1. Introduction
The first fossils of the gigantic, semiaquatic predatory dinosaur Spinosaurus aegyptiacus, which lived during the Cretaceous period c. 97 million years ago and was larger than Tyrannosaurus rex, were described by Stromer in Reference Stromer1915. He was perplexed by the presence of many large predators such as crocodilia, despite the lack of any major terrestrial herbivores (Martill et al. Reference Martill, Ibrahim, Brito, Baider, Zhouri, Loveridge, Naish and Hing2011). Unfortunately, the remains collected by Stromer were lost during the Second World War. Based on newly collected items, a fascinating account of Spinosaurus was recently given by Ibrahim et al. (Reference Ibrahim, Sereno, Sasso, Maganuco, Fabbri, Martill, Zouhri, Myhrvold and Iurino2014) who have provided arguments for Spinosaurus’ skill in swimming and aquatic hunting, including the adapted anatomical proportions, a centre of body mass towards the front, high-density bones, flattened pedal unguals, a longirostral piscivorous snout and a small nostril located far back on the head. Their findings, together with our knowledge of the presence of large river systems in North Africa at the time of Spinosaurus, point to Spinosaurus’ semiaquatic life and its aquatic prey.
Nevertheless, after solving Stromer's old riddle, Ibrahim et al. (Reference Ibrahim, Sereno, Sasso, Maganuco, Fabbri, Martill, Zouhri, Myhrvold and Iurino2014) were confronted by another inexplicable puzzle: the dorsal ‘sail’, supported by major spinal processes. The authors discussed three hypotheses. The first, that the sail was a thermoregulatory structure, was dismissed because of the lack of canals for blood vessels in the spinal processes. A second, that the spines supported a muscle or fat-lined hump (Bailey, Reference Bailey1997) was dismissed in favour of Stromer's (Reference Stromer1915) hypothesis of convergent evolution with the skin-covered neural spines of the crested chameleon. Based on the idea that the sail was tightly enveloped in skin, the authors proposed that it was used largely for display on land and in water to deter foes and competitors or to impress potential sexual partners, and that it would have remained visible while swimming.
We would like to extend this hypothesis. In water, Spinosaurus’ semiaquatic life would always have risked submerging the sail by accident. The sail would unleash completely different forces under water than when used for display in air. Accordingly, the anatomical characteristics of the animal must have taken such occurrences into account. Given these features, the question of why the animal would not have taken advantage of a hydrodynamically stable sail must be asked.
Spinosaurus’ sail was almost rectangular, unlike the rounded, arc-shaped sails of related non-aquatic spinosaurids (see Ibrahim et al. Reference Ibrahim, Sereno, Sasso, Maganuco, Fabbri, Martill, Zouhri, Myhrvold and Iurino2014). The size and shape of the spinal sail relative to Spinosaurus’ body roughly resembles the anatomical geometry of the dorsal fins of sailfish (Domenici et al. Reference Domenici, Wilson, Kurvers, Marras, Herbert-Read, Steffensen, Krause, Viblanc, Couillaud and Krause2014), which in our view suggests homologous functions.
2. Discussion
Despite their poorer adaptation to semiaquatic life, earlier spinosaurids were assumed to be predominantly piscivorous (Ibrahim et al. Reference Ibrahim, Sereno, Sasso, Maganuco, Fabbri, Martill, Zouhri, Myhrvold and Iurino2014). Spinosaurus’ advanced adaptation together with other anatomical features – for example its skull's ability to attack or grab prey under water through dorsoventral excursions, or its bone density, resembling the degree of adaptation of the modern hippopotamus and early whales to an aquatic life (Ibrahim et al. Reference Ibrahim, Sereno, Sasso, Maganuco, Fabbri, Martill, Zouhri, Myhrvold and Iurino2014) – lead us to assume that Spinosaurus was both a deep-water hunter and an excellent swimmer. Our view is supported by Amiot et al. (Reference Amiot, Buffetaut, Lecuyer, Wang, Boudad, Ding, Fourel, Hutt, Martineau, Medeiros, Mo, Simon, Suteethorn, Sweetman, Tong, Zhang and Zhou2010) who argued that oxygen isotopes suggest that spinosaurids closely related to Spinosaurus were probably aquatic.
Comparison of the feeding mechanics of different spinosaurids with existing crocodilians by Cuff & Rayfield (Reference Cuff and Rayfield2013) suggests that spinosaurids were not mandatory piscivores, with their diet determined by individual animal size. Nevertheless, Foffa et al. (Reference Foffa, Cuff, Sassoon, Rayfield, Mavrogordato and Benton2014) believe that large size alone made the spinosaurids omnivores. The narrow spinosaurid rostrum is distinct from that of every other theropod dinosaur. When corrected for size and compared to that of modern crocodiles, the narrow Spinosaurus snout is closest to that of the gharial (Gavialis gangeticus), which is almost exclusively piscivorous. Gharials use rapid, swiping lateral strikes of the head to capture fish. For more efficient ‘lateral grabbing’, Spinosaurus’ rostrum seems to form two additional, laterally outwards-directed rows of incisors for snapping with while rapidly turning the head. Other evidence for the piscivory of Spinosaurus includes a large claw on manual digit I that may have functioned as a hook for catching fish and fish scales etched by gastric acid found in a spinosaurid rib cage (Cuff & Rayfield, Reference Cuff and Rayfield2013).
To develop another anatomical argument, let us assume that the dorsal sail had been intended solely for display. On land it would have been a hindrance because it could not be folded like a peacock's fan, a fact that might have been problematic (especially for more vulnerable juveniles). In water, the sail would have needed to survive accidental submerging and provide the stability required to avoid spinal injuries from lateral bending by hydrodynamic forces. Injury of the sail's spinal processes would have destabilized the peripheral spinal tendons at the tips of the spinal processes, leading to destabilization of the neck and tail. The subsequent canting of the vertebrae might have resulted in injuries to the spinal cord. In other words: an insufficiently hydrodynamically stable sail was not an option.
These considerations have led us to the hypothesis that the dorsal sail of Spinosaurus had to be submergible. If so, then hydrodynamically speaking the sail was homologous to the dorsal fins of sailfish (Fig. 1; Domenici et al. Reference Domenici, Wilson, Kurvers, Marras, Herbert-Read, Steffensen, Krause, Viblanc, Couillaud and Krause2014). Sailfish, together with marlin and swordfish, belong to the two families of billfish characterized by their prominent bills and large size, weighing up to 1000 kg and as large as 4 m in length. All billfish are apex predators feeding on a wide variety of smaller prey such as fish, crustaceans and cephalopods, just like Spinosaurus. They also use their bills to hunt mackerel and tuna, some of the fastest seawater fish of all. Swordfish are known to charge through schools of fish while slashing with their swords to kill or stun their prey before returning to consume them (Helfman, Collette & Facey, Reference Helfman, Collette and Facey1997). To reduce hydrodynamic friction for fast swimming, sailfish can fully withdraw their dorsal and long pectoral fins, unlike other billfish (Domenici et al. Reference Domenici, Wilson, Kurvers, Marras, Herbert-Read, Steffensen, Krause, Viblanc, Couillaud and Krause2014).
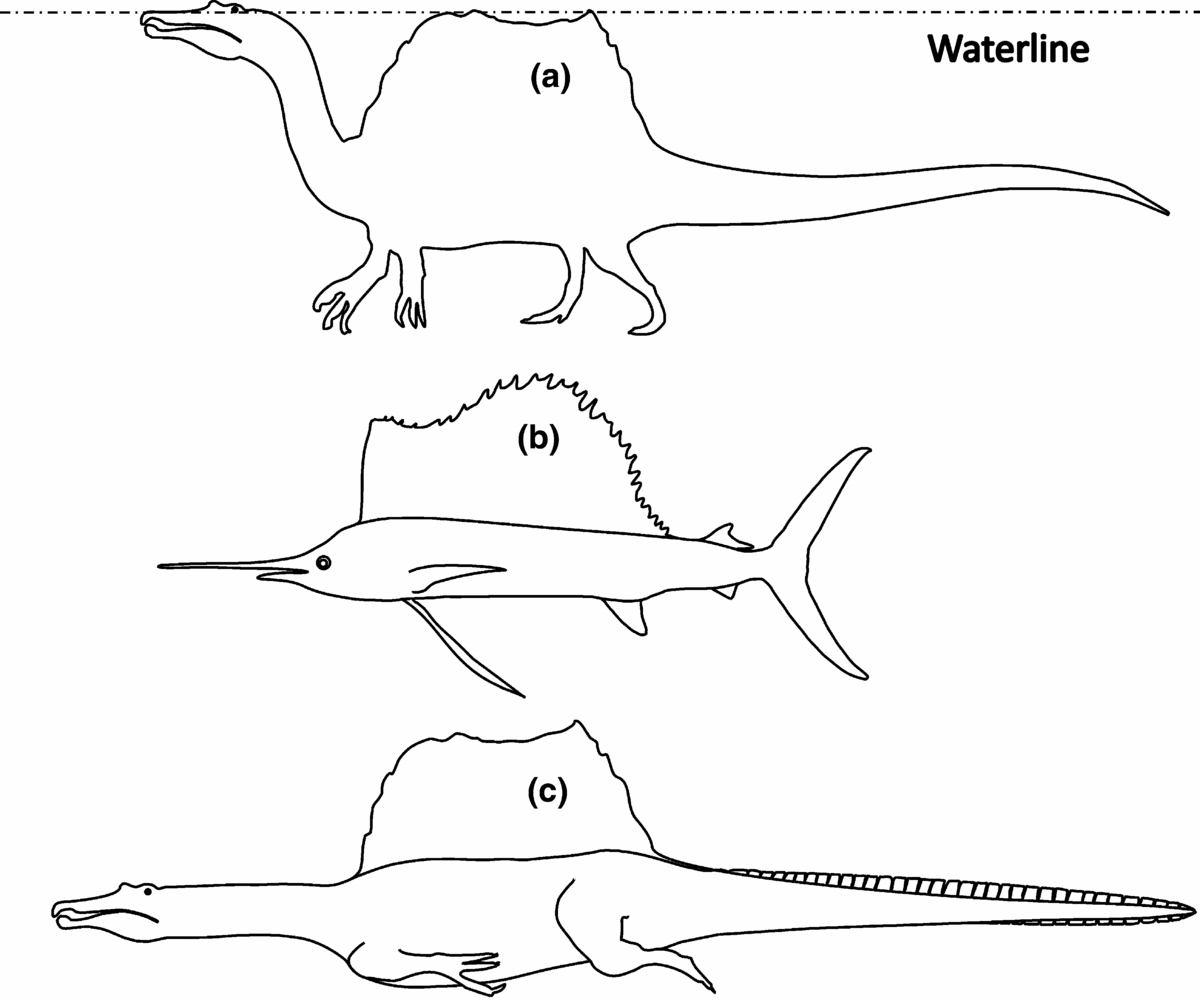
Figure 1. Body shapes of Spinosaurus aegyptiacus and sailfish. Circumferences were drawn in line with the skeleton published in Ibrahim et al. (Reference Ibrahim, Sereno, Sasso, Maganuco, Fabbri, Martill, Zouhri, Myhrvold and Iurino2014), and averaged from images in the internet. (a) Spinosaurus in resting or stalking posture. (b) Sailfish with sail raised. (c) Spinosaurus swimming submerged. The hind leg is shown in stroke position and the tail is shown extended with hypothetical crocodile-like horny scales, improving the efficiency of the undulating propulsion.
It remains to be determined whether Spinosaurus mainly submerged the sail for predation or while swimming longer distances, during acceleration or at various velocities. To approach this problem, the velocity-dependent costs of locomotion need to be compared for swimming near the surface with the sail out of the water to avoid sail friction as against the additional hydrodynamic costs of the submerged sail (Blake, Reference Blake1983).
If Spinosaurus submerged the sail for predation, it might be assumed that special tendon and skeletal features existed to absorb any shocks and lateral bending forces acting on the sail. Shock-absorbing bone structures in swordfish are known. Gudger (Reference Gudger1940) described incidents of swordfish that were unable to extract their swords from the hulls of rammed vessels. They broke off, leaving their swords behind; no fish has ever been found with a broken neck or a dislocated spinal column, however. Gudger (Reference Gudger1940) found the anatomical reason for this stability in honeycombed ethmoid bones filled with oil at the base of the sword, acting as a multiple-cell shock absorber. Here we hypothesize that the pronounced proximal flanges, which are especially pronounced in Spinosaurus, may have had a similar structure and an analogous function (Ibrahim et al. Reference Ibrahim, Sereno, Sasso, Maganuco, Fabbri, Martill, Zouhri, Myhrvold and Iurino2014).
Spinosaurus’ anatomy exhibits another feature that may have a modern homology: its long tail resembled that of the thresher shark, employed to slap the water to herd and stun shoals of fish before devouring them (Oliver et al. Reference Oliver, Turner, Gann, Silvosa and D’Urban Jackson2013).
The strategies that sailfish and thresher sharks employ against shoaling fish are more effective when the shoal is first concentrated into a ‘bait ball’ (Helfman, Collette & Facey, Reference Helfman, Collette and Facey1997; Oliver et al. Reference Oliver, Turner, Gann, Silvosa and D’Urban Jackson2013; Domenici et al. Reference Domenici, Wilson, Kurvers, Marras, Herbert-Read, Steffensen, Krause, Viblanc, Couillaud and Krause2014). Since this is difficult for individual predators to achieve, they cooperate in this effort. When herding a shoal of fish or squid, sailfish also raise their sails to make themselves appear larger. When they slash or wipe their bills through shoaling fish by turning their heads, their dorsal sail and fins are outstretched to stabilize their bodies hydrodynamically (Lauder & Drucker, Reference Lauder and Drucker2004). Domenici et al. (Reference Domenici, Wilson, Kurvers, Marras, Herbert-Read, Steffensen, Krause, Viblanc, Couillaud and Krause2014) postulate that these fin extensions enhance the accuracy of tapping and slashing. The sail can reduce yaw rotation by counteracting the lateral force in the direction opposite to the slash. This means that prey is less likely to recognize the massive trunk as being part of an approaching predator (Marras et al. Reference Marras, Noda, Steffensen, Svendsen, Krause, Wilson, Kurvers, Herbert-Read, Boswell and Domenici2015; Webb & Weihs Reference Webb and Weihs2015). Film footage available online impressively demonstrates the hunting strategies of sailfish and thresher sharks.
Interestingly, Spinosaurus exhibited the anatomical features required to combine all three hunting strategies: a sail for herding prey more efficiently, as well as flexible tail and neck to slap the water for stunning, injuring or killing prey. The submerged dorsal sail would have provided a strong centreboard-like counterforce for powerful sidewards movements of the strong neck and long tail, as performed by sailfish (Domenici et al. Reference Domenici, Wilson, Kurvers, Marras, Herbert-Read, Steffensen, Krause, Viblanc, Couillaud and Krause2014) or thresher sharks (Oliver et al. Reference Oliver, Turner, Gann, Silvosa and D’Urban Jackson2013). While smaller dorsal sails or fins make the dorsal water volume better accessible for slashing, it can be speculated that their smaller stabilization effect makes lateral slashing less efficient (e.g. for thresher sharks). Forming a hydrodynamic fulcrum and hydrodynamically stabilizing the trunk along the dorsoventral axis, Spinosaurus’ sail would also have compensated for the inertia of the lateral neck by tail movements and vice versa not only for predation but also for accelerated swimming. This behaviour might also have been one reason for Spinosaurus’ muscular chest and neck reported by Ibrahim et al. (Reference Ibrahim, Sereno, Sasso, Maganuco, Fabbri, Martill, Zouhri, Myhrvold and Iurino2014).
When feeding on smaller prey, it is not the ability of a predator to swim rapidly that is important but rather its manoeuvrability and precise head movements. The ability to perform quick movements with a long rostrum is a known key factor for aquatic predators (Foffa et al. Reference Foffa, Cuff, Sassoon, Rayfield, Mavrogordato and Benton2014). This view is backed by new findings on the hunting behaviour of billfish (Marras et al. Reference Marras, Noda, Steffensen, Svendsen, Krause, Wilson, Kurvers, Herbert-Read, Boswell and Domenici2015).
One additional benefit of the submerged sail was the hydrodynamic stabilization of the trunk against tilt around the craniocaudal axis. For the increased reach and radius of head movements, this stabilization must have been effective for lateral movements with a stretched or slightly arched neck at certain angles with respect to the craniocaudal axis. Especially when grabbing prey at dorsoventral locations underwater, the lowest underwater points of the feeding envelope (compare to Schwarz-Wings, Reference Schwarz-Wings2014) would become more comfortable for the animal despite the increasing torque induced by lateral neck movement around the craniocaudal axis. The submerged sail would have counteracted the tilt, practically increasing the reach and swiftness of the head: a prerequisite for slashing fish and the efficient exploitation of Spinosaurus’ large underwater feeding envelope. Clearly, if the animal surfaced in order to display the sail, this would have moved the top part of the feeding envelope into the air, making the creatures ineffective in pursuing aqueous prey.
Ethological reasons for submerging the sail also exist: a dorsal sail protruding from the water would not have allowed Spinosaurus to stalk prey on land from the water. Behaving like crocodiles, which sneak up while submerged (hidden except for their nose and eyes), would seem a more reasonable assumption. A submerged sail would naturally be more efficient to encircle aquatic prey, and this efficiency could have been further increased when hunting in groups as practised by today's sailfish and thresher sharks against shoaling fish (Oliver et al. Reference Oliver, Turner, Gann, Silvosa and D’Urban Jackson2013; Domenici et al. Reference Domenici, Wilson, Kurvers, Marras, Herbert-Read, Steffensen, Krause, Viblanc, Couillaud and Krause2014).
Finally, let us consider Spinosaurus’ diving abilities. For homeothermic, air-breathing animals with similar proportions, simplified allometric considerations show a proportionality of the basic metabolic rate to mass to the power of 2/3 (Heldmaier, Neuweiler & Rössler, Reference Heldmaier, Neuweiler and Rössler2013). While the mass-specific basic metabolic rate is reduced for larger animals, their oxygen is stored in lungs which, along with bodily fluids and tissue, scale isometrically to body mass (White & Seymour, Reference White and Seymour2005). Accordingly, an animal with twice the characteristic length of a geometrically similar animal could submerge for around twice as long. Even though body mass is a poor determinant of the diving times of reptiles, which may assume a resting state with reduced heart rates and oxygen consumption (Bennett & Dawson, Reference Bennett, Dawson, Gans and Dawson1976; Hochscheid et al. Reference Hochscheid, McMahon, Bradshaw, Maffucci, Bentivegna and Hays2007; Seebacher, Franklin & Read, Reference Seebacher, Franklin and Read2005; Campbell et al. Reference Campbell, Sullivan, Read, Gordos and Franklin2010), it is tempting to assume that Spinosaurus took advantage of the potentials of a submerged animal of 10 tonnes during active dives associated with swimming and hunting.
We imagine that in a hunting scenario, a Spinosaurus would slink up on a swarm of prey such as fish by means of undulating lateral tail and body movements with its extremities withdrawn (Ibrahim et al. Reference Ibrahim, Sereno, Sasso, Maganuco, Fabbri, Martill, Zouhri, Myhrvold and Iurino2014). While stunning or injuring the prey by laterally swinging its head, it would stretch its front-pedal web-footed unguals almost perpendicularly to the craniocaudal axis, like the stretched pectoral fins of sailfish, before feeding on the sinking, injured prey with dorsoventral movements of its head, with the powerful forelimbs acting as fulcrums. This behaviour would require less energy than relocating the massive body in pursuit of the injured prey. After the attack, Spinosaurus might have hovered like a newt with its extremities stretched out, kept stable along all three axes by the hydrodynamic leverage action of its dorsal sail and unguals. To restrict a ferocious hunter such as Spinosaurus to the surface, with its sail raised in the air for display, would be unsatisfactory for a starving animal and a hypothetical observer of the scene.
Future numerical simulations combining stability considerations of the sail's spines, biomechanics and hydrodynamics, as already performed for the much easier problem of the feeding biomechanics of a pliosaur (Foffa et al. Reference Foffa, Cuff, Sassoon, Rayfield, Mavrogordato and Benton2014), hold promise for determining that the main function of the dorsal sail was hydrodynamic. Nevertheless, whether Spinosauri were solitary creatures or hunted in packs (which was perhaps also connected to their age and size), whether they used just their necks or also their tails for lacerating and whether they relied on their sails as screens when encircling prey all represent additional puzzles. Certainly, when displayed the sail would have been an impressive feature of a ferocious hunter for foes and competitors alike, as Ibrahim et al. (Reference Ibrahim, Sereno, Sasso, Maganuco, Fabbri, Martill, Zouhri, Myhrvold and Iurino2014) have suggested.
Acknowledgement
This manuscript was partly motivated by discussions with colleagues of the Institute of Biology at the University of Rostock, Germany on the importance of biophysics in the education of biology students.
Declaration of interest
The authors declare no conflict of interest. There are no sources of financial support.



