25 results
Association between job role and coronavirus disease 2019 (COVID-19) among healthcare personnel, Iowa, 2021
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 01 December 2022, e188
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
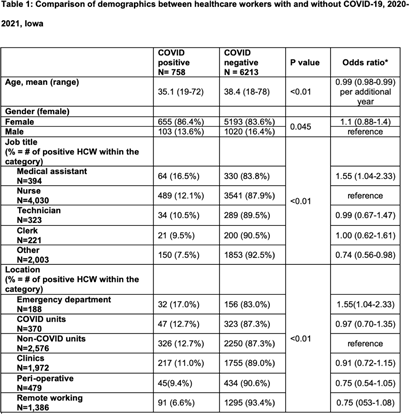
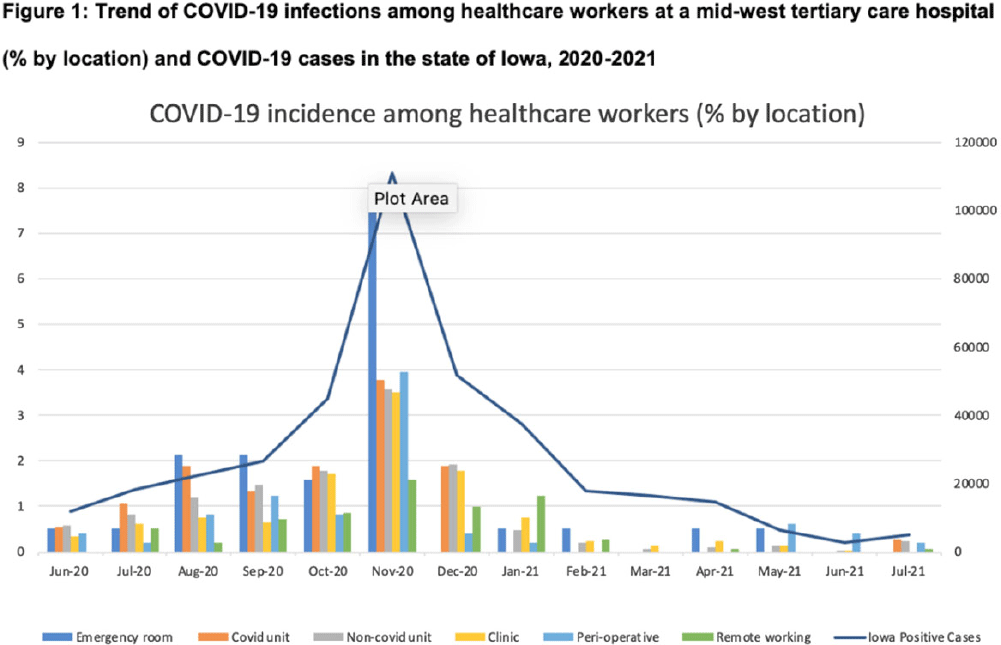
Coronavirus disease 2019 (COVID-19) among nonphysician healthcare personnel by work location at a tertiary-care center, Iowa, 2020–2021
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 8 / August 2023
- Published online by Cambridge University Press:
- 02 June 2022, pp. 1351-1354
- Print publication:
- August 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
COVID-19 incidence among nonphysician healthcare workers at a tertiary care center–Iowa, 2020–2021
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s6-s7
-
- Article
-
- You have access
- Open access
- Export citation
Blood-culture ordering practices in patients with a central line at an academic medical center–Iowa, 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s30
-
- Article
-
- You have access
- Open access
- Export citation
Blood culture practices in patients with a central line at an academic medical center—Iowa, 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 13 April 2022, e64
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
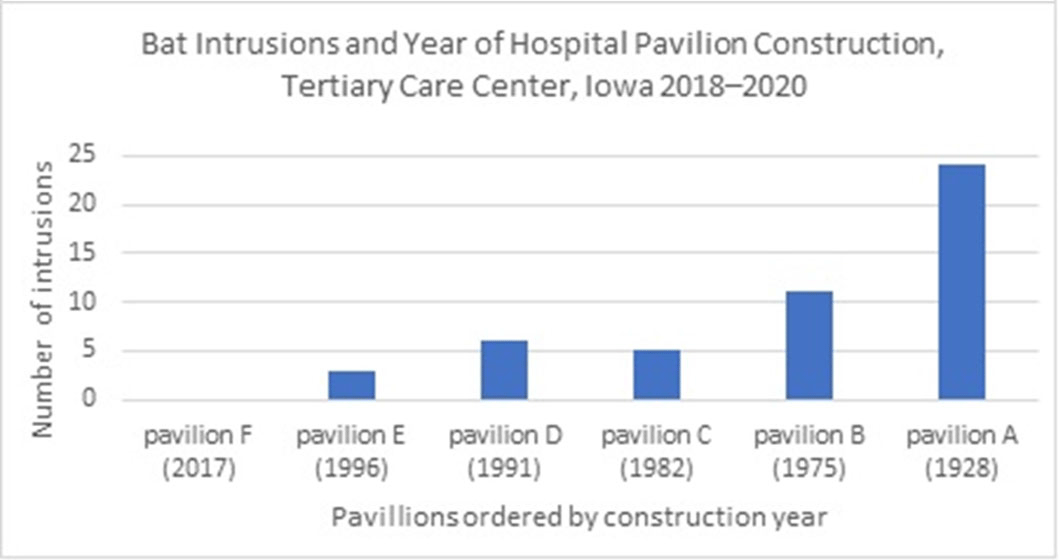
Bat intrusions at a tertiary care center in Iowa, 2018–2020
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 12 / December 2022
- Published online by Cambridge University Press:
- 16 August 2021, pp. 1948-1950
- Print publication:
- December 2022
-
- Article
- Export citation
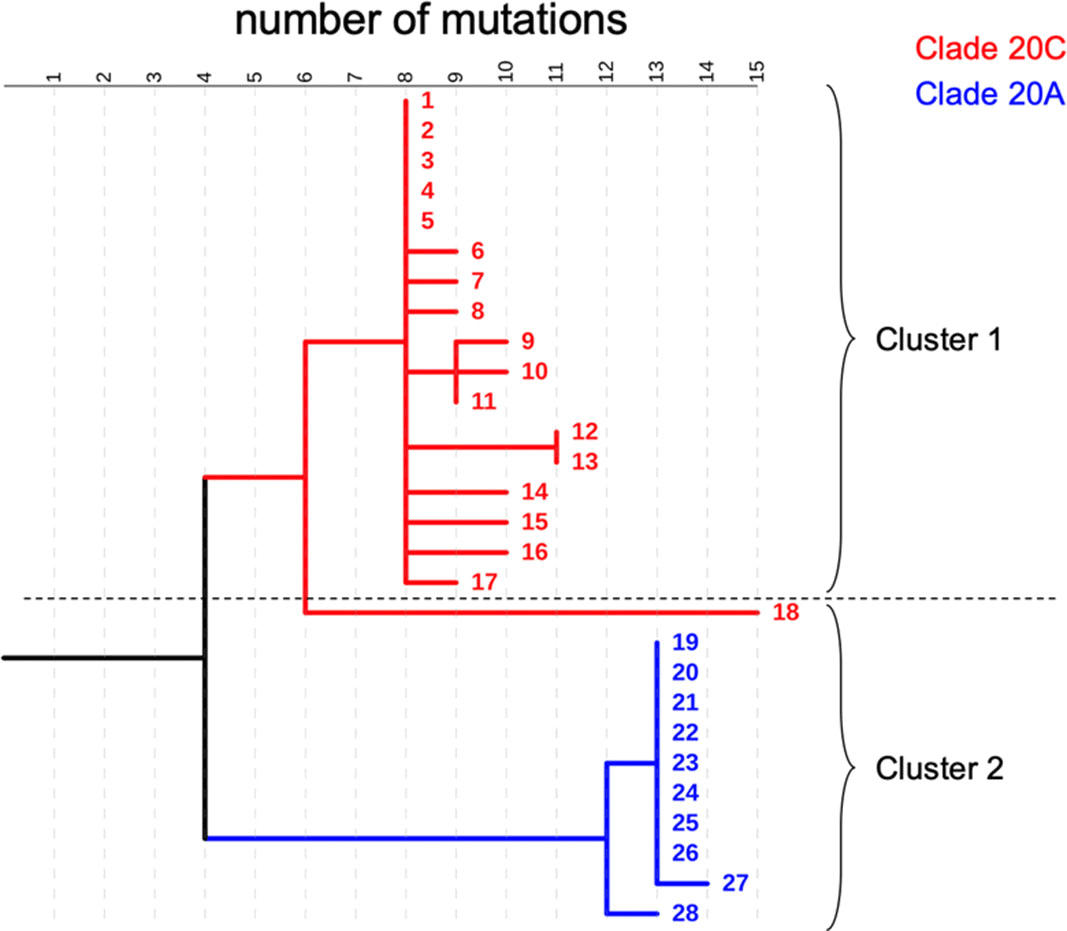
Molecular Epidemiology of Large COVID-19 Clusters at an Academic Medical Center, March–October 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s10-s11
-
- Article
-
- You have access
- Open access
- Export citation
Bat Intrusions at a Tertiary Care Center, Iowa 2018–2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s16
-
- Article
-
- You have access
- Open access
- Export citation
COVID-19 Conversion after Exposure in a Semiprivate Room at a Tertiary Care Center in Iowa, July–December 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s20-s21
-
- Article
-
- You have access
- Open access
- Export citation
Coronavirus Disease 2019 (COVID-19) Admission Screening at a Tertiary-Care Center, Iowa 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s1
-
- Article
-
- You have access
- Open access
- Export citation
Suspected COVID-19 Reinfections at a Tertiary Care Center, Iowa 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s19
-
- Article
-
- You have access
- Open access
- Export citation
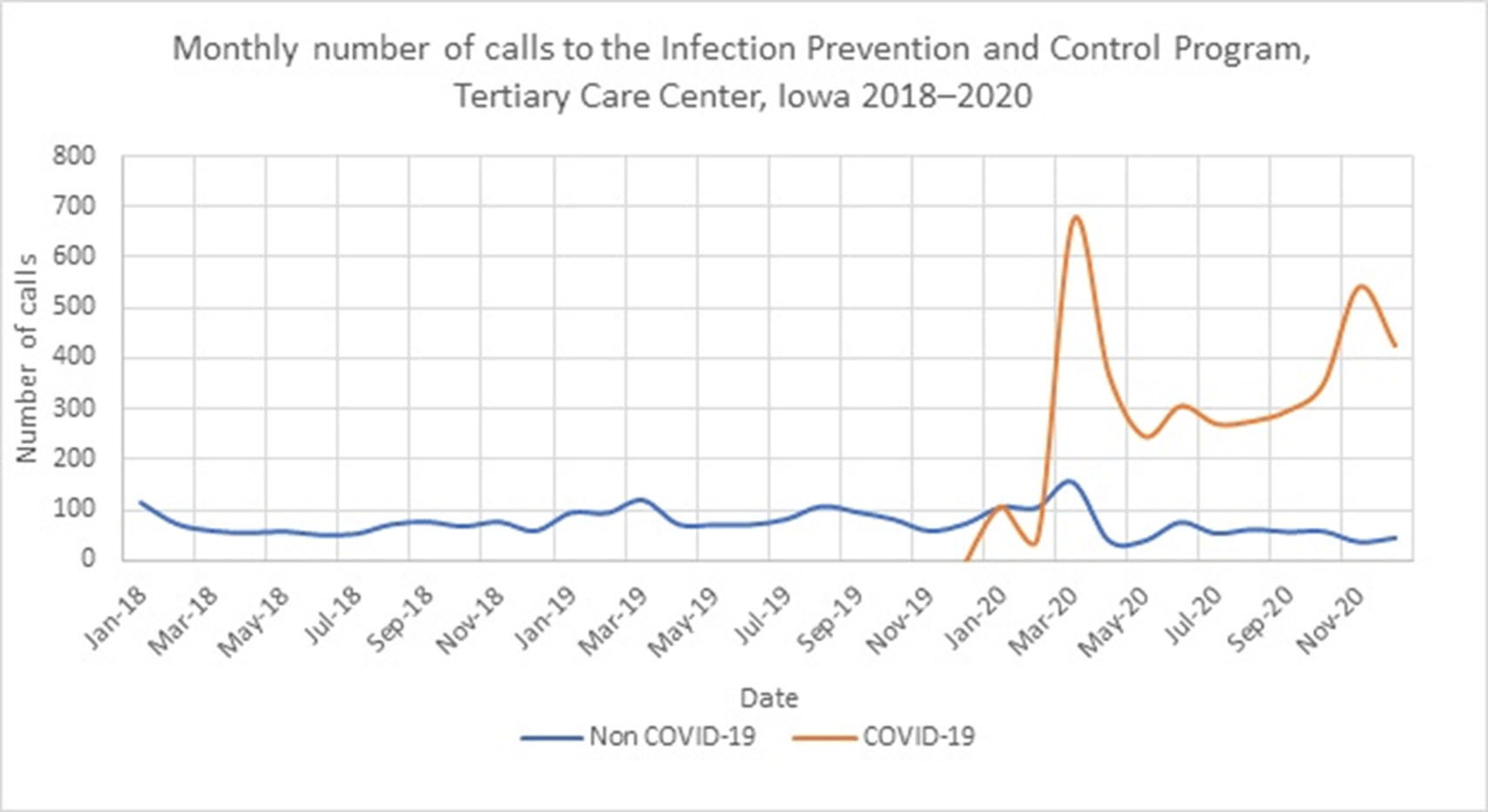
Impact of COVID-19 on Volume of Infection Prevention and Control Calls at a Tertiary-Care Center in Iowa, 2018–2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s53-s54
-
- Article
-
- You have access
- Open access
- Export citation
Coronavirus disease 2019 (COVID-19) incidence after exposures in shared patient rooms in a tertiary-care center in Iowa, July 2020–May 2021
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 12 / December 2022
- Published online by Cambridge University Press:
- 12 July 2021, pp. 1910-1913
- Print publication:
- December 2022
-
- Article
- Export citation
Coronavirus disease 2019 (COVID-19) admission screening and assessment of infectiousness at an academic medical center in Iowa, 2020
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 8 / August 2022
- Published online by Cambridge University Press:
- 24 June 2021, pp. 974-978
- Print publication:
- August 2022
-
- Article
- Export citation
Molecular epidemiology of large coronavirus disease 2019 (COVID-19) clusters before and after the implementation of routine serial testing at an academic medical center in Iowa, 2020
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 12 / December 2021
- Published online by Cambridge University Press:
- 24 June 2021, pp. 1514-1516
- Print publication:
- December 2021
-
- Article
- Export citation
Assessment of room quality of manual cleaning and turnaround times with and without ultraviolet light at an academic medical center
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 1 / January 2021
- Published online by Cambridge University Press:
- 29 October 2020, pp. 107-108
- Print publication:
- January 2021
-
- Article
- Export citation
Reduction in Abdominal Hysterectomy Surgical Site Infection Rates After the Addition of Anaerobic Antimicrobial Prophylaxis
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s47
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Communications and Screening for 2019 Novel Coronavirus at a Tertiary-Care Medical Center
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s84
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Impact of UV-Light Use on the Quality of Manual Cleaning and Room Turnover Times at a Large Tertiary-Care Hospital, 2019
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s266-s267
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Impact of an Enhanced Prevention Bundle on Central-Line–Associated Bloodstream Infection Incidence in Adult Oncology Units
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s256-s258
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation