106 results
Somatic multicomorbidity and disability in patients with psychiatric disorders in comparison to the general population: a quasi-epidemiological investigation in 54,826 subjects from 40 countries (COMET-G study)
-
- Journal:
- CNS Spectrums / Volume 29 / Issue 2 / April 2024
- Published online by Cambridge University Press:
- 25 January 2024, pp. 126-149
-
- Article
-
- You have access
- HTML
- Export citation
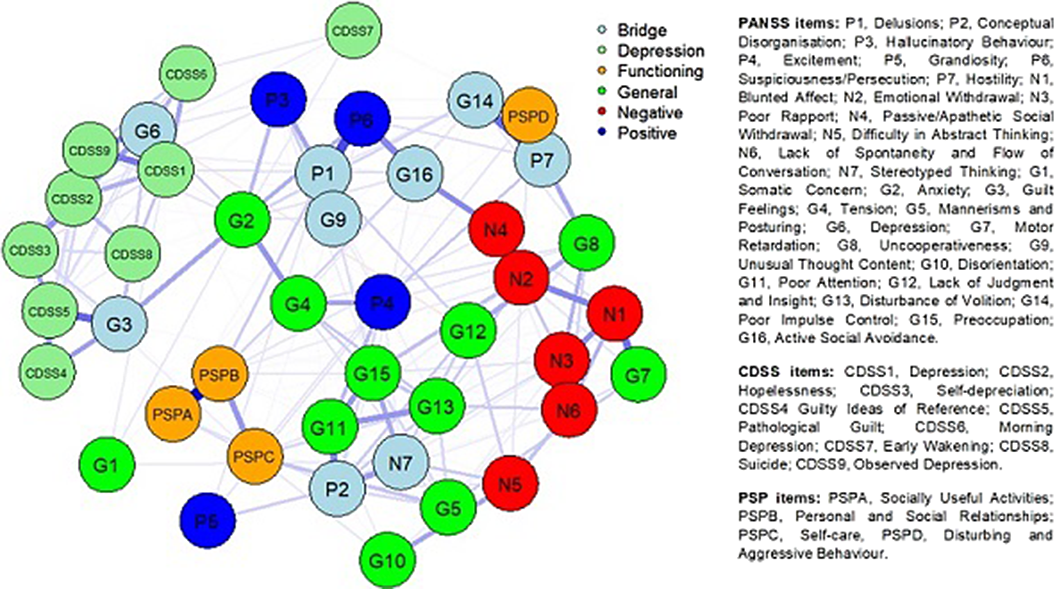
Searching for bridges between psychopathology and real-world functioning in first-episode psychosis: a network analysis from the OPTiMiSE trial
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S262-S263
-
- Article
-
- You have access
- Open access
- Export citation
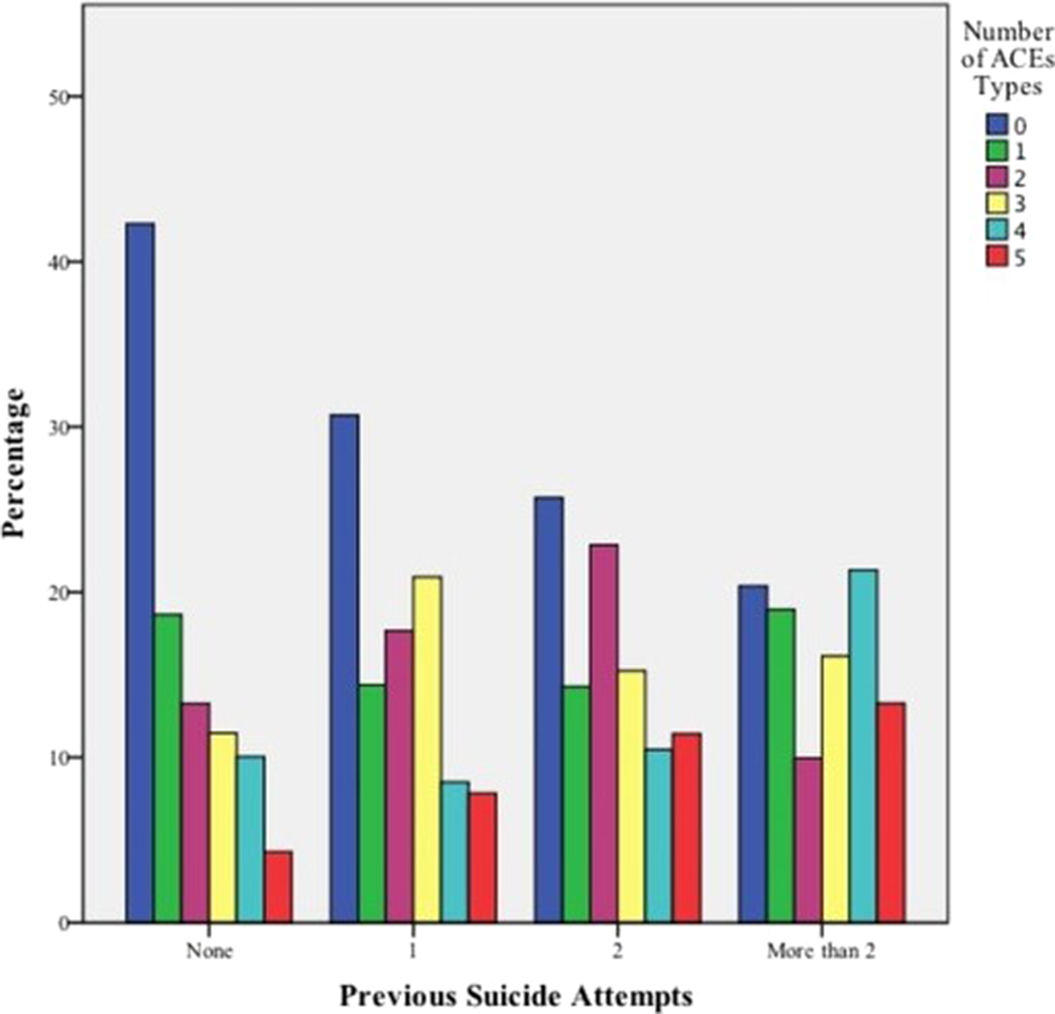
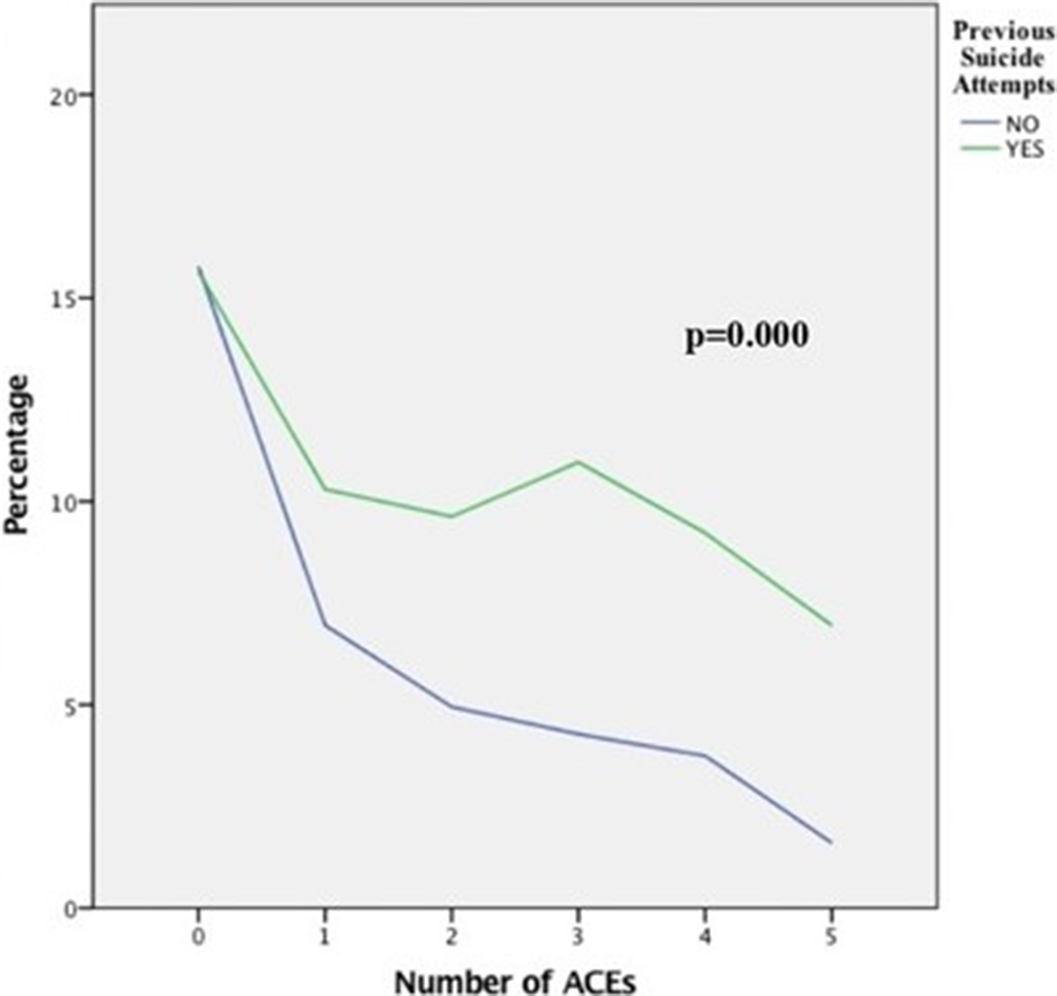
Association between adverse childhood experiences and the number of suicide attempts in lifetime
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S561-S562
-
- Article
-
- You have access
- Open access
- Export citation
Gut permeability and low-grade inflammation in bipolar disorder
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S385-S386
-
- Article
-
- You have access
- Open access
- Export citation
The response to unfolded proteins in schizophrenia and bipolar disorder
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S636-S637
-
- Article
-
- You have access
- Open access
- Export citation
SOD and CAT as potential preliminary biomarkers for the differential diagnosis of schizophrenia and bipolar disorder in the first episode of psychosis
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S449-S450
-
- Article
-
- You have access
- Open access
- Export citation
An empirical staging model for schizophrenia using machine learning
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S626-S627
-
- Article
-
- You have access
- Open access
- Export citation
Tobacco use in first-episode psychosis, a multinational EU-GEI study
-
- Journal:
- Psychological Medicine / Volume 53 / Issue 15 / November 2023
- Published online by Cambridge University Press:
- 26 April 2023, pp. 7265-7276
-
- Article
- Export citation
Neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, and monocyte-to-lymphocyte ratio in depressed patients with suicidal behavior: A systematic review
-
- Journal:
- European Psychiatry / Accepted manuscript
- Published online by Cambridge University Press:
- 16 April 2023, pp. 1-25
-
- Article
-
- You have access
- Open access
- Export citation
Trends in the incidence of hospital-treated suicide attempts during the COVID-19 pandemic in Oviedo, Spain
-
- Journal:
- European Psychiatry / Volume 66 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 03 February 2023, e23
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Genetic and psychosocial stressors have independent effects on the level of subclinical psychosis: findings from the multinational EU-GEI study
-
- Journal:
- Epidemiology and Psychiatric Sciences / Volume 31 / 2022
- Published online by Cambridge University Press:
- 27 September 2022, e68
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Endocrine biomarkers related to sleep-wake cycle and sleep disturbances in patients with bipolar disorder: A systematic review
-
- Journal:
- European Psychiatry / Volume 65 / Issue S1 / June 2022
- Published online by Cambridge University Press:
- 01 September 2022, p. S360
-
- Article
-
- You have access
- Open access
- Export citation
Orexins and bipolar disorder: A review
-
- Journal:
- European Psychiatry / Volume 65 / Issue S1 / June 2022
- Published online by Cambridge University Press:
- 01 September 2022, p. S400
-
- Article
-
- You have access
- Open access
- Export citation
National Center for In-situ Tomographic Ultramicroscopy and the Waffle Method: An Approach for Cryo-FIB/SEM Thin Lamellae Preparation
-
- Journal:
- Microscopy and Microanalysis / Volume 28 / Issue S1 / August 2022
- Published online by Cambridge University Press:
- 22 July 2022, p. 1248
- Print publication:
- August 2022
-
- Article
-
- You have access
- Export citation
23 - Paleontology and Geology of the Mursi Formation
- from Part III - Eastern and Central Africa
-
-
- Book:
- African Paleoecology and Human Evolution
- Published online:
- 19 May 2022
- Print publication:
- 09 June 2022, pp 278-288
-
- Chapter
- Export citation
Factors associated with real-world functioning in first stages of schizophrenia disorder
-
- Journal:
- European Psychiatry / Volume 64 / Issue S1 / April 2021
- Published online by Cambridge University Press:
- 13 August 2021, p. S805
-
- Article
-
- You have access
- Open access
- Export citation
Insomnia associated with neutrophil/lymphocyte ratio in female patients with schizophrenia
-
- Journal:
- European Psychiatry / Volume 64 / Issue S1 / April 2021
- Published online by Cambridge University Press:
- 13 August 2021, p. S806
-
- Article
-
- You have access
- Open access
- Export citation
The mediating role of impulsivity in the relationship between suicidal behaviour and early traumatic experiences in depressed subjects
-
- Journal:
- European Psychiatry / Volume 64 / Issue S1 / April 2021
- Published online by Cambridge University Press:
- 13 August 2021, p. S583
-
- Article
-
- You have access
- Open access
- Export citation
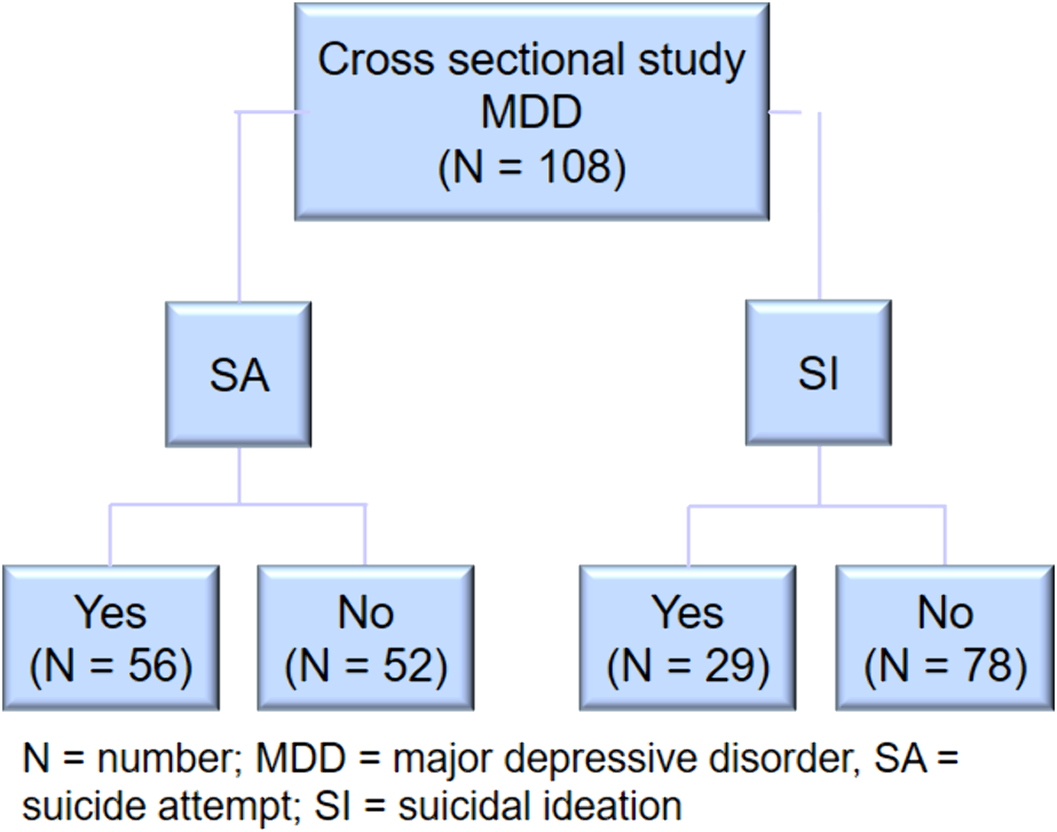
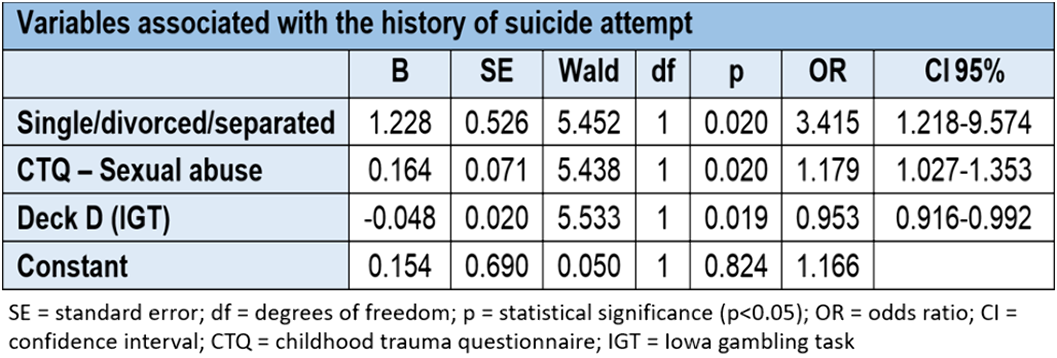
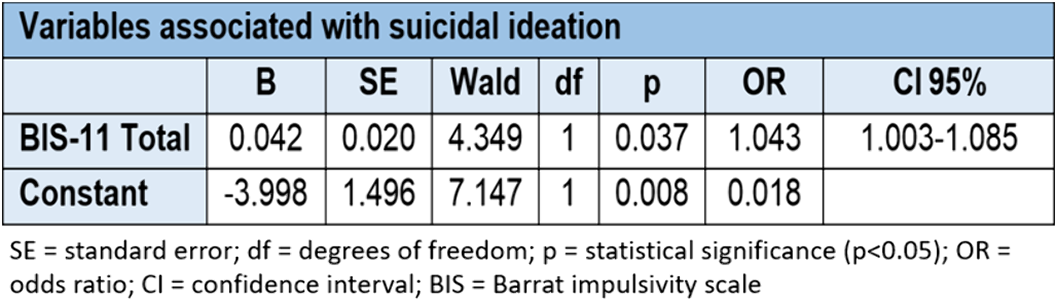
Association of traumatic events in childhood, impulsivity and decision-making with previous suicide attempt and/or current suicidal ideation in adult patients with major depressive disorder
-
- Journal:
- European Psychiatry / Volume 64 / Issue S1 / April 2021
- Published online by Cambridge University Press:
- 13 August 2021, pp. S177-S178
-
- Article
-
- You have access
- Open access
- Export citation
COVID-19 psychological impact in patients with depressive disorder: Differences based on their age
-
- Journal:
- European Psychiatry / Volume 64 / Issue S1 / April 2021
- Published online by Cambridge University Press:
- 13 August 2021, pp. S674-S675
-
- Article
-
- You have access
- Open access
- Export citation