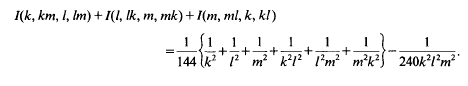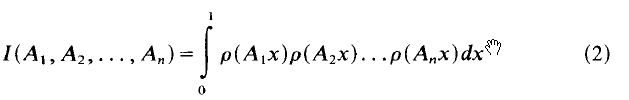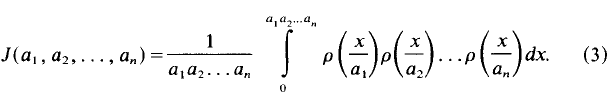16 results
Structure and ionic transport in disordered silicates
-
- Journal:
- Mineralogical Magazine / Volume 64 / Issue 3 / June 2000
- Published online by Cambridge University Press:
- 05 July 2018, pp. 441-446
-
- Article
- Export citation
TW Hydrae: multi-wavelength interferometry of a transition disk
-
- Journal:
- Proceedings of the International Astronomical Union / Volume 8 / Issue S299 / June 2013
- Published online by Cambridge University Press:
- 06 January 2014, pp. 104-108
- Print publication:
- June 2013
-
- Article
-
- You have access
- Export citation
The osmotic pressure, after re-solution, of serum dried from the frozen state (F.D. serum)
-
- Journal:
- Journal of Hygiene / Volume 40 / Issue 5 / September 1940
- Published online by Cambridge University Press:
- 15 May 2009, pp. 548-554
-
- Article
-
- You have access
- Export citation
Foreword
-
-
- Book:
- Sieve Methods, Exponential Sums, and their Applications in Number Theory
- Published online:
- 03 February 2010
- Print publication:
- 30 January 1997, pp vii-vii
-
- Chapter
- Export citation

Sieve Methods, Exponential Sums, and their Applications in Number Theory
-
- Published online:
- 03 February 2010
- Print publication:
- 30 January 1997
Participants in the Symposium
-
- Book:
- Sieve Methods, Exponential Sums, and their Applications in Number Theory
- Published online:
- 03 February 2010
- Print publication:
- 30 January 1997, pp xi-xii
-
- Chapter
- Export citation
Contents
-
- Book:
- Sieve Methods, Exponential Sums, and their Applications in Number Theory
- Published online:
- 03 February 2010
- Print publication:
- 30 January 1997, pp v-vi
-
- Chapter
- Export citation
Frontmatter
-
- Book:
- Sieve Methods, Exponential Sums, and their Applications in Number Theory
- Published online:
- 03 February 2010
- Print publication:
- 30 January 1997, pp i-iv
-
- Chapter
- Export citation
Index of Authors
-
- Book:
- Sieve Methods, Exponential Sums, and their Applications in Number Theory
- Published online:
- 03 February 2010
- Print publication:
- 30 January 1997, pp viii-x
-
- Chapter
- Export citation
Multiple Franel integrals: Corrigendum
-
- Journal:
- Mathematika / Volume 41 / Issue 2 / December 1994
- Published online by Cambridge University Press:
- 26 February 2010, p. 401
- Print publication:
- December 1994
-
- Article
- Export citation
Multiple Franel integrals
- Part of
-
- Journal:
- Mathematika / Volume 40 / Issue 1 / June 1993
- Published online by Cambridge University Press:
- 26 February 2010, pp. 51-70
- Print publication:
- June 1993
-
- Article
- Export citation
Elucidating Structural Mechanisms for Cordierite Ceramic Formation using Synchrotron Radiation
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 307 / 1993
- Published online by Cambridge University Press:
- 15 February 2011, 39
- Print publication:
- 1993
-
- Article
- Export citation
The Changing Environments of Dopants in Amorphous Silicon at Various Stages of Annealing
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 307 / 1993
- Published online by Cambridge University Press:
- 15 February 2011, 21
- Print publication:
- 1993
-
- Article
- Export citation
Glancing Angle X-ray Absorption Spectroscopy
-
- Journal:
- Advances in X-ray Analysis / Volume 34 / 1990
- Published online by Cambridge University Press:
- 06 March 2019, pp. 13-22
- Print publication:
- 1990
-
- Article
- Export citation
High Throughput Rate Solid State Detector Systems for Fluorescence EXAFS
-
- Journal:
- Advances in X-ray Analysis / Volume 34 / 1990
- Published online by Cambridge University Press:
- 06 March 2019, pp. 187-191
- Print publication:
- 1990
-
- Article
- Export citation
Current and Future Energy Dispersive EXAFS Detector Systems
-
- Journal:
- Advances in X-ray Analysis / Volume 34 / 1990
- Published online by Cambridge University Press:
- 06 March 2019, pp. 177-185
- Print publication:
- 1990
-
- Article
- Export citation