41 results
Research agenda for antibiotic stewardship within the Veterans’ Health Administration, 2024–2028
-
- Journal:
- Infection Control & Hospital Epidemiology , First View
- Published online by Cambridge University Press:
- 02 February 2024, pp. 1-7
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Variations in implementation of antimicrobial stewardship via telehealth at select Veterans Affairs medical centers
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s38
-
- Article
-
- You have access
- Open access
- Export citation
Using telehealth to support antimicrobial stewardship at four rural VA medical centers: Interim analysis
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s110
-
- Article
-
- You have access
- Open access
- Export citation
Perceptions of an automated benchmarking dashboard for antimicrobial stewardship programs among antimicrobial stewards within the veterans’ health administration: a multicenter qualitative study
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 10 July 2023, e118
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
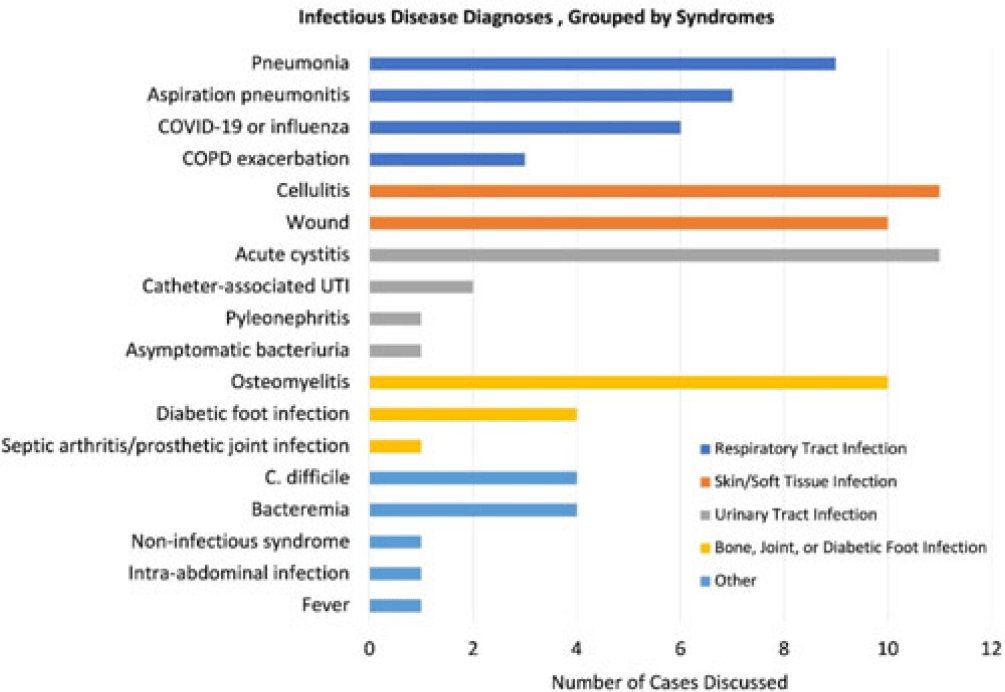
The use of telehealth-supported stewardship activities in acute-care and long-term care settings: An implementation effectiveness trial
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 12 / December 2023
- Published online by Cambridge University Press:
- 14 June 2023, pp. 2028-2035
- Print publication:
- December 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Tracking antimicrobial stewardship activities beyond days of therapy (DOT): Comparison of days of antibiotic spectrum coverage (DASC) and DOT at a single center
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 6 / June 2023
- Published online by Cambridge University Press:
- 10 January 2023, pp. 934-937
- Print publication:
- June 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Is the number of prescriptions an appropriate metric for outpatient antimicrobial consumption? A comparison between the prescription counts and days supplied
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 6 / June 2023
- Published online by Cambridge University Press:
- 23 August 2022, pp. 941-944
- Print publication:
- June 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
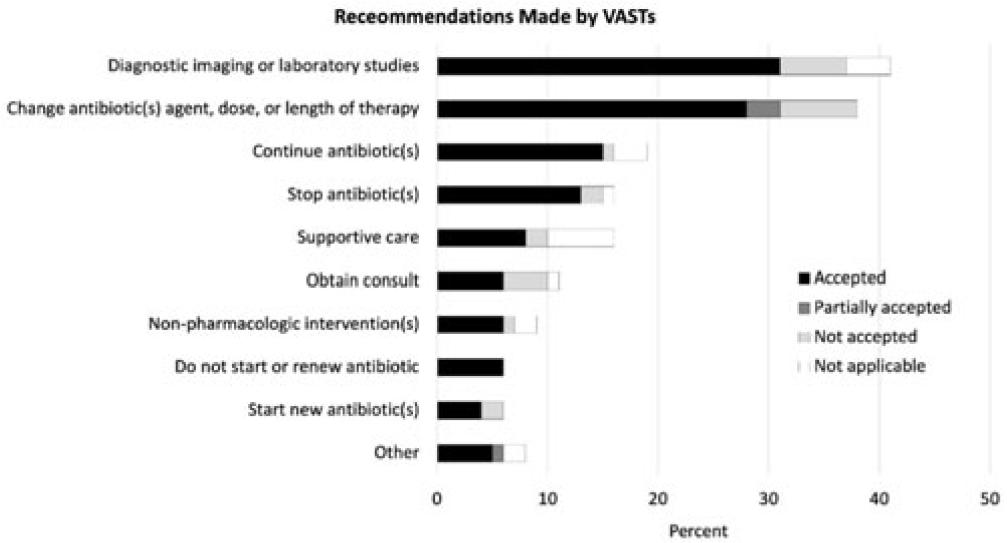
Investigation of factors influencing inpatient antibiotic prescribing decisions in the Veterans’ Health Administration
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 23 June 2022, e99
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Antibiotic-prescribing practices and associated outcomes after common urologic procedures in an integrated healthcare system
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s22
-
- Article
-
- You have access
- Open access
- Export citation
Recommendations for antimicrobial stewardship during end-of-life patient care
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s24
-
- Article
-
- You have access
- Open access
- Export citation
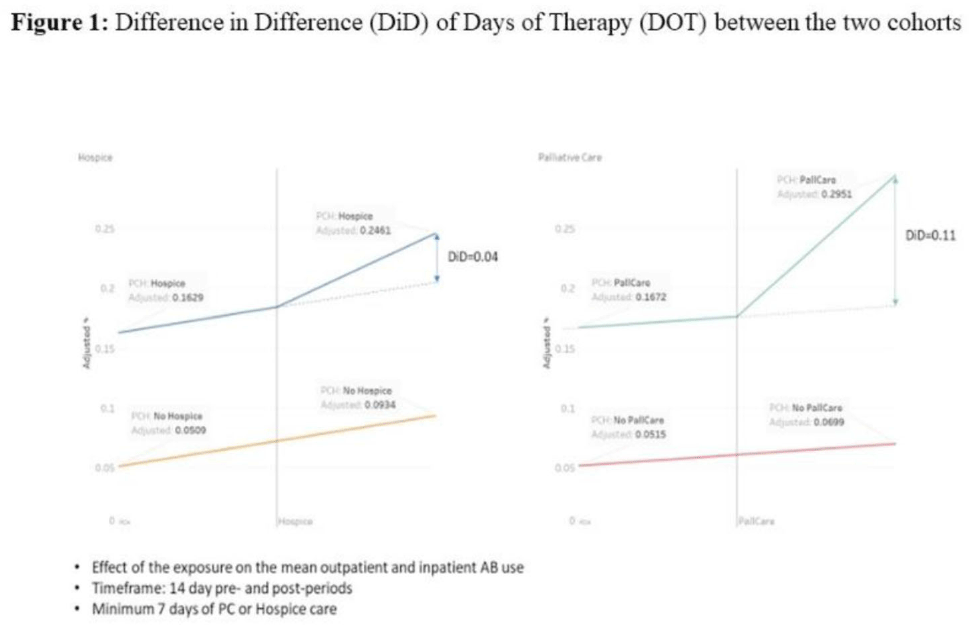
Antibiotic use in end-of-life care patients: A nationwide Veterans’ Health Administration cohort study
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s19-s20
-
- Article
-
- You have access
- Open access
- Export citation
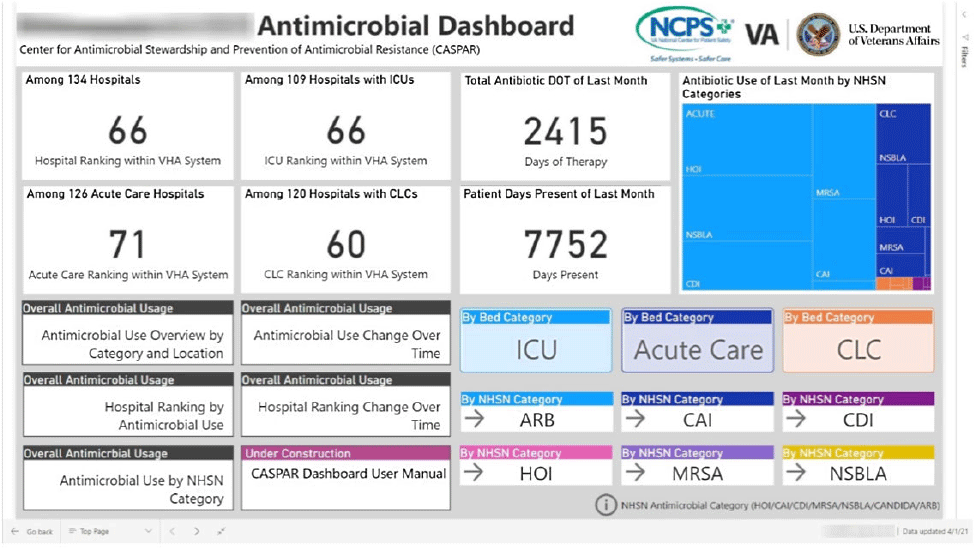
Qualitative Evaluation of an automated nationwide benchmarking antimicrobial utilization dashboard for the VHA
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s4
-
- Article
-
- You have access
- Open access
- Export citation
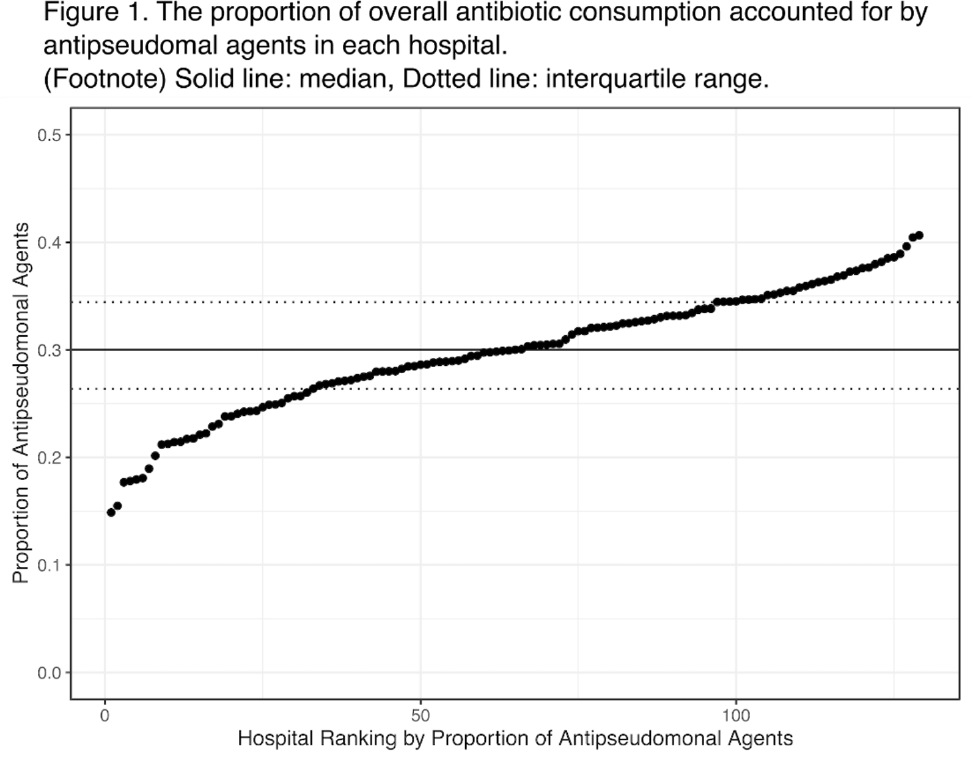
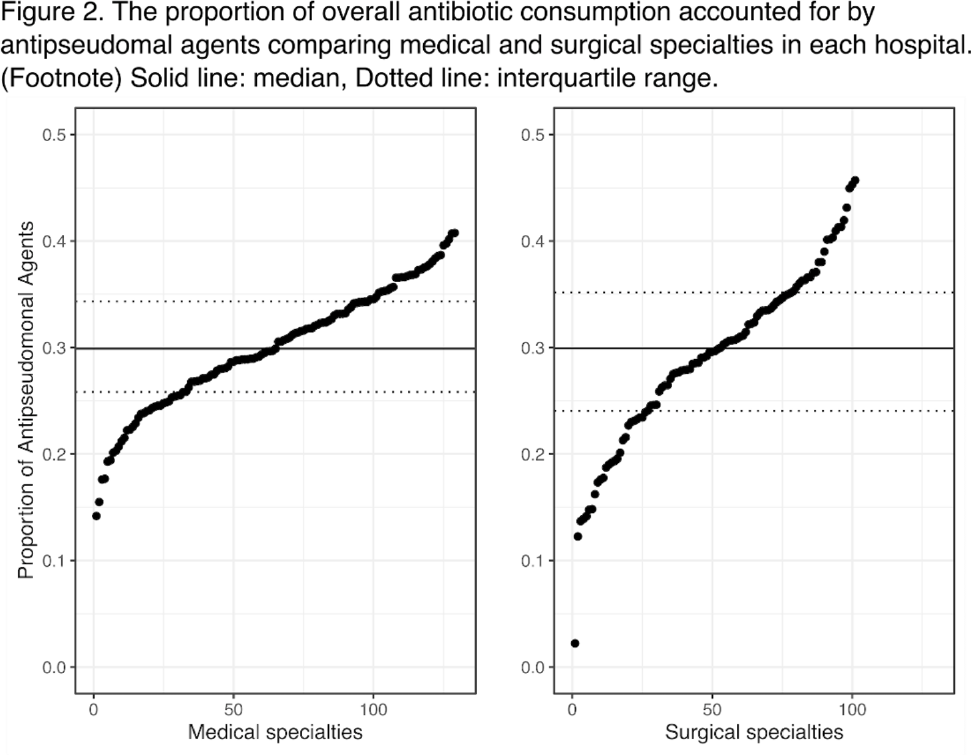
Hospital-level variation in the utilization of antipseudomonal antibiotics: A nationwide cross-sectional study at the VHA
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s21
-
- Article
-
- You have access
- Open access
- Export citation
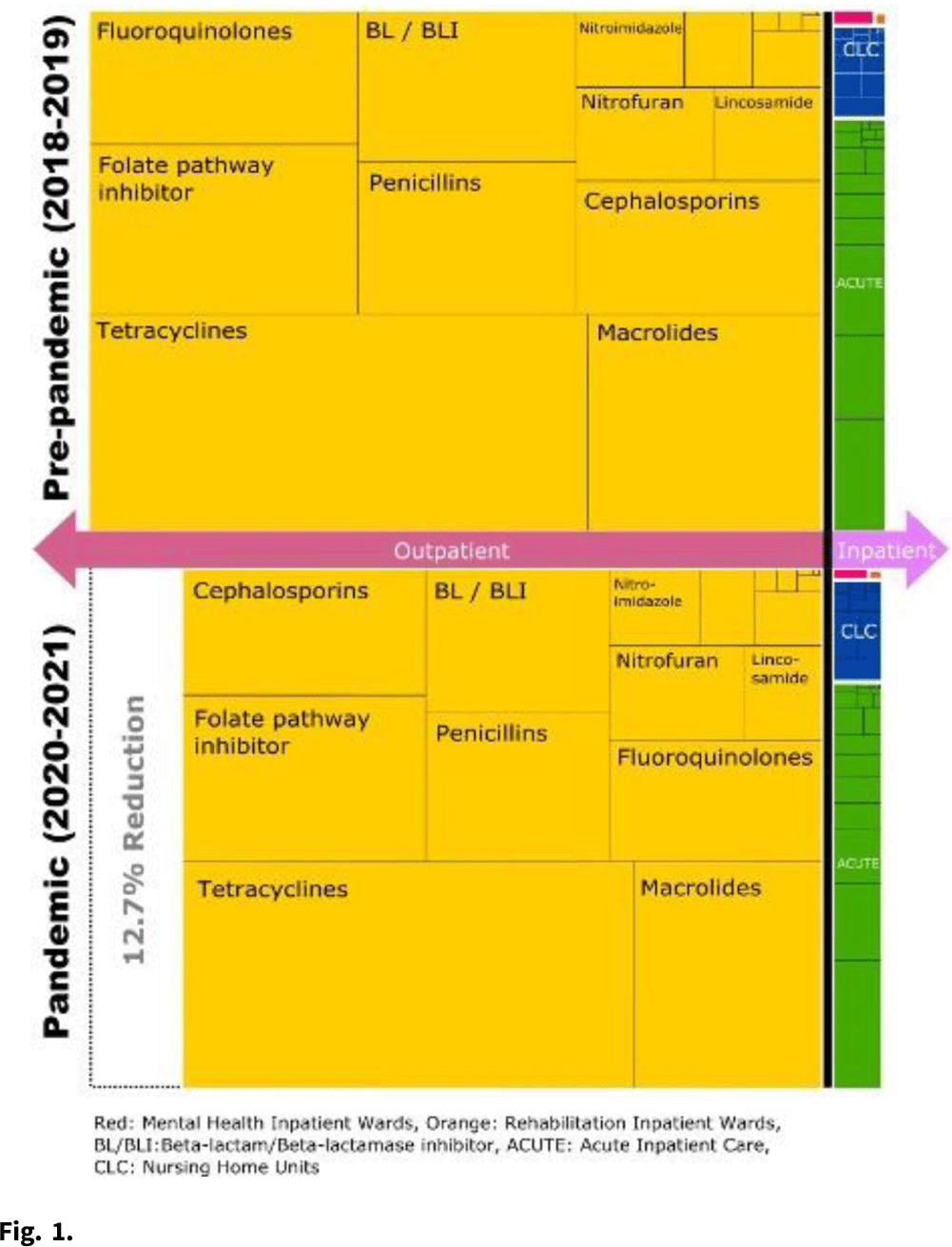
Reduction in outpatient antibiotic utilization: An unintended benefit of the COVID-19 pandemic?
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s64
-
- Article
-
- You have access
- Open access
- Export citation
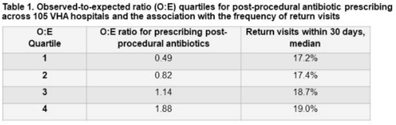
Implementation of a consensus protocol for antibiotic use for bone and joint infection to reduce unnecessary outpatient parenteral antimicrobial therapy: A quality improvement initiative
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 12 January 2022, e6
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Leveraging implementation science to advance antibiotic stewardship practice and research
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 2 / February 2022
- Published online by Cambridge University Press:
- 02 December 2021, pp. 139-146
- Print publication:
- February 2022
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Antibiotic prescribing in mental health units across the Veterans’ Health Administration: How much and how appropriate?
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 2 / February 2023
- Published online by Cambridge University Press:
- 21 October 2021, pp. 308-311
- Print publication:
- February 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Opportunities and challenges in improving antimicrobial use during the era of telehealth expansion: A narrative review
- Part of
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue 1 / 2021
- Published online by Cambridge University Press:
- 06 October 2021, e26
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Outpatient antibiotic prescribing for common infections via telemedicine versus face-to-face visits: Systematic literature review and meta-analysis
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue 1 / 2021
- Published online by Cambridge University Press:
- 31 August 2021, e24
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
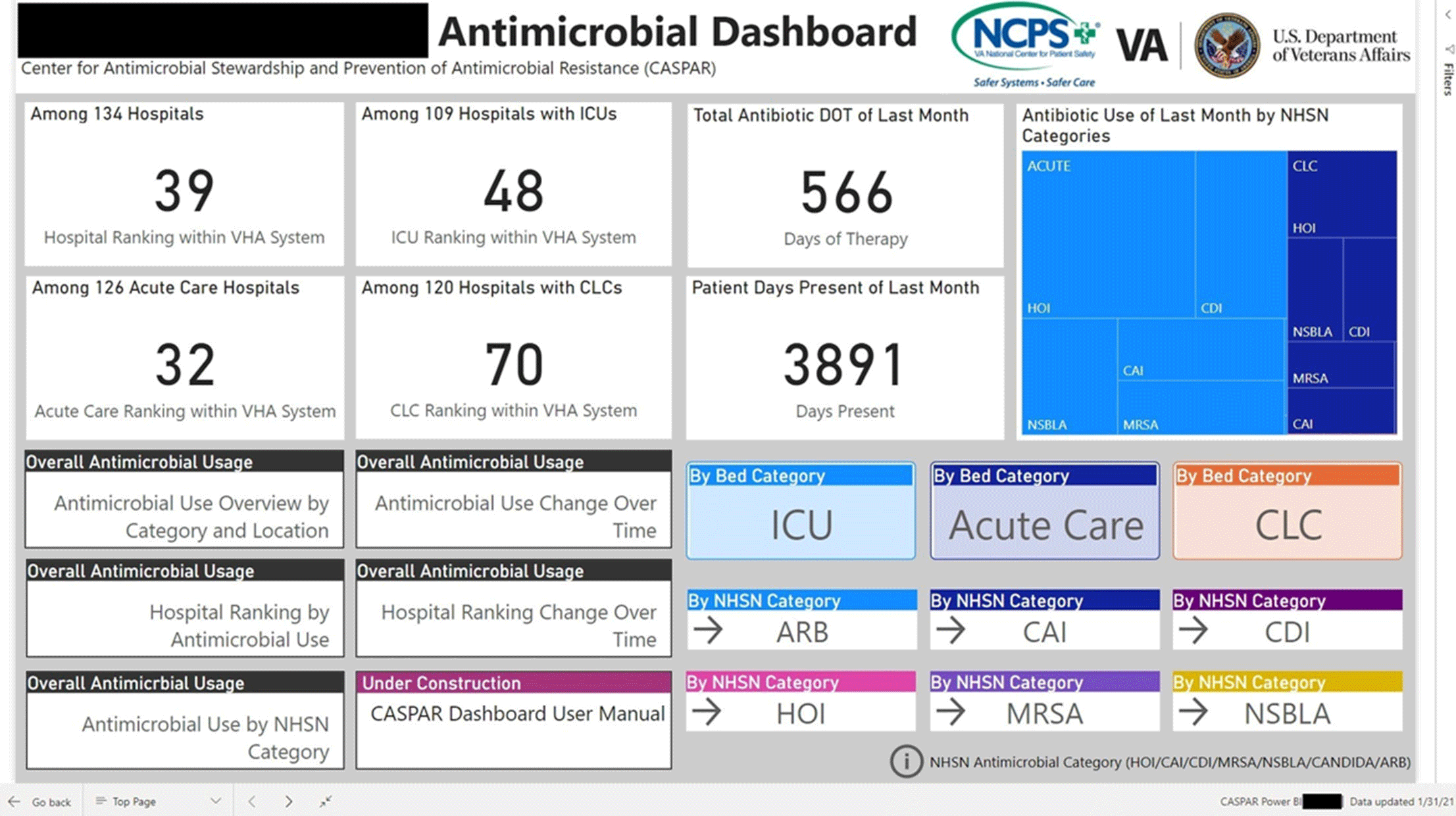
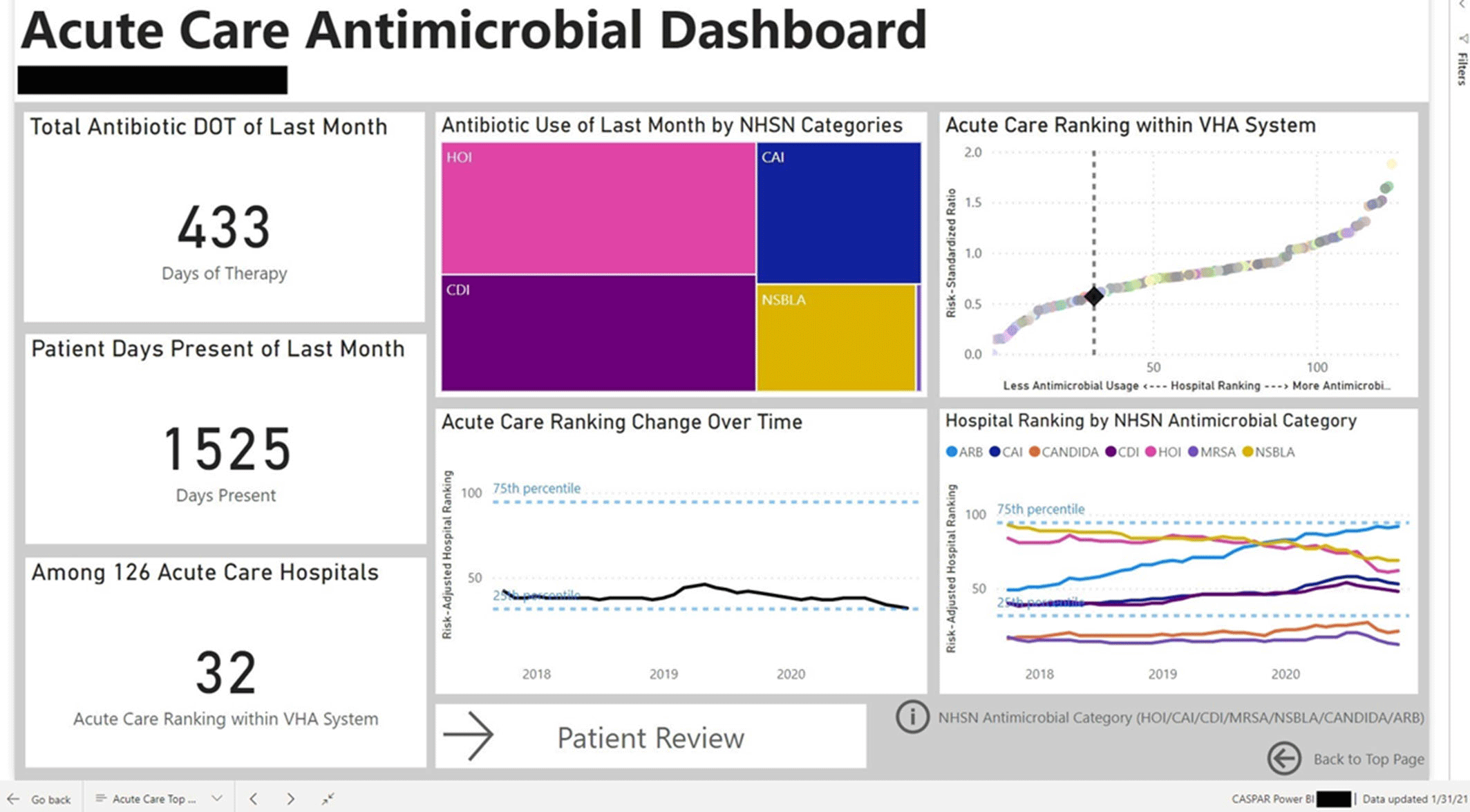
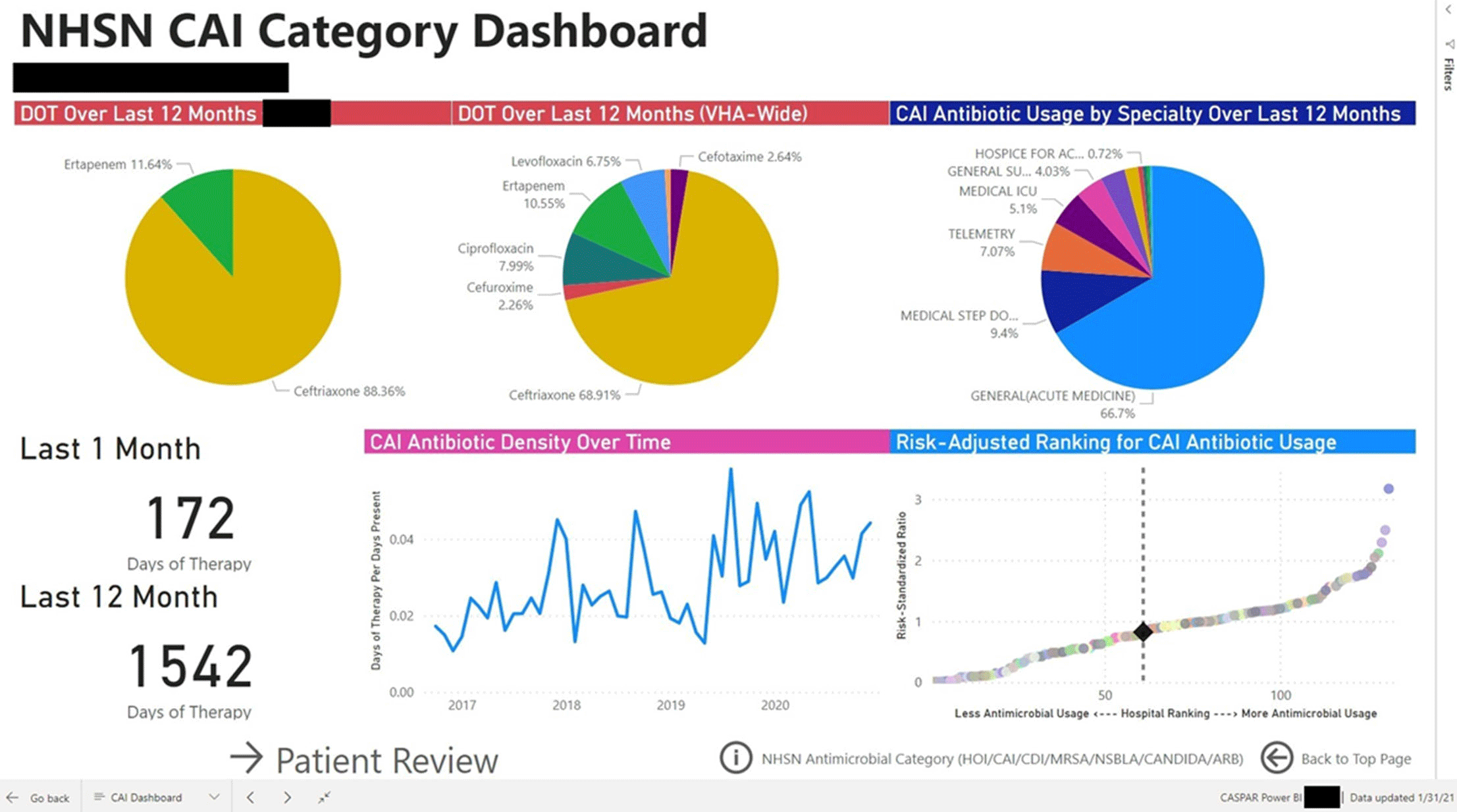
Automated Nationwide Benchmarking Dashboard for Antimicrobial Stewardship Programs within the Veterans’ Health Administration
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s23-s24
-
- Article
-
- You have access
- Open access
- Export citation