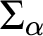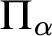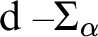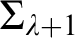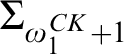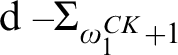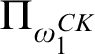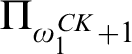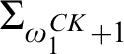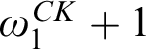15 results
Consolidated health economic evaluation reporting standards 2022 (CHEERS 2022) statement: updated reporting guidance for health economic evaluations
-
- Journal:
- International Journal of Technology Assessment in Health Care / Volume 38 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 11 January 2022, e13
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
SCOTT COMPLEXITY OF COUNTABLE STRUCTURES
- Part of
-
- Journal:
- The Journal of Symbolic Logic / Volume 86 / Issue 4 / December 2021
- Published online by Cambridge University Press:
- 01 February 2021, pp. 1706-1720
- Print publication:
- December 2021
-
- Article
- Export citation
OP230 How Legitimate Is The Process Of Updating the Benefits Package In Israel? A 20 Year Overview
-
- Journal:
- International Journal of Technology Assessment in Health Care / Volume 36 / Issue S1 / December 2020
- Published online by Cambridge University Press:
- 28 December 2020, pp. 3-4
-
- Article
-
- You have access
- Export citation
RELATIONSHIPS BETWEEN COMPUTABILITY-THEORETIC PROPERTIES OF PROBLEMS
- Part of
-
- Journal:
- The Journal of Symbolic Logic / Volume 87 / Issue 1 / March 2022
- Published online by Cambridge University Press:
- 05 October 2020, pp. 47-71
- Print publication:
- March 2022
-
- Article
- Export citation
STRONG JUMP-TRACEABILITY
-
- Journal:
- Bulletin of Symbolic Logic / Volume 24 / Issue 2 / June 2018
- Published online by Cambridge University Press:
- 07 August 2018, pp. 147-164
- Print publication:
- June 2018
-
- Article
- Export citation
The finite intersection principle and genericity
-
- Journal:
- Mathematical Proceedings of the Cambridge Philosophical Society / Volume 160 / Issue 2 / March 2016
- Published online by Cambridge University Press:
- 26 November 2015, pp. 279-297
- Print publication:
- March 2016
-
- Article
- Export citation
CHARACTERIZING LOWNESS FOR DEMUTH RANDOMNESS
-
- Journal:
- The Journal of Symbolic Logic / Volume 79 / Issue 2 / June 2014
- Published online by Cambridge University Press:
- 25 June 2014, pp. 526-560
- Print publication:
- June 2014
-
- Article
- Export citation
COMPUTING K-TRIVIAL SETS BY INCOMPLETE RANDOM SETS
-
- Journal:
- Bulletin of Symbolic Logic / Volume 20 / Issue 1 / March 2014
- Published online by Cambridge University Press:
- 13 May 2014, pp. 80-90
- Print publication:
- March 2014
-
- Article
- Export citation
CONSOLIDATED HEALTH ECONOMIC EVALUATION REPORTING STANDARDS (CHEERS) STATEMENT
-
- Journal:
- International Journal of Technology Assessment in Health Care / Volume 29 / Issue 2 / April 2013
- Published online by Cambridge University Press:
- 15 April 2013, pp. 117-122
-
- Article
- Export citation
Rogues' Gallery of Contributing Authors
-
-
- Book:
- Core Clinical Competencies in Anesthesiology
- Published online:
- 06 July 2010
- Print publication:
- 12 April 2010, pp xi-xii
-
- Chapter
- Export citation
The process of updating the National List of Health Services in Israel: Is it legitimate? Is it fair?
-
- Journal:
- International Journal of Technology Assessment in Health Care / Volume 25 / Issue 3 / July 2009
- Published online by Cambridge University Press:
- 21 July 2009, pp. 255-261
-
- Article
- Export citation
Diffusion of published cost-utility analyses in the field of health policy and practice
-
- Journal:
- International Journal of Technology Assessment in Health Care / Volume 21 / Issue 3 / July 2005
- Published online by Cambridge University Press:
- 04 August 2005, pp. 399-402
-
- Article
- Export citation
Decisions to adopt new technologies at the hospital level: Insights from Israeli medical centers
-
- Journal:
- International Journal of Technology Assessment in Health Care / Volume 21 / Issue 2 / April 2005
- Published online by Cambridge University Press:
- 26 April 2005, pp. 219-227
-
- Article
- Export citation
DECISION MAKING IN ACQUIRING MEDICAL TECHNOLOGIES IN ISRAELI MEDICAL CENTERS: A Preliminary Study
-
- Journal:
- International Journal of Technology Assessment in Health Care / Volume 19 / Issue 1 / January 2003
- Published online by Cambridge University Press:
- 22 January 2003, pp. 194-201
-
- Article
- Export citation
PREFERENCE-BASED OUTCOME MEASURES IN COST-UTILITY ANALYSES: A 20-year Overview
-
- Journal:
- International Journal of Technology Assessment in Health Care / Volume 18 / Issue 3 / 22 2002
- Published online by Cambridge University Press:
- 19 December 2002, pp. 461-466
-
- Article
- Export citation