Scientific interest in anxiety disorders in young children has increased in the past decade. Anxiety symptoms and diagnostic categories in young children resemble those in older children (Mian, Godoy, Briggs-Gowan, & Carter, Reference Mian, Godoy, Briggs-Gowan and Carter2012). In young children, prevalence rates of anxiety disorders ranging from 9.4% (Egger & Angold, Reference Egger and Angold2006) up to 22.2% (Paulus, Backes, Sander, Weber, & von Gontard, Reference Paulus, Backes, Sander, Weber and von Gontard2015) have been found. Unfortunately, anxiety disorders are often unrecognised in young children because anxious children are considered to be shy, cooperative and compliant (Albano, Chorpita, & Barlow, Reference Albano, Chorpita, Barlow, Mash and Barkley2003). If left unnoticed and untreated, this can have harmful consequences, as early-onset anxiety disorders can become chronic (Essau, Lewinsohn, Lim, Ho, & Rohde, Reference Essau, Lewinsohn, Lim, Ho and Rohde2018; Essau, Lewinsohn, Olaya, & Seeley, Reference Essau, Lewinsohn, Olaya and Seeley2014). Research also shows that a diagnosis of an anxiety disorder in early childhood predicts anxiety and depression in adolescence, and significantly increases the risk of having psychiatric disorders in adolescence and adulthood (Bienvenu & Ginsburg, Reference Bienvenu and Ginsburg2007; Bittner et al., Reference Bittner, Egger, Erkanli, Costello, Foley and Angold2007; Copeland et al., Reference Copeland, Adair, Smetanin, Stiff, Briante, Colman and Angold2013; Copeland, Shanahan, Costello, & Angold, Reference Copeland, Shanahan, Costello and Angold2009; Costello, Mustillo, Erkanli, Keeler, & Angold, Reference Costello, Mustillo, Erkanli, Keeler and Angold2003; Essau et al., Reference Essau, Lewinsohn, Lim, Ho and Rohde2018; Moffitt et al., Reference Moffitt, Caspi, Harrington, Milne, Melchior, Goldberg and Poulton2007). Moreover, childhood anxiety disorders are associated with social (de Lijster et al., Reference de Lijster, Dieleman, Utens, Dierckx, Wierenga, Verhulst and Legerstee2018), school (de Lijster et al., Reference de Lijster, Dieleman, Utens, Dierckx, Wierenga, Verhulst and Legerstee2018; Mychailyszyn, Mendez, & Kendall, Reference Mychailyszyn, Mendez and Kendall2010), and familial (Towe-Goodman, Franz, Copeland, Angold, & Egger, Reference Towe-Goodman, Franz, Copeland, Angold and Egger2014) problems. Anxiety disorders have a significant impact on societal costs due to poorer academic outcomes, financial dependence and unemployment in adulthood (Barrett, Cooper, & Teoh, Reference Barrett, Cooper and Teoh2014; Bodden, Dirksen, & Bögels, Reference Bodden, Dirksen and Bögels2008).
Considering these alarming outcomes, early intervention is urgently needed. Providing intervention at a young age has important advantages. First, anxious thoughts and behaviours may be easier to modify in younger children, as anxiety symptoms are likely to be less ingrained and neuroplasticity in young children is high (Hirshfeld-Becker & Biederman, Reference Hirshfeld-Becker and Biederman2002). Second, intervening early in the lifespan can minimise the impact of anxiety symptoms on the development and future of the child (Connolly & Bernstein, Reference Connolly and Bernstein2007; Donovan & March, Reference Donovan and March2014; Fox et al., Reference Fox, Warner, Lerner, Ludwig, Ryan, Colognori and Brotman2012; Hirshfeld-Becker & Biederman, Reference Hirshfeld-Becker and Biederman2002)
Despite the serious need for an evidence-based intervention for anxious young children, only a few studies have been conducted into interventions for this age group (Anticich, Barrett, Silverman, Lacherez, & Gillies, Reference Anticich, Barrett, Silverman, Lacherez and Gillies2013; Fox et al., Reference Fox, Warner, Lerner, Ludwig, Ryan, Colognori and Brotman2012; von Klitzing, Dohnert, Kroll, & Grube, Reference von Klitzing, Dohnert, Kroll and Grube2015; for a complete overview, see Fisak & Barrett, Reference Fisak and Barrett2019). An intervention that has been developed for 4- to 7-year-old children with anxiety disorders is the cognitive behavioural Fun FRIENDS program (Barrett, Reference Barrett2007b; Pahl & Barrett, Reference Pahl and Barrett2007). The Fun FRIENDS program is an adaptation for young children of the evidence-based FRIENDS for Life program (Barrett & Turner, Reference Barrett and Turner2000), which was based on the Coping Cat program (Kendall, Reference Kendall1994). The Fun FRIENDS program aims to increase children's emotional resilience, social-emotional skills and coping skills, and to reduce emotional and behavioural problems. The program consists of 12 group sessions and is provided in a play-based manner, based on an experiential learning approach.
Until now, only two studies have examined the effectiveness of Fun FRIENDS delivered as a preventive program (Anticich et al., Reference Anticich, Barrett, Silverman, Lacherez and Gillies2013; Pahl & Barrett, Reference Pahl and Barrett2010). In addition, three studies have studied the outcomes of Fun FRIENDS delivered as a treatment program for young children with clinical internalising symptoms or anxiety disorders (Barrett, Fisak, & Cooper, Reference Barrett, Fisak and Cooper2015; Carlyle, Reference Carlyle2014; Fisak, Gallegos-Guarjardo, Verreynne, & Barrett, Reference Fisak, Gallegos-Guajardo, Verreynne and Barrett2018).
Both prevention studies were randomised controlled trials in which Fun FRIENDS was delivered in a universal, classroom-based manner by psychology students or classroom teachers. In the first prevention study (N = 263, mean age = 4.56, SD = 0.51; Pahl & Barrett, Reference Pahl and Barrett2010), both the Fun FRIENDS intervention group and the waitlist control group showed comparable improvements on parent reports of anxiety, behavioural inhibition, and social-emotional strength. Regarding teacher reports, however, the Fun FRIENDS intervention group showed greater improvements than the waitlist control group in behavioural inhibition and social-emotional strength, especially for girls. For ethical reasons, 12-month follow-up assessments were only completed for the intervention group. From preintervention to 12-month follow-up, the Fun FRIENDS intervention group showed improvements in anxiety and social-emotional strength, and for girls in behavioural inhibition. In the second prevention study (N = 488, age range 4–7 years, mean age = 5.42, SD = 0.67; Anticich et al., Reference Anticich, Barrett, Silverman, Lacherez and Gillies2013), children who had participated in Fun FRIENDS showed greater improvements in behavioural and emotional strength, and behavioural inhibition than children from the active control group (cognitive behavioural ‘You Can Do It’ program; Ashdown & Bernard, Reference Ashdown and Bernard2011) and waitlist control group.
As for Fun FRIENDS as a treatment program, the first study consisted of a pilot study (N = 6, age range 4–7 years). This study suggested that Fun FRIENDS was effective in reducing anxiety of young children referred to a mental health service for anxiety symptoms (Carlyle, Reference Carlyle2014). The second treatment study was an open trial that included young children (N = 31, age range 5–7, mean age = 5.68, SD = 0.54) who were diagnosed with one or more anxiety disorders (Barrett et al., Reference Barrett, Fisak and Cooper2015). From preintervention to immediate postintervention, significant improvements in anxiety symptoms, shyness, number of anxiety disorder diagnoses and resilience were found. These results were maintained at 12-month follow-up. The third treatment study was also an open trial targeting young children (N = 178, age range 5–7, mean age = 5.27, SD = 0.93) with internalising symptoms (Fisak et al., Reference Fisak, Gallegos-Guajardo, Verreynne and Barrett2018). Their parents simultaneously received a resilience-building program (Barrett, Reference Barrett2012a, Reference Barrett2012b). For child outcomes, from preintervention to immediate postintervention, significant reductions in internalising symptoms and significant improvements in resilience were found.
Considering these promising outcomes, the Fun FRIENDS protocol was translated and adjusted for the Netherlands (Utens, Reference Utens2011a, Reference Utens2011b). The aim of the current study is to examine whether young children with anxiety disorders show fewer anxiety symptoms after participating in the Dutch version of the Fun FRIENDS program, and to identify predictors of treatment progress. We thereby aim to add to the limited available knowledge concerning evidence-based treatment for young children with anxiety disorders and to contribute to the cross-cultural knowledge regarding this innovative cognitive behavioural program. We hypothesised that anxiety symptoms and the number of anxiety diagnoses would decrease after participating in Fun FRIENDS.
Methods
Participants
Children who were 4–8 years old and met the Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV; American Psychiatric Association, 1994) diagnostic criteria for at least one anxiety disorder were eligible to participate in the Fun FRIENDS program. DSM-IV anxiety disorder criteria were assessed based on the parent version of the Anxiety Disorders Interview Schedule for Children (ADIS-C; Silverman, Saavedra, & Pina, Reference Silverman, Saavedra and Pina2001). All participants were referred to the Department of Child and Adolescent Psychiatry of the Erasmus Medical Center – Sophia Children's Hospital in Rotterdam between December 2008 and November 2013. Children with an IQ below 70 or a diagnosis of a posttraumatic stress disorder without a comorbid anxiety disorder were excluded from participation. In total, 28 children participated in the Fun FRIENDS program. Participant characteristics can be found in Table 1. As parental education is associated with persistence and severity of mental disorders (McLaughlin et al., Reference McLaughlin, Breslau, Green, Lakoma, Sampson, Zaslavsky and Kessler2011), we have presented maternal education levels. Participants’ primary anxiety disorder diagnoses are shown in Table 2 and all anxiety disorder diagnoses are shown in Table 3.
Table 1. Participant characteristics.

Note: aConforms to Dutch classification system (Centraal Bureau voor de Statistiek [Statistics Netherlands], 2017).
Table 2. Participants’ primary anxiety disorder diagnoses based on the ADIS-C at preintervention and postintervention
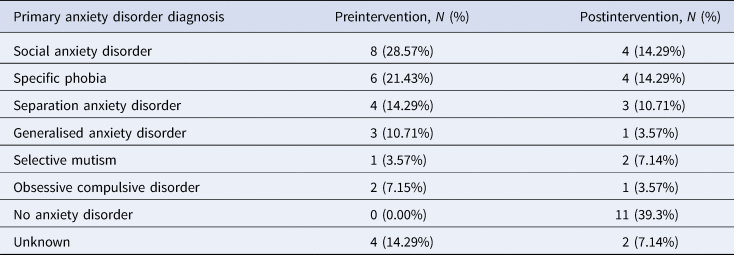
Table 3. All anxiety disorder diagnoses based on the ADIS-C at preintervention and postintervention

Procedure
This retrospective open trial study was conducted using a one-group pretest-posttest design. All parents were asked to complete assessments as part of the routine intake procedure (preintervention) and directly after the Fun FRIENDS intervention (postintervention). As the Fun FRIENDS program was provided within the framework of regular treatment, assessments were completed as usual and data were analysed retrospectively, this study was not subject to the Dutch Medical Research Involving Human Subjects Act. The local research ethics committee was informed about the study and confirmed that full ethical approval of the study was not required. Participants were informed that collected data would be used anonymously in scientific research and that they could always opt out without any consequences for the treatment of their child.
Treatment
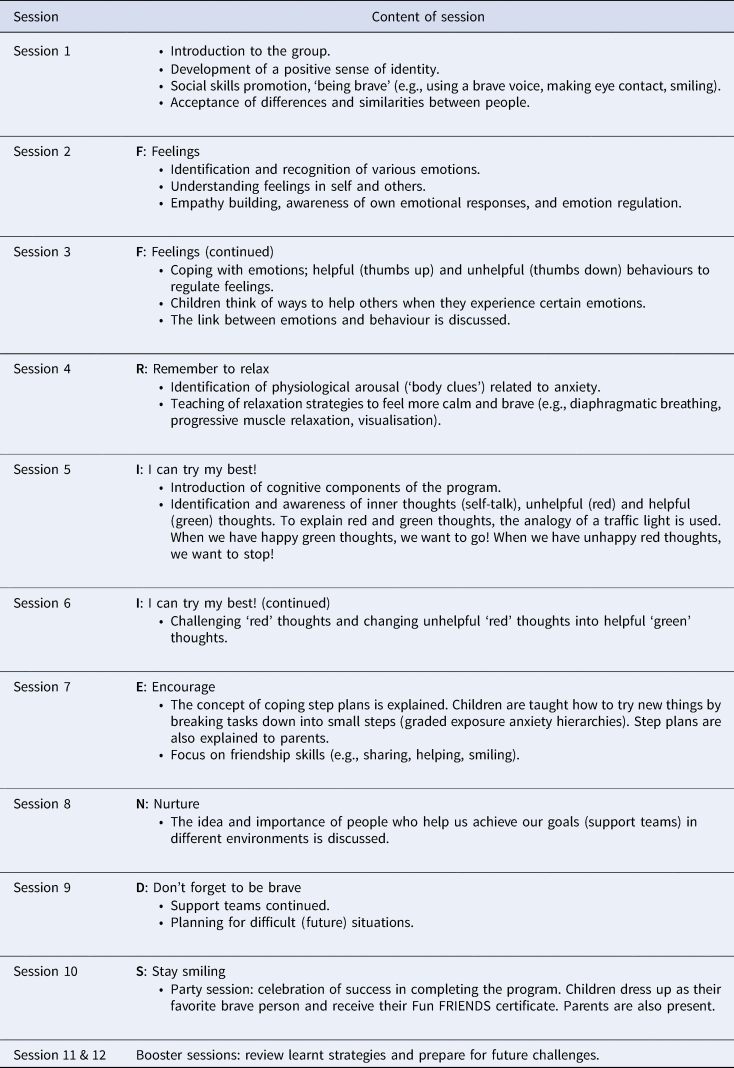
All children participated in the Dutch version of the Fun FRIENDS program (Barrett, Reference Barrett2007b; Utens, Reference Utens2011b). The program was delivered to seven consecutive treatment groups. Five groups consisted of 4 children, one group of 5 children, and one group of 3 children (n = 28). The children received 12 weekly 1.5-hour sessions. On average, the program was delivered over a time period of 3.5 months. All sessions were led by two licensed, experienced psychologists. One of them received training from the developer of the Fun FRIENDS program. At each session, a master's student in psychology was present to make observations, take notes, and assist the psychologists. The content of each session is described in Table 4. During the last 15 minutes of each session, the master's student observed the children during free play, while in a separate room, the psychologists gave the group of parents further information about the home assignments and the exercises performed during the session. The last child session was a booster session in which parents were present and actively involved. All families received a Fun FRIENDS Workbook (Barrett, Reference Barrett2007a; Utens, Reference Utens2011a), which contained home assignments and additional information about the program.
Table 4. Outline of Fun FRIENDS sessions
Measures
Anxiety Disorders Interview Schedule for Children (ADIS-C)
The ADIS-C (Siebelink & Treffers, Reference Siebelink and Treffers2001; Silverman et al., Reference Silverman, Saavedra and Pina2001) is a semi-structured interview that was used to assess the presence and severity of DSM-IV anxiety disorders in children and adolescents. The ADIS-C was conducted with parents to assess the following DSM-IV diagnoses: selective mutism, generalised anxiety disorder, social phobia, specific phobia, separation anxiety disorder, panic disorder, agoraphobia, obsessive-compulsive disorder, and posttraumatic stress disorder. For each diagnosis confirmed based on the interview, the parent was asked to rate to what extent the symptoms interfered with the child's daily life on a 9-point scale (i.e., 0–8, higher scores indicating a higher level of interference). Subsequently, the interviewer rated the level of interference on the same 9-point scale, yielding the Clinician Severity Rating (CSR). A CSR of 4 or higher indicates that a DSM-IV diagnosis can be confirmed and assigned. Strong interrater reliability, retest reliability and concurrent validity have been found for the ADIS-C (Lyneham, Abbott, & Rapee, Reference Lyneham, Abbott and Rapee2007; Silverman et al., Reference Silverman, Saavedra and Pina2001). Preintervention and postintervention interviews were conducted by a different interviewer. All ADIS-C interviews were administered by trained psychologists or trained master's students in psychology. To ensure that all interviewers conducted reliable and valid scoring, the master's students were thoroughly trained by observing live and videotaped interviews. Moreover, they received regular supervision regarding their ADIS-C interviews by their supervising experienced clinical psychologist or psychiatrist, and all ADIS-C interviews were reviewed and discussed in multidisciplinary meetings.
Child Behavior Checklist (CBCL)
The CBCL 1½−5 (100 items; for 5-year-olds; Achenbach & Rescorla, Reference Achenbach and Rescorla2001) and CBCL/6-18 (120 items; for 6- to 8-year-olds; Achenbach & Rescorla, Reference Achenbach and Rescorla2001) were completed by parents to assess emotional and behavioural problems in children before and after the intervention. Response categories range from 0 to 2, higher scores indicating more problems. The CBCL yields two broadband scales of externalising and internalising behaviours and an overall total score. Furthermore, both the CBCL 1½–5 and the CBCL/6–18 encompass the Anxious/Depressed syndrome scale and the DSM-oriented Anxiety Problems scale. Adequate psychometric properties have been found (Achenbach et al., Reference Achenbach, Becker, Dopfner, Heiervang, Roessner, Steinhausen and Rothenberger2008).
Statistical Analysis
First, differences in baseline characteristics between children with complete assessments and children with incomplete assessments were examined using independent samples t tests for continuous data and chi-squared tests or Fisher's exact tests for categorical data.
Second, for the ADIS-C, a Wilcoxon signed-rank test was computed to assess the difference between the number of anxiety disorders at preintervention and postintervention. The difference between average preintervention and postintervention interference scores rated by parents was examined through a paired samples t test. Unfortunately, too many CSRs were missing at preintervention and postintervention to complete statistically warranted reliable analyses on these data. CSRs were missing due to the retrospective design of the study. As all assessments were conducted as part of regular clinical care, data were not systematically entered into a scientific database. Moreover, changes in digital medical file systems caused logistical difficulties in retrieving a sufficient number of CSRs.
Third, CBCL scores were standardised using t scores as two different versions were used (i.e., CBCL 1½−5 and CBCL/6-18). Differences between preintervention and postintervention CBCL scores were examined using paired samples t tests.
Finally, it was examined whether sex, age, or preintervention anxiety scores independently predicted treatment progress. Treatment progress was calculated by subtracting postintervention anxiety problem scores on the CBCL from preintervention anxiety problem scores (primary outcome). To examine whether children's sex predicted treatment progress, an independent samples t test was conducted. To examine whether children's age at the start of participation in the Fun FRIENDS program predicted treatment progress, a simple linear regression analysis was performed. Another simple linear regression analysis was performed to examine whether preintervention scores on the anxiety problems subscale of the CBCL predicted treatment progress.
Results
Mean scores are presented in Table 5. One child missed 6 out of 12 sessions due to logistical reasons, but was included in the analyses according to the intention-to-treat principle. For two children, both the ADIS-C and the CBCL were not fully completed. Therefore, these children were excluded from all analyses.
Table 5. Mean scores for outcome variables and statistical comparisons between preintervention and postintervention
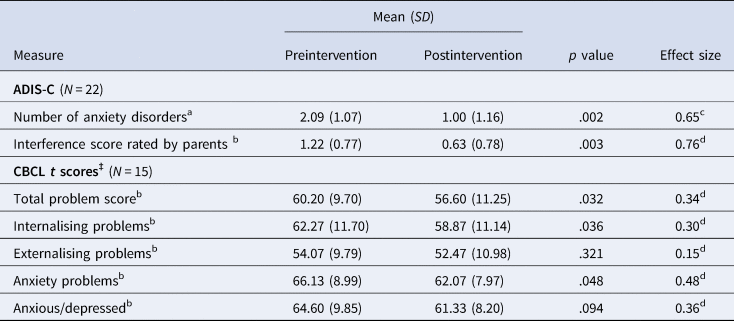
Note: ‡Mother-report. aWilcoxon signed-rank test, bPaired samples t test. cr = z / √N, dCohen's d.
ADIS-C
Parents of 22 children completed the ADIS-C at both preintervention and postintervention. Children with complete ADIS-C assessments did not differ from children with incomplete ADIS-C assessments in terms of age, sex, total IQ, and maternal education level (all p ≥ .08).
The mean number of anxiety disorders decreased significantly from preintervention (M = 2.09, SD = 1.07) to postintervention (M = 1.00, SD = 1.16), z = −3.04, p = .002. The effect size was r = .65, indicating a large to very large effect (Cohen, Reference Cohen1988). The average interference score rated by parents also significantly decreased from preintervention (M = 1.22, SD = 0.77) to postintervention (M = 0.63, SD = 0.78), t(22) = 3.34, p = .003. The effect size was d = 0.76, indicating a large effect (Cohen, Reference Cohen1988).
CBCL
Unfortunately, the number of CBCLs completed by fathers was too small to analyse. Therefore, only CBCL data reported by mothers were analysed. Mothers of 15 children completed the CBCL at both pre- and postintervention. Children with complete CBCL assessments did not differ from children with incomplete CBCL assessments in terms of age, sex, total IQ, and maternal education level (all p > .27).
A significant decrease in CBCL total problem scores was found, t(14) = 2.38, p = .032, d = 0.34, which indicates that overall, emotional and behavioural problems decreased from preintervention to postintervention. A significant decrease was also found for internalising problems, t(14) = 2.32, p = .036, d = 0.30, and anxiety problems, t(14) = 2.17, p = .048, d = 0.48. The effect sizes indicate small to medium effects (Cohen, Reference Cohen1988). The observed decrease in scores of the anxious/depressed subscale was not significant, p = .094, d = 0.36. The decrease in externalising problems was also not significant, p = .321, d = 0.15.
Predictors of Treatment Progress
Treatment progress was defined as the difference between preintervention and postintervention CBCL anxiety problems score (primary outcome). A positive score indicates treatment progress (i.e., a lower anxiety problems score at postintervention than at preintervention).
Sex and age
Treatment progress of boys (mean Δ = 3.00, SD = 7.94) and girls (mean Δ = 5.00, SD = 7.05) did not significantly differ, p = .614, d = 0.27, indicating that sex does not predict treatment progress. Moreover, children's age at start of participation in Fun FRIENDS did not significantly predict treatment progress, p = .73.
Anxiety problems at preintervention
The level of preintervention anxiety problems did significantly predict treatment progress, β = .537, F(1) = 5.27, p = .04, R 2 = .29. This indicates that a higher preintervention anxiety problems score predicts more treatment progress (demonstrated by a larger positive difference between preintervention and postintervention anxiety problems).
Discussion
The current study examined whether anxiety in young children with anxiety disorders decreases after participating in the cognitive behavioural Fun FRIENDS program. As expected, we found significant decreases in the number of anxiety disorder diagnoses and symptom interference with young children's daily lives as reported by parents. Moreover, we found significant decreases in emotional and behavioural problems, internalising problems and anxiety problems. These results suggest that the Dutch version of the Fun FRIENDS program is promising in treating anxiety disorders in young children in a clinical setting, which is in line with previous findings (Barrett et al., Reference Barrett, Fisak and Cooper2015; Carlyle, Reference Carlyle2014). Children with higher levels of preintervention anxiety problems seemed to benefit most from the Fun FRIENDS program, which is also in line with previous findings (Barrett et al., Reference Barrett, Fisak and Cooper2015). Sex and age did not predict treatment progress.
The decrease in anxious/depressed symptoms, however, was not significant. This might be surprising, as the Fun FRIENDS program specifically targets issues such as anxiety and depression (Pahl & Barrett, Reference Pahl and Barrett2007). Moreover, previous studies have shown that the FRIENDS for Life program (for children aged 8–12 years), on which the Fun FRIENDS program was based, is effective in reducing both anxiety and depressive symptoms (e.g., Ahlen, Breitholtz, Barrett, & Gallegos, Reference Ahlen, Breitholtz, Barrett and Gallegos2012; Essau, Conradt, Sasagawa, & Ollendick, Reference Essau, Conradt, Sasagawa and Ollendick2012; World Health Organization, 2004). The difference in results may be explained by the outcome measures used. To measure depressive symptoms, previous studies used the Children's Depression Inventory (CDI; Kovacs, Reference Kovacs1985) and the Revised Child Anxiety and Depression Scale (RCADS; Chorpita, Yim, Moffitt, Umemoto, & Francis, Reference Chorpita, Yim, Moffitt, Umemoto and Francis2000), whereas the current study used the CBCL. Moreover, the children who participated in the current study received treatment because they were diagnosed with anxiety disorders. Based on preintervention ADIS-C scores, no children were diagnosed with a depressive disorder at baseline. It should also be noted that, considering the trend towards significance, the decrease in anxious/depressive symptoms may have reached the level of significance if the sample size had been larger.
It is not surprising that externalising problems did not significantly decrease after participating in the Fun FRIENDS program because Fun FRIENDS mainly targets internalising problems (Pahl & Barrett, Reference Pahl and Barrett2007). In addition, overall, participants’ preintervention externalising problem scores were relatively low and not the main target of treatment. Therefore, a significant decrease in externalising problems might not have been likely.
This study has several strengths. First, it adds to the limited evidence-based knowledge body concerning treatment for young children with clinical levels of anxiety. Second, this is the first European study examining the Fun FRIENDS program. It is important to cross-validate findings across countries using the same validated assessment instruments and protocols. This enables us to draw more robust conclusions as to outcomes after participating in the Fun FRIENDS program. Third, the senior psychologist providing the Fun FRIENDS program was trained by the program developer; and, fourth, treatment was fully manualised and standardised.
However, although promising, the results of the current study should be interpreted with caution as the study did not include a control group, which limits the internal validity (Shadish, Cook, & Campbell, Reference Shadish, Cook and Campbell2002). In this study, internal validity refers to whether the decline in symptoms can be attributed to participating in the Fun FRIENDS program. Without the use of a control group, it is not possible to draw definite conclusions as to the effectiveness of an intervention, because other influences on the outcome cannot be ruled out. For example, the decline in symptoms may also be influenced by maturation (i.e., naturally occurring changes over time) or regression to the mean (i.e., the tendency to score less extremely on a posttest assessment than on a pretest assessment). In the future, randomised controlled trials with larger groups of participants should be conducted in order to draw definite conclusions as to the effectiveness of the Fun FRIENDS program. It would also be useful to include a long-term follow-up assessment to examine whether the obtained results remain over a longer period of time.
Moreover, to optimise treatment for young children with anxiety disorders, future research should focus on which elements of the Fun FRIENDS program are most useful. Future research could also consider the format in which the intervention is delivered. A randomised controlled trial has shown that the FRIENDS for Life program (Barrett & Turner, Reference Barrett and Turner2000; translated by Utens, de Nijs, & Ferdinand, Reference Utens, de Nijs and Ferdinand2001) is as equally effective in diminishing anxiety through individual treatment as through group treatment (Liber et al., Reference Liber, Van Widenfelt, Utens, Ferdinand, Van der Leeden, Van Gastel and Treffers2008). Whether this also holds true for the Fun FRIENDS program should be examined in future research. The psychologists, parents and children involved in the current study considered the group format to be beneficial. The group format seemed to enable children to learn from each other and to encourage each other in learning the cognitive behavioural techniques. However, when delivering an intervention in a group format, children may drop out due to different issues (e.g., change in parents’ working schedule), which may also have a negative influence on the rest of the group. For this reason, prior to participating in the program, we asked parents to fully commit to the treatment.
In conclusion, the Fun FRIENDS program is one of the very few cognitive behavioural treatment programs for young children with anxiety disorders. The current study shows promising results as to the outcomes after participating in the Fun FRIENDS program. To determine the effectiveness of the program in a clinical setting, randomised controlled trials with longer follow-up periods are needed.
Acknowledgments
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Declaration of interest
The authors declare that they have no conflict of interest. EU translated the FRIENDS for Life and Fun FRIENDS manuals to Dutch but does not receive remuneration for this.
Ethical standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.







