38 results
Impact of infectious diseases consultation for hospitalized patients with Clostridioides difficile infection
-
- Journal:
- Infection Control & Hospital Epidemiology , First View
- Published online by Cambridge University Press:
- 15 February 2024, pp. 1-4
-
- Article
- Export citation
Introduction to A Compendium of Strategies to Prevent Healthcare-Associated Infections In Acute-Care Hospitals: 2022 Updates
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 10 / October 2023
- Published online by Cambridge University Press:
- 19 October 2023, pp. 1533-1539
- Print publication:
- October 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
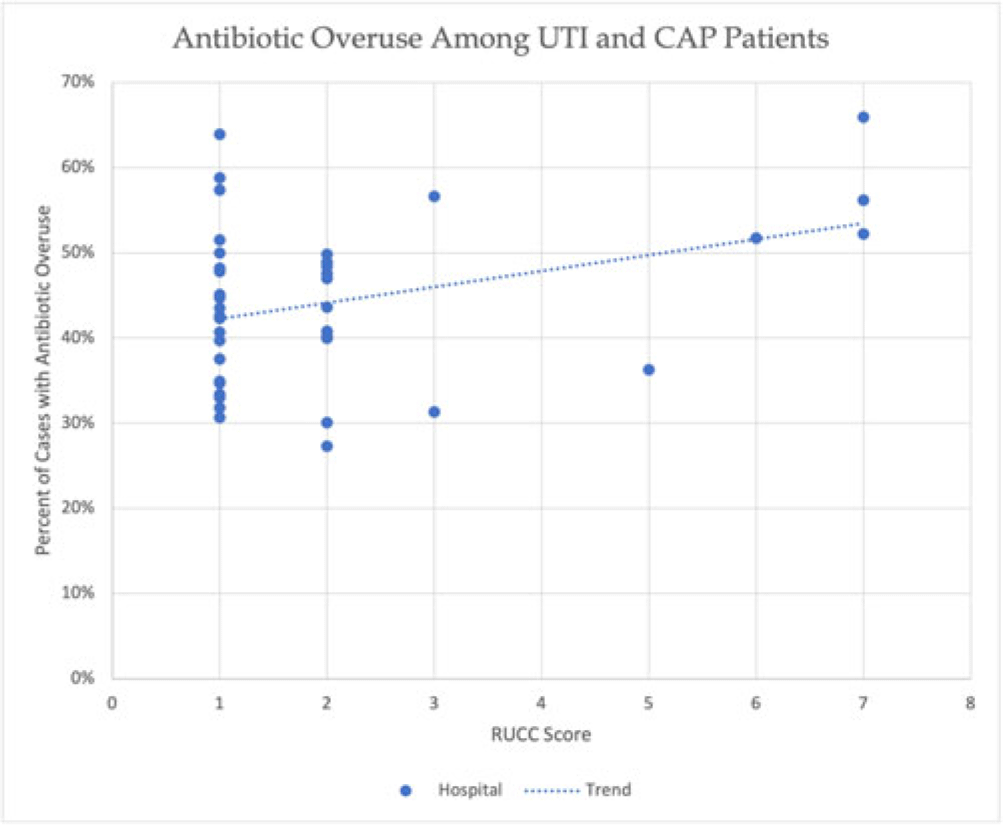
Identifying the relationship between hospital rurality and antibiotic overuse
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s34-s35
-
- Article
-
- You have access
- Open access
- Export citation
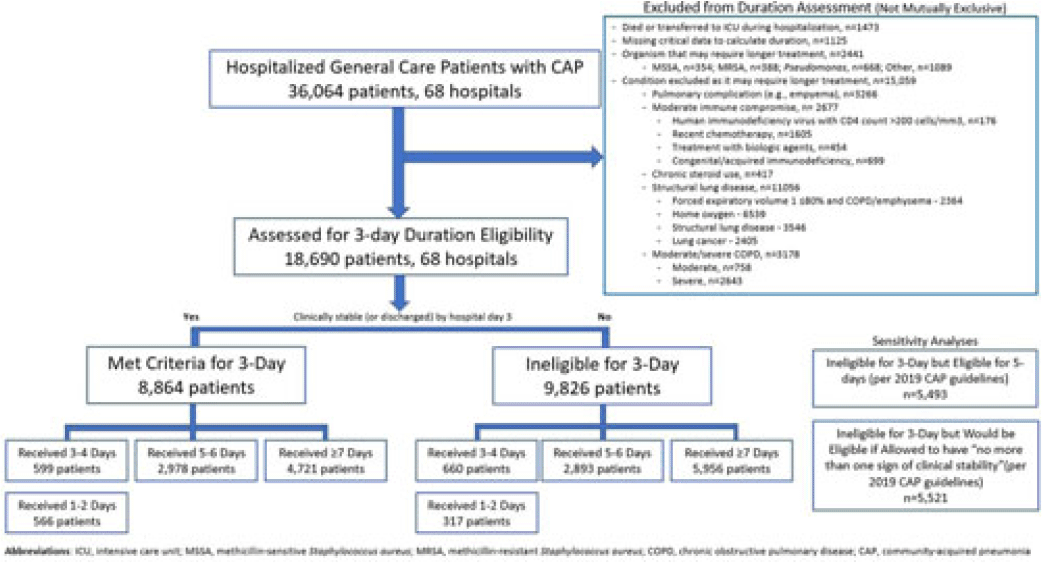
Three-day antibiotic duration in patients with pneumonia: A sixty-eight–hospital cohort
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s22
-
- Article
-
- You have access
- Open access
- Export citation
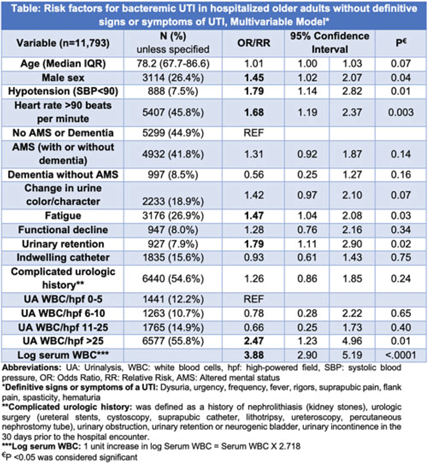
Prevalence of and risk factors for bacteremic UTIs in hospitalized adults without definitive signs or symptoms of UTI
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s27-s28
-
- Article
-
- You have access
- Open access
- Export citation
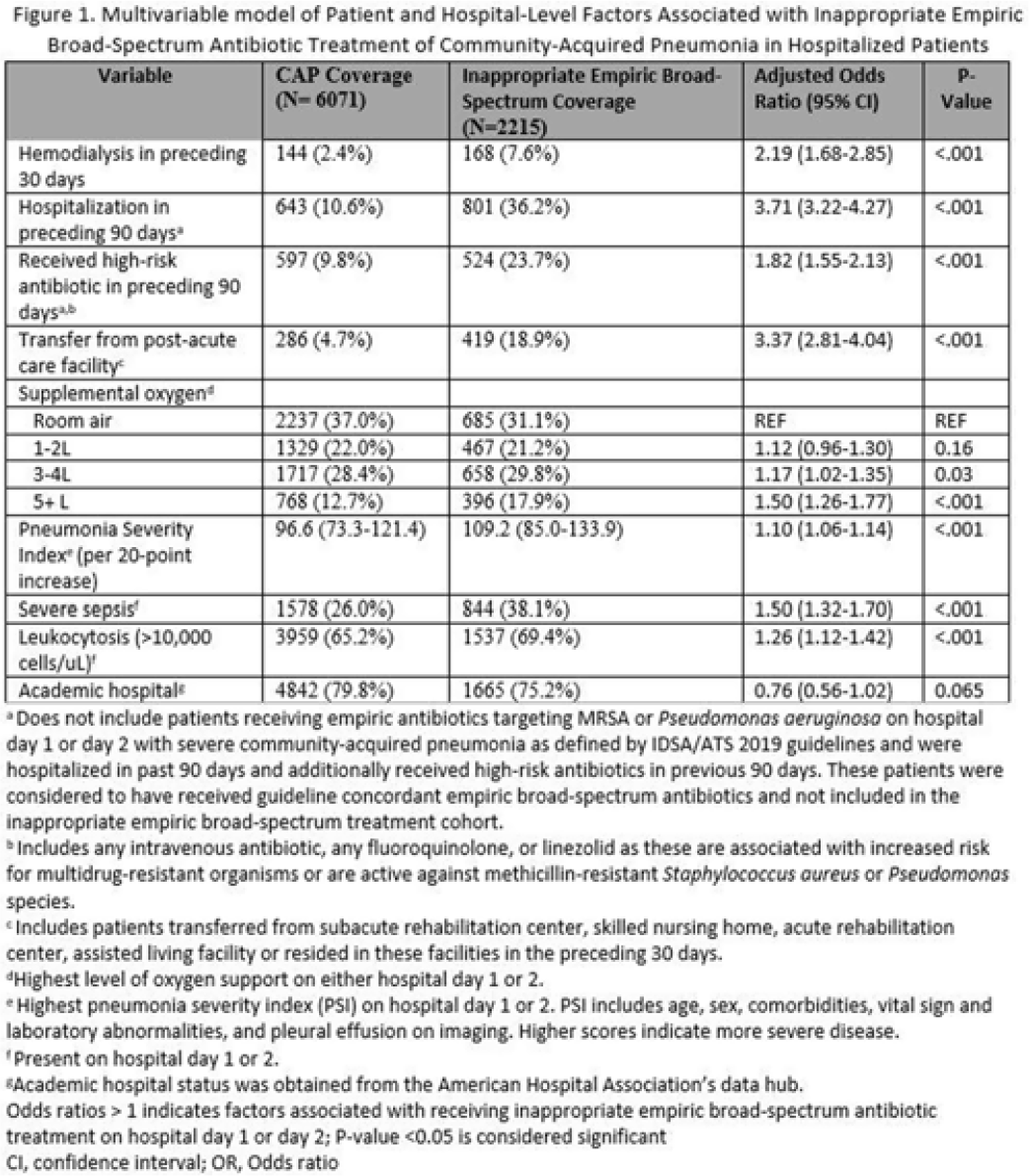
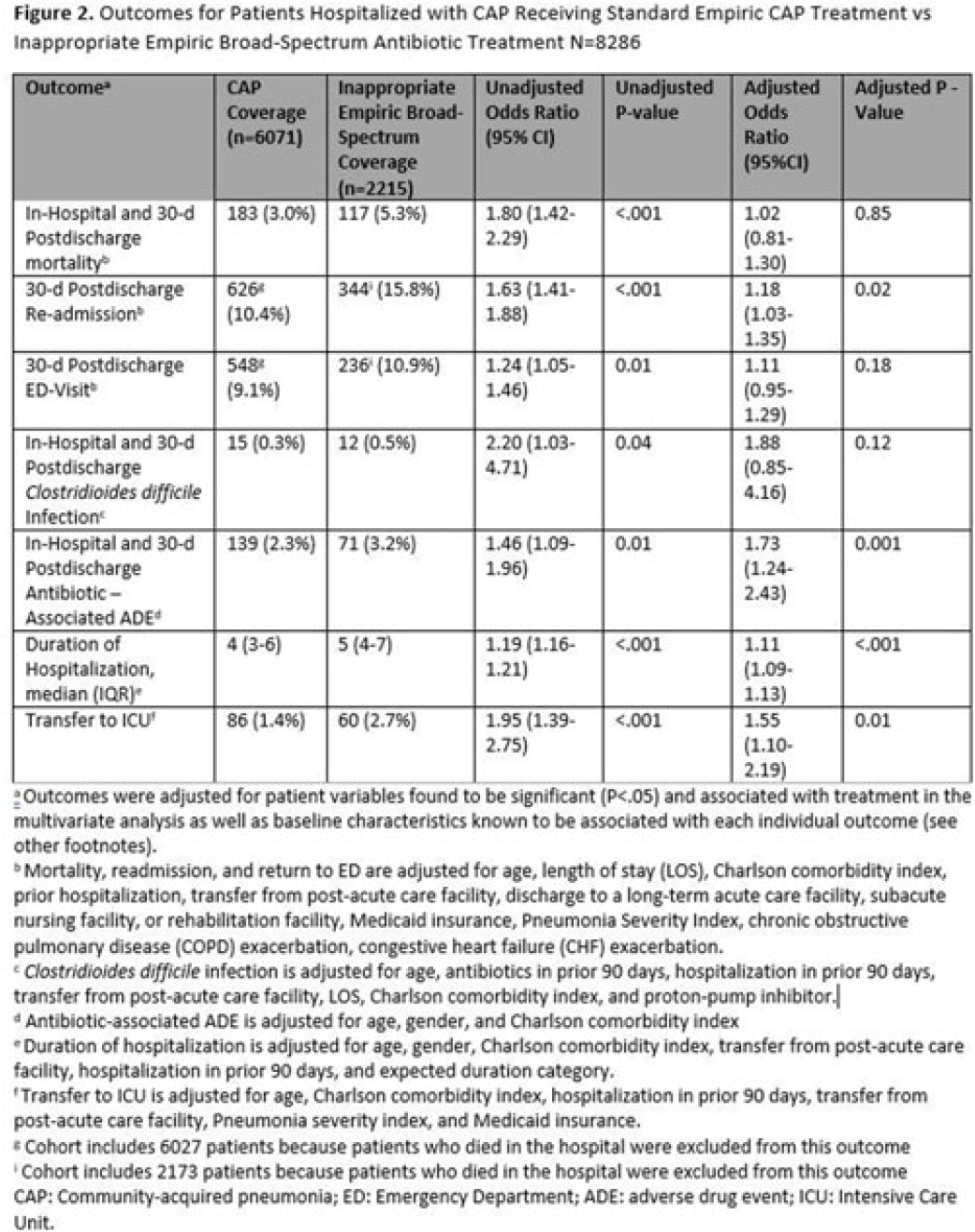
Risk Factors and outcomes associated with inappropriate empiric broad-spectrum antibiotic use in hospitalized patients with community-acquired pneumonia
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s31-s32
-
- Article
-
- You have access
- Open access
- Export citation
Executive Summary: A Compendium of Strategies to Prevent Healthcare-Associated Infections in Acute-Care Hospitals: 2022 Updates
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 10 / October 2023
- Published online by Cambridge University Press:
- 22 August 2023, pp. 1540-1554
- Print publication:
- October 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
A bronchoscopy-associated pseudo-outbreak of Mycobacterium chelonae and Mycobacterium mucogenicum associated with contaminated ice machine water and ice
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 12 / December 2023
- Published online by Cambridge University Press:
- 05 June 2023, pp. 2056-2058
- Print publication:
- December 2023
-
- Article
- Export citation
Asymptomatic screening for severe acute respiratory coronavirus virus 2 (SARS-CoV-2) as an infection prevention measure in healthcare facilities: Challenges and considerations
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 1 / January 2023
- Published online by Cambridge University Press:
- 21 December 2022, pp. 2-7
- Print publication:
- January 2023
-
- Article
-
- You have access
- HTML
- Export citation
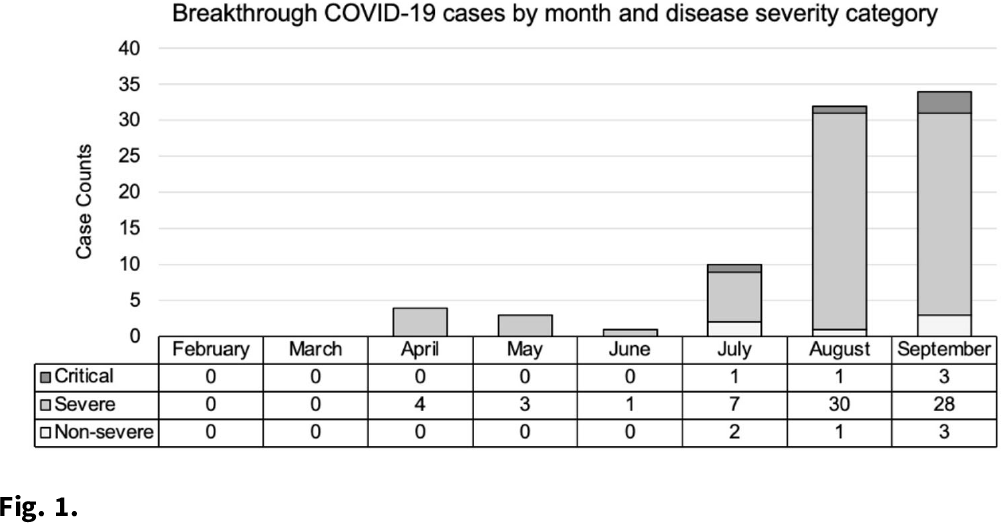
SARS-CoV-2 breakthrough infections among hospitalized patients in southeastern Michigan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s40-s41
-
- Article
-
- You have access
- Open access
- Export citation
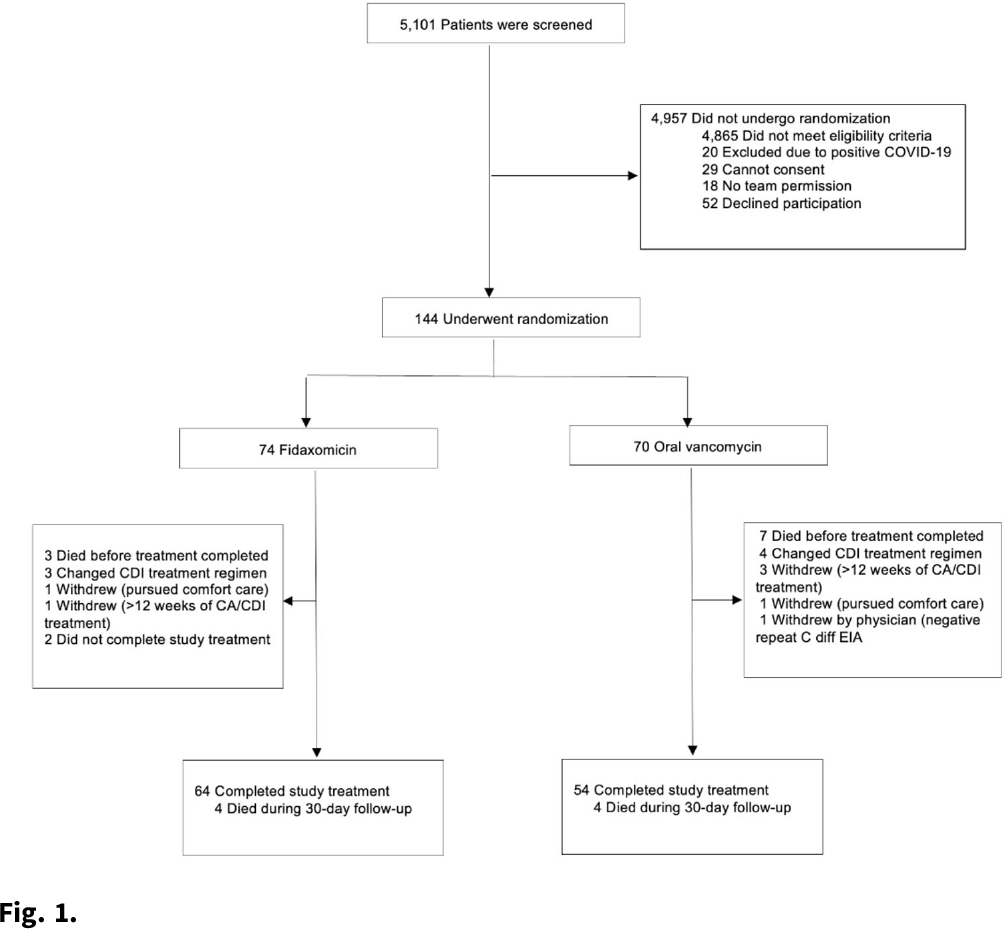
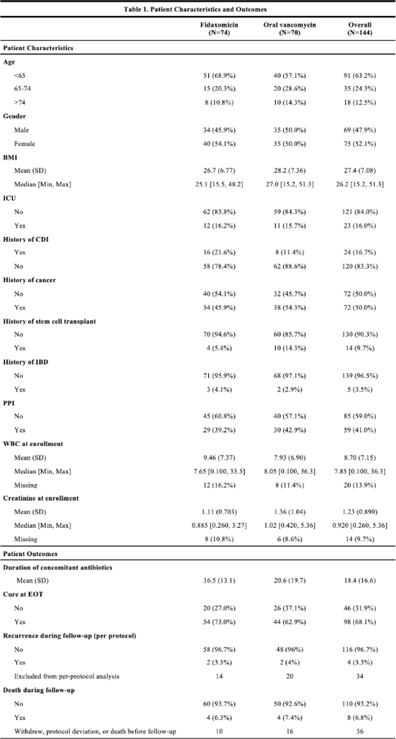
Comparison of fidaxomicin to oral vancomycin for the treatment of Clostridioides difficile infection in hospitalized patients
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s72-s73
-
- Article
-
- You have access
- Open access
- Export citation
Risk factors and outcomes associated with community-onset and hospital-acquired coinfection in patients hospitalized for coronavirus disease 2019 (COVID-19): A multihospital cohort study
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 9 / September 2022
- Published online by Cambridge University Press:
- 26 July 2021, pp. 1184-1193
- Print publication:
- September 2022
-
- Article
- Export citation
Multisociety statement on coronavirus disease 2019 (COVID-19) vaccination as a condition of employment for healthcare personnel
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 1 / January 2022
- Published online by Cambridge University Press:
- 13 July 2021, pp. 3-11
- Print publication:
- January 2022
-
- Article
-
- You have access
- HTML
- Export citation
Assessing the healthcare epidemiology environment—A roadmap for SHEA’s future
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 9 / September 2021
- Published online by Cambridge University Press:
- 18 June 2021, pp. 1111-1114
- Print publication:
- September 2021
-
- Article
- Export citation
Local, state and federal face mask mandates during the COVID-19 pandemic
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 4 / April 2021
- Published online by Cambridge University Press:
- 05 January 2021, pp. 455-456
- Print publication:
- April 2021
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Service, science, and fortitude: Our thanks and salute to Dr. Anthony S. Fauci, October 2020
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 3 / March 2021
- Published online by Cambridge University Press:
- 05 November 2020, p. 331
- Print publication:
- March 2021
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Preparing nursing homes for a second wave of coronavirus disease 2019 (COVID-19)
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 10 / October 2021
- Published online by Cambridge University Press:
- 20 October 2020, pp. 1251-1254
- Print publication:
- October 2021
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Antibiotic Overuse at Discharge in Hospitalized Patients with Bacteriuria or Treated for Pneumonia: A Multihospital Study
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s459-s461
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Influence of Infectious Disease Physician Approval on Appropriateness of PICC Use and Outcomes
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s485-s487
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
When Legionnaires’ Disease Isn’t: Case Presentation and Implications of the Council of State and Territorial Epidemiologists (CSTE) Changes to Case Definitions
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s512-s513
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation