17 results
The Mechanics, Art, and Value of Central Line Stewardship
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s71-s72
-
- Article
-
- You have access
- Open access
- Export citation
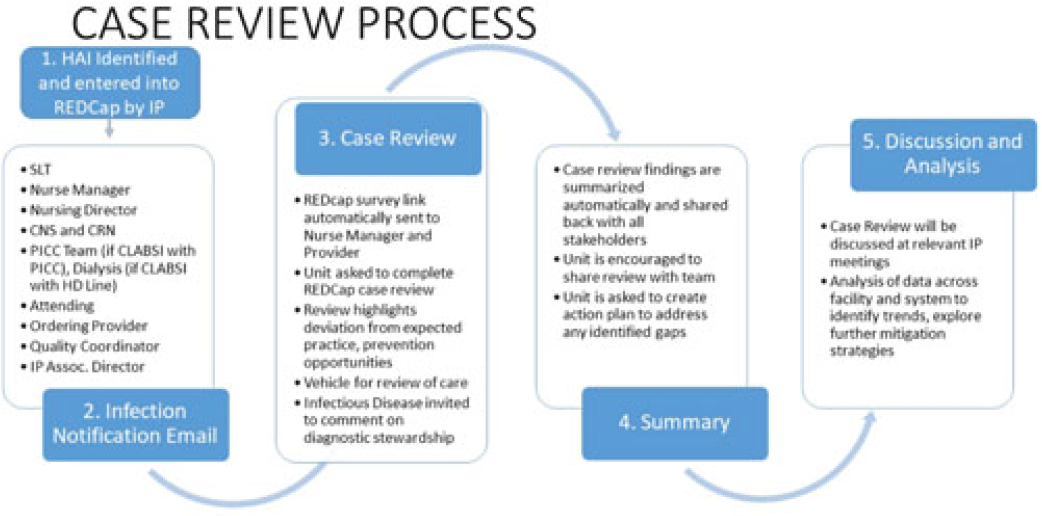
Electrifying the case review process for better speed, reach, and impact
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s95
-
- Article
-
- You have access
- Open access
- Export citation
Benefit and cost of repeating a severe acute respiratory coronavirus virus 2 (SARS-CoV-2) polymerase chain reaction (PCR) test after the second day of hospitalization in five hospitals during various community prevalences and vaccination rates
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 1 / January 2023
- Published online by Cambridge University Press:
- 16 June 2022, pp. 106-109
- Print publication:
- January 2023
-
- Article
- Export citation
Infection preventionist staffing levels and rates of 10 types of healthcare-associated infections: A 9-year ambidirectional observation
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 11 / November 2022
- Published online by Cambridge University Press:
- 17 January 2022, pp. 1641-1646
- Print publication:
- November 2022
-
- Article
- Export citation
To err is human, to forget is device-related: A cautionary note for endoscopists
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 6 / June 2022
- Published online by Cambridge University Press:
- 16 April 2021, pp. 807-808
- Print publication:
- June 2022
-
- Article
-
- You have access
- HTML
- Export citation
A cluster-control approach to a coronavirus disease 2019 (COVID-19) outbreak on a stroke ward with infection control considerations for dementia and vascular units
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 11 / November 2021
- Published online by Cambridge University Press:
- 11 January 2021, pp. 1333-1339
- Print publication:
- November 2021
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Amidst the CAUTI Metrics Hurley Burly, a Sustained SURly Success Adaptable for Reducing Other Nosocomial Infections
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s115
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Outcomes of Extended-Spectrum Beta-Lactamase Gram-Negative Bacteremia Cases Treated With Carbapenem Versus Noncarbapenem Antibiotics
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s339
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
To Err is Human, To Forget is Device-related: A Cautionary Note for Endoscopists
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s412-s413
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Sustainably reducing device utilization and device-related infections with DeCATHlons, device alternatives, and decision support
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue 11 / November 2020
- Published online by Cambridge University Press:
- 30 June 2020, pp. 1344-1347
- Print publication:
- November 2020
-
- Article
- Export citation
Mitigating and preventing hepatitis B virus exposures during hemodialysis across a large regional health system
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 40 / Issue 9 / September 2019
- Published online by Cambridge University Press:
- 20 June 2019, pp. 1066-1069
- Print publication:
- September 2019
-
- Article
- Export citation
Got GAS? Ease the Bloat with Real-Time Whole-Genome Sequencing
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 39 / Issue 6 / June 2018
- Published online by Cambridge University Press:
- 02 April 2018, pp. 760-762
- Print publication:
- June 2018
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Importation, Mitigation, and Genomic Epidemiology of Candida auris at a Large Teaching Hospital
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 39 / Issue 1 / January 2018
- Published online by Cambridge University Press:
- 06 December 2017, pp. 53-57
- Print publication:
- January 2018
-
- Article
- Export citation
Relationships Among Cleaning, Environmental DNA, and Healthcare-Associated Infections in a New Evidence-Based Design Hospital
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 36 / Issue 10 / October 2015
- Published online by Cambridge University Press:
- 08 July 2015, pp. 1130-1138
- Print publication:
- October 2015
-
- Article
- Export citation
Amount of Usage and Involvement in Explosions Not Associated with Increased Contamination of Prehospital Vehicles with Multi-drug-resistant Organisms
-
- Journal:
- Prehospital and Disaster Medicine / Volume 28 / Issue 2 / April 2013
- Published online by Cambridge University Press:
- 07 January 2013, pp. 107-109
- Print publication:
- April 2013
-
- Article
- Export citation
Detection of qacA/B in Clinical Isolates of Methicillin-Resistant Staphylococcus aureus from a Regional Healthcare Network in the Eastern United States
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 32 / Issue 11 / November 2011
- Published online by Cambridge University Press:
- 02 January 2015, pp. 1116-1119
- Print publication:
- November 2011
-
- Article
- Export citation
Gram-Negative Multidrug-Resistant Organism Colonization in a US Military Healthcare Facility in Iraq
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 32 / Issue 6 / June 2011
- Published online by Cambridge University Press:
- 02 January 2015, pp. 545-552
- Print publication:
- June 2011
-
- Article
- Export citation