The eating disorders (ED) anorexia nervosa (AN) and bulimia nervosa (BN), and eating disorder not otherwise specified affect about 5–7 % of women of childbearing age. There is an increasing literature from large cohorts and data registers showing clear effects of active and past ED on obstetric outcomes(Reference Micali, Simonoff and Treasure1–Reference Sollid, Wisborg and Hjort4). These studies all point to a lower birth weight, especially in the offspring of women with lifetime AN. Although previous studies have shown that ED symptoms tend to improve during pregnancy(Reference Micali, Simonoff and Treasure1, Reference Bulik, Von Holle and Siega-Riz2), only one study to date has investigated exact dietary intakes in pregnancy in women with active/past (before pregnancy) ED(Reference Siega-Riz, Haugen and Meltzer5). This large study on a population sample of Norwegian pregnant women showed differences in early pregnancy in food group intakes in women with active BN compared with controls and higher nutritional intakes in women with active binge eating disorder. Although this well-conducted study investigated women with active or last 6 months' history of binge eating disorder and BN, no subjects with active/past AN were available for study.
Clinical experience highlights an uncertainty among women with active/past ED in relation to nutritional requirements in pregnancy. We previously highlighted an important role for pre-pregnancy BMI in mediating the effect of maternal AN on birth weight(Reference Micali, Simonoff and Treasure1). Dietary consumption in pregnancy might also be an important factor contributing to adverse fetal outcomes(Reference Micali and Treasure6). Therefore, clarifying differences in nutrient and food group consumption and overall dietary patterns in pregnancy in women with lifetime ED could help clarify the risk pathways.
The present study aims to build on and complement the evidence provided by Siega-Riz et al. (Reference Siega-Riz, Haugen and Meltzer5) by investigating the frequency of consumption of various food groups and quality of intake (macronutrient intakes) in a large general population cohort based in the UK: the Avon Longitudinal Study of Parents and Children (ALSPAC). Due to the high correlation and biological interaction between food and nutrient intakes, we were also interested in studying dietary patterns of pregnant women with lifetime ED obtained using principal components analysis to identify underlying dietary patterns in the data.
We hypothesised that women with lifetime ED would have a lower energy, lower sugar and lower fat intake compared with unexposed women.
Subjects and methods
Participants and procedures
The ALSPAC is a longitudinal, prospective study designed to examine the effects of environment, genetics and other factors on health and development(Reference Golding, Pembrey and Jones7). All pregnant women living in the geographical area of Avon, UK, who were expected to deliver their baby between 1 April 1991 and 31 December 1992, were recruited; 14 472 women were enrolled. Primary data collection was via self-completion questionnaires.
At 12 weeks of gestation, women were asked whether they had ever had any psychiatric problems, including depression, schizophrenia, alcohol abuse, AN, BN or any other disorder. Women were excluded from the present study if they had not completed this questionnaire (n 2019), or if they reported in this questionnaire any lifetime history of psychiatric problems (other than ED) only (n 1166), as detailed data on psychopathology in this group were lacking. Women were excluded if they had non-singleton pregnancies (n 199) and if they had a miscarriage for the current pregnancy (n 36).
The final sample eligible for the study was 11 052 women. Information on ethnicity and maternal education was missing for 691 women (6·2 %). Complete baseline and dietary data were available for 10 137 (91·7 %) women.
Women were divided into four groups according to their answer to the 12 weeks questionnaire on having ever had AN, BN or both(Reference Micali, Simonoff and Treasure1): women with a self-reported lifetime AN (n 151); women with a self-reported lifetime BN (n 186); women who reported both lifetime AN and BN (n 77); a group of unexposed women representative of the general population (n 9723).
Measures
Sociodemographic and weight and height data were obtained by a self-completion questionnaire at 12, 18 and 32 weeks of gestation; BMI was calculated as pre-pregnant weight (kg)/height (m2). Dietary information was collected via the FFQ, completed by women at 32 weeks of gestation.
Food group consumption
The FFQ contained a set of questions enquiring about the frequency of consumption of a wide variety of foods and drinks. The women were given the following options to indicate how often they were currently consuming a variety of food types: (1) never or rarely; (2) once in 2 weeks; (3) 1–3 times/week; (4) 4–7 times/week; (5) more than once daily. The women were also asked to record how many cups of tea or coffee, the number of glasses of cola and the number of slices of bread they usually consumed daily. Dietary supplements were not included in the FFQ. The frequency of consumption data were numerically transformed into times consumed per week, in order to apply quantitative meaning to the frequency categories, as follows: (1) 0, (2) 0·5, (3) 2, (4) 5·5 and (5) 10 times/week.
In order to summarise dietary data, we grouped foods, where it could be done in a meaningful way. For example, frequencies of eating red meat, poultry, sausages/burgers and pies/pasties were summed into a variable called ‘all meat’. ‘Fish’ included white fish, oily fish and shellfish. ‘Potatoes’ included chips, roast potatoes and boiled or baked potatoes, and ‘vegetables’ included peas, maize or similar, cabbage or similar, green vegetables, carrots, root vegetables and salad. Fruit and fruit juice were combined. ‘Sweets/chocolate/cakes/biscuits’ included puddings, cakes or buns, chocolates, sweets, biscuits and chocolate bars. Soya and meat substitutes were also summed together. Women were also asked to indicate whether they were vegetarian or ate meat.
Dietary patterns
Dietary patterns were obtained from the FFQ (detailed above) using principal components analysis. This has been described in detail elsewhere(Reference Northstone, Emmett and Rogers8). Briefly, the number of components best representing the data was primarily chosen on the basis of a scree plot and the interpretability of the components. Women were excluded from the principal components analysis if they had more than ten dietary items missing. If ten or fewer items were missing, we made the assumption that the women did not consume those items and they were given a value of 0. A component score was created for each woman for each of the components identified, calculated by multiplying the factor loadings by the corresponding standardised value for each food and summing across the food items. Each score had a mean of 0 and a higher score indicated closer adherence to that dietary pattern. The following five components were obtained: ‘health conscious’ (high loadings for salad, fruit, rice, pasta, oat and bran-based breakfast cereals, fish, pulses, fruit juices and non-white bread); ‘traditional’ (high consumption of all types of vegetables and red meat and poultry); ‘processed’ (high intakes of high-fat processed foods, such as meat pies, sausages and burgers, fried foods, pizza, chips and baked beans); ‘confectionery’ (high intakes of foods with high sugar content such as chocolate, sweets, biscuits, cakes and other puddings); ‘vegetarian’ (high loadings for meat substitutes, pulses, nuts and herbal teas, and high negative loadings for red meat and poultry).
Nutrients
Daily nutrient intakes were estimated from the FFQ using the 5th edition of McCance and Widdowson's ‘The Composition of Food’ and supplements based on standard portion sizes; detailed information on the methodology has been published elsewhere(Reference Rogers and Emmett9). Previous analysis of these data(Reference Rogers and Emmett9) showed this questionnaire to produce mean nutrient intakes similar to those obtained for women in the British National Diet and Nutritional survey for adults(Reference Gregory, Foster and Tyler10). Data for analysis of nutrient intakes were missing for forty-eight women (0·5 %) and available for 10 073 women (91·1 %). Missingness was independent of exposure status.
Statistical analyses
Preliminary analyses determined distribution patterns of all variables; variables that were found to be skewed underwent logarithmic transformation before analyses. Group comparisons used parametric (one-way ANOVA) tests as appropriate, after testing for normality. Multinomial and binary logistic regression models examined predictors of categorical and binary outcomes, respectively.
Potential covariates previously found to be associated with relevant outcomes in this sample(Reference Northstone, Emmett and Rogers8) and likely to influence outcomes were first tested in bivariate models and then included in multivariate models if associated with the outcomes. The final model accounted for main effects of each covariate. All analyses of nutrient intakes were adjusted for total energy intake. All analyses were performed using Stata 10 for Windows(11). All statistical tests presented are two-tailed. Statistical significance was defined as a P value of less than 0·05.
Multiple testing
In order to take into account the effect of multiple testing on potential chance significant findings, two steps were taken. First, women with lifetime ED were compared with controls on selected outcomes; only if a difference was found were comparisons extended to ED subgroups (AN, BN and AN+BN). At this second stage of analyses, the Bonferroni–Holm(Reference Holm12) procedure was used to adjust for multiple testing. This procedure is more powerful and less conservative than a Bonferroni correction and allows adjusting P values obtained during multiple testing.
We did not apply a method to adjust for multiple testing across outcomes, as this would have reduced power to an unacceptable level, and as this is an exploratory study. Instead, we have provided significance for different levels of type I error(Reference Bender and Lange13).
Ethics
The present study was approved by the Institute of Psychiatry Ethics Committee (reference no. 110/02), the ALSPAC Law and Ethics Committee and the Local Research Ethic Committees. All women gave written informed consent for participation in the study.
Results
Sociodemographic data
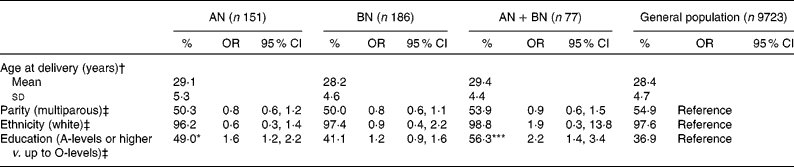
Age at delivery, parity and ethnic distribution were comparable across the groups. Women with lifetime AN and AN+BN were more likely to have obtained A-levels compared with unexposed women (OR 1·6, 95 % CI 1·2, 2·2; OR 2·2, 95 % CI 1·4, 3·4, respectively; see Table 1).
Table 1 Sociodemographic data: comparisons between index groups and unexposed women from ANOVA and logistic regression (Percentages, odds ratios and 95 % confidence intervals)

AN, anorexia nervosa; BN, bulimia nervosa; AN+BN, both anorexia nervosa and bulimia nervosa.
Values are shown for comparisons between each index group and unexposed women: *P < 0·05, ***P < 0·001 (ANOVA, binary logistic regression).
† F = 2·4 (df = 3); ANOVA.
‡ Logistic regression.
Food group consumption and dietary patterns
Women with ED were 2·8 times more likely to describe themselves as vegetarian (OR 2·8, 95 % CI 2·1, 3·8) compared with unexposed women; all subgroups differed from the unexposed (AN: OR 2·8, 95 % CI 1·7, 4·4; BN: OR 2·3, 95 % CI 1·4, 3·7; AN+BN: OR 4·3, 95 % CI 2·3, 7·7).
Women with ED consumed less meat than unexposed women (b coefficient − 1·6, 95 % CI − 2·1, − 1·1; P < 0·001 (respectively AN: − 1·8, 95 % CI − 2·6, − 1·0; BN: − 1·4, 95 % CI − 2·1, − 0·7; AN+BN: − 1·8, 95 % CI − 2·9, − 0·6), and fewer potatoes (b coefficient − 0·6, 95 % CI − 0·9, − 0·2). However, they ate more pulses (b coefficient 0·2, 95 % CI 0·1, 0·3). Women with lifetime ED were more likely to consume soya and soya products (b coefficient 0·3, 95 % CI 0·2, 0·5; see Table 2).
Table 2 Food group consumption (times/week) and dietary patterns from the FFQ: adjusted‡ ANOVA§ (B coefficients and 95 % confidence intervals) and logistic regression (odds ratios and 95 % confidence intervals) by group

ED, eating disorders; AN, anorexia nervosa; BN, bulimia nervosa; AN+BN, both anorexia and bulimia nervosa.
Subgroup differences were not calculated if no statistically significant difference was present when comparing ED v. unexposed: **P ≤ 0·01, ***P ≤ 0·001.
† Subgroup differences were not calculated if no statistically significant difference was present when comparing ED v. unexposed (P < 0·1).
‡ Adjusted for maternal age, maternal education and ethnicity.
§ Comparing each group with the unexposed.
Women with ED consumed greater amounts of bread per d: on average, 1·1 slices/d more than women in the general population (b coefficient 1·1, 95 % CI 0·2, 1·9); this difference was evident for women with BN (b coefficient 1·9, 95 % CI 0·6, 3·2).
Women with ED scored higher on the ‘vegetarian pattern’ compared with unexposed women (b coefficient 0·3, 95 % CI 0·2, 0·4; see Table 2). This association was consistent across the three ED subgroups. Women with lifetime AN+BN had higher scores on the ‘traditional’ dietary pattern (b coefficient 0·3, 95 % CI 0·1, 0·5) and the ‘health conscious’ pattern (b coefficient 0·2, 95 % CI 0·01, 0·4) compared with the unexposed. Women with lifetime AN scored higher on the ‘traditional’ dietary pattern (b coefficient 0·2, 95 % CI 0·02, 0·3) compared with unexposed women (see Table 2; Table S1, available online).
Use of fats, milk and caffeine
Women with lifetime ED were less likely to use butter compared with unexposed women (OR 0·7, 95 % CI 0·5, 0·8); however, they used margarine, low-fat spread and vegetable oil similarly to unexposed women. A lower use of full-fat milk (OR 0·7, 95 % CI 0·6, 0·9) and a higher use of skimmed milk (OR 1·4, 95 % CI 1·1, 1·8) and soya milk (OR 3·1, 95 % CI 1·8, 5·2) compared with unexposed women were also evident.
A high weekly caffeine intake (>2500 mg) was more common in women with lifetime ED than in the controls (OR 1·9, 95 % CI 1·3, 2·8); women with both lifetime AN and lifetime AN+BN differed from unexposed women (OR 2·6, 95 % CI 1·4, 4·8 and OR 2·7, 95 % CI 1·1, 7·0) (see Table 3).
Table 3 Use of fats, milk and caffeine: odds ratios‡ (95 % confidence intervals) from logistic regression for each group compared with unexposed women (Odds ratios and 95 % confidence intervals)

ED, eating disorders; AN, anorexia nervosa; BN, bulimia nervosa; AN+BN, both anorexia nervosa and bulimia nervosa.
Subgroup differences were not calculated if no statistically significant difference was present when comparing ED v. unexposed: *P ≤ 0·05, **P ≤ 0·01, ***P ≤ 0·001.
† Subgroup differences were not calculated if no statistically significant difference was present when comparing ED v. unexposed (P ≤ 0·1).
‡ Adjusted for maternal age, maternal education and ethnicity.
Nutrient intake
In relation to energy, carbohydrate, fat and protein consumption, no differences were highlighted between women with ED and unexposed women (see Table 4).
Table 4 Nutrients: adjusted†B coefficients from ANOVA (B Coefficients and 95 % confidence intervals)

ED, eating disorder.
* P < 0·1.
† Adjusted for maternal education, age and ethnicity.
‡ Carbohydrate, fat and protein intakes were also adjusted for total energy.
Across the ED subgroups, only minor differences in macronutrient consumption were observed (see Table S2, available online): a lower sugar and non-milk extrinsic sugar, and a higher polyunsaturated fat intake in women with lifetime BN. A lower intake of saturated fat was observed in women with lifetime AN and AN+BN. A higher fibre (NSP) intake was also observed in women with lifetime AN, BN and AN+BN.
Mineral and vitamin intakes were not compromised in women with lifetime ED. Across the subgroups, all index groups had higher intakes of Mg and Se; women with AN and AN+BN of Fe and K; and women with AN+BN of P, Ca and Zn compared with unexposed women.
Vitamin intake was higher for folate (b coefficient 10·9, 95 % CI 2·7, 19·1) in women with AN; lower in log-transformed retinol (b coefficient − 0·1, 95 % CI − 0·2, − 0·02) and higher in thiamin (b coefficient 0·1, 95 % CI 0·05, 0·2), vitamin C (b coefficient 14·4, 95 % CI 7·3, 21·4) and folate (b coefficient 24·2, 95 % CI 12·8, 35·6) in women with lifetime AN+BN compared with unexposed women (see Table S2, available online). Women with lifetime BN had a slightly higher vitamin E intake compared with the unexposed (b coefficient 0·1, 95 % CI 0·02, 0·15).
Discussion
This is the first study to investigate various aspects of dietary intake in the third trimester of pregnancy in women with lifetime ED using data from a large longitudinal general population study.
The present results show that, in general, dietary intake in pregnancy in women with lifetime ED was comparable to women from the general population without other psychiatric disorders. Some differences were highlighted in relation to food group consumption, in that women with lifetime ED consumed less meat and fewer potatoes in favour of soya products and pulses.
In relation to dietary patterns, there was a trend towards women with lifetime ED scoring higher on the ‘health conscious’ and ‘traditional’ dietary pattern, than unexposed women. In addition, women with ED scored higher on the ‘vegetarian’ dietary pattern and were three times more likely to describe themselves as vegetarians. This was reflected in more common use of soya milk in women with ED than in the unexposed. Siega-Riz et al. (Reference Siega-Riz, Haugen and Meltzer5) showed a lower intake of high-fat meats in the second trimester of pregnancy among women with BN before and during pregnancy in a large general population study.
Although there is evidence that a vegetarian diet is adequate in pregnancy and might actually result in positive infant health outcomes(Reference Craig and Mangels14), maternal soya intake in pregnancy has been shown to have some effects on sexual maturation in animal studies(Reference Guerrero-Bosagna, Sabat and Valdovinos15), in particular earlier pubertal maturation in female offspring.
Women with lifetime ED were also less likely to use butter and drink full-fat milk (in favour of skimmed and soya milk). This is consistent with the ‘vegetarian’ dietary pattern highlighted.
Interestingly, women with lifetime ED were almost twice as likely to consume ≥ 2500 mg caffeine/week (this is equivalent to about twenty-five cups of brewed coffee/week) compared with healthy women and higher than the recommendations of the Department of Health for pregnant women. This was particularly true for women with lifetime AN and AN+BN. The Food Standards Agency recommends that pregnant women should drink less than 200 mg caffeine/d(16). Siega-Riz et al. (Reference Siega-Riz, Haugen and Meltzer5) showed a slight increase in mean coffee consumption in women with binge eating disorder during pregnancy in their study.
Previous studies on clinical and non-clinical samples have highlighted increased consumption of caffeine in women with ED and have suggested that this might be driven by a wish to suppress appetite and/or for its stimulating properties(Reference Hart, Abraham and Luscombe17, Reference Striegel-Moore, Franko and Thompson18). Caffeine crosses the placenta and there is increasing evidence that caffeine intake in pregnancy affects the fetus. A detrimental effect of caffeine has been shown on fetal weight and length(Reference Parazzini, Chiaffarino and Chatenoud19, Reference Bakker, Steegers and Obradov20).
Few differences were evident in relation to macronutrient intake in women with lifetime ED. Across maternal ED, there was evidence of a lower consumption of non-milk extrinsic sugars (for women with BN and AN+BN) and saturated fat (particularly women with lifetime AN and AN+BN) and more fibre (NSP) compared with unexposed women.
There was very little difference in terms of micronutrient intakes in pregnancy in women with lifetime ED compared with healthy women, consistent with Siega-Riz et al. (Reference Siega-Riz, Haugen and Meltzer5). In particular, no deficiencies in relation to vitamin and mineral intakes were shown for women with lifetime ED. This is encouraging, as it suggests that the majority of women with lifetime ED have adequate vitamin and mineral intake in pregnancy, and it might be secondary to general improved nutrition (as highlighted by the lack of difference in macronutrient intake).
The present study relied on a large general population sample of pregnant women, and it is the first study to investigate nutrition in the third trimester of pregnancy in women with lifetime ED. The sample of pregnant women was representative of the general population in the geographical area under study.
The main weakness in the present study relates to the ascertainment of ED history. This was obtained by self-report. In particular, one question ascertained the lifetime history of ED, resulting in a possible misclassification of exposure. It is likely that this represents an underestimate of ED, given the tendency of screening measures for ED in community samples to miss cases. However, self-reported ED was validated using behavioural and cognitive characteristics in this sample(Reference Micali, Simonoff and Treasure1). Moreover, self-report of ED has been shown to be comparable to longer and widely used ED screening instruments for screening purposes in general population studies(Reference Keski-Rahkonen, Sihvola and Raevuori21). In addition, the prevalence of self-reported ED (3·7 %) in this sample is consistent with general population lifetime prevalence.
Unfortunately, it was not possible to ascertain active v. lifetime ED during pregnancy.
The other major weakness was that standard portion sizes were used to assess nutrient intakes from the FFQ. Clinically, women with past/active ED often report difficulties in determining what was a good portion size. It is possible that women with lifetime AN ate small portions and those with BN binged on certain foods. The FFQ used for the present study only asked about the frequency with which each food group was consumed. It is possible that women with ED, albeit eating a good-quality diet, did not eat enough in quantity.
Conclusions
Despite being more likely to be vegetarian and consuming less meat, women with lifetime ED had similar protein, fat and carbohydrate intake compared with unexposed women. In general, good macronutrient and vitamin and mineral intake were apparent. This is encouraging and suggests that the quality of maternal diets in pregnancy was reasonable in women with lifetime ED. There was some evidence that they made more healthy choices compared with unexposed women. However, we were unable to account for portion sizes. Future analyses should investigate actual gestational weight gain in this sample. A high caffeine intake was shown in women with ED and future research needs to clarify the impact of this on fetal growth.
Acknowledgements
We are extremely grateful to all the families who took part in this study, the midwives for their help in recruiting them, and the whole ALSPAC team, which includes interviewers, computer and laboratory technicians, clerical workers, research scientists, volunteers, managers, receptionists and nurses. The UK Medical Research Council, the Wellcome Trust and the University of Bristol provided core support for the ALSPAC. This study was funded by the National Alliance for Schizophrenia and Depression, the Department of Health via the ARIADNE programme (Applied Research into Anorexia Nervosa and Not Otherwise Specified Eating Disorders) and a Department of Health National Institute for Health Research (NIHR) Programme Grant for Applied Research (reference no. RP-PG-0606-1043) to U. Schmidt, J. Treasure, K. Tchanturia, H. Startup, S. Ringwood, S. Landau, M. Grover, I. Eisler, I. Campbell, J. Beecham, M. Allen, G. Wolff. The views expressed herein are not necessarily those of the Department of Health/NIHR. N. M. devised the study hypothesis, analysed the data and wrote the paper, and has primary responsibility for the final content. K. N. and P. E. were involved in the original cohort design, performed the data extraction and the general data analyses, helped with the data interpretation and revised the manuscript. U. N. advised on the data analytic strategies and advised on the interpretation of the data analyses. J. L. T. supervised the data analyses, the interpretation of the data and revised the manuscript. The authors declare that there are no conflicts of interest.
Supplementary tables are available online at http://www.journals.cambridge.org/bjn