Although the association between social context and late-life health and functioning is well established (Cacioppo et al., Reference Cacioppo, Hawkley, Crawford, Ernst, Burleson, Kowalewski and Berntson2002; Cohen, Reference Cohen2004), the mechanisms for these associations or how social context relates to the biological and genetic factors known to contribute to later life functioning has yet to be fully understood. The advantages of twin studies are the strengthening of causal inference through cotwin control methods (McGue et al., Reference McGue, Osler and Christensen2010), the use of biometric models to quantify genetic and environmental variance (Rijsdijk & Sham, Reference Rijsdijk and Sham2002), studying sex effects by leveraging data from opposite-sex pairs, determining the extent to which associations between risk and outcome are driven by the same genetic or the same environmental influences, and testing whether familial factors (genetic and rearing effects) on the outcome may change as a function of the exposure (van der Sluis et al., Reference van der Sluis, Posthuma and Dolan2012). As an international consortium with harmonized measures of risk, pathway and contextual factors, assessed longitudinally on a large number of twins, the Interplay of Genes and Environment across Multiple Studies (IGEMS) consortium is particularly well suited for investigating the contribution of gene–environment (GE) interplay to functioning in multiple domains across adulthood.
The IGEMS consortium includes more than 76,000 twins from 18 studies representing five countries (Sweden, Denmark, Finland, United States and Australia). The sample spans a wide age range (14–103 years at intake) and has sufficient power to address issues that typically elude most studies. IGEMS also includes a set of well-characterized longitudinal phenotypes, including measures of physical health, cognitive health and emotional health, and measures of multiple facets of adult socioeconomic status (SES; e.g., occupation, education, financial strain), as well as rearing SES, which are harmonized over time and across studies. The twin structure of the dataset permits using established twin methods to test key hypotheses on the nature of GE interplay, while the dense genotyping of a large subset of IGEMS participants allows us to confirm and extend these twin analyses through analyses of polygenic risk scores (PRSs) for health outcomes and for education. Importantly, IGEMS cohorts span multiple countries and historical periods, allowing us to determine whether models of GE interplay established at the micro (i.e., individual) level also apply at the macro (i.e., country and historical period) level.
There are two substantive emphases in the IGEMS consortium: First, socioeconomic conditions are a major social determinant of health (Freedman et al., Reference Freedman, Martin, Schoeni and Cornman2008; Mensah et al., Reference Mensah, Mokdad, Ford, Greenlund and Croft2005; Mirowsky & Ross, Reference Mirowsky and Ross2003; Sattler et al., Reference Sattler, Toro, Schonknecht and Schroder2012; Sharp & Gatz, Reference Sharp and Gatz2011). The oft-cited ‘gradient’ for SES represents the association between health and SES as continuous and monotonic, and not fully explained by poorer health among those who are impoverished (Adler et al., Reference Adler, Boyce, Chesney, Cohen, Folkman, Kahn and Syme1994). Most research focuses on individual-level SES — social status that accrues to occupational classification, education and income, as well as access to social, human and income capital. Others consider the macro-economic environment, such as the extent of social inequality in a country (Kawachi et al., Reference Kawachi, Levine, Miller, Lasch and Amick1994; Lynch et al., Reference Lynch, Smith, Kaplan and House2000, Reference Lynch, Smith, Harper, Hillemeier, Ross, Kaplan and Wolfson2004). IGEMS is unusual in integrating individual- and country-level contributors to health gradients. Further, although both genetic and environmental factors are known to contribute to the SES-health gradient (Lahey et al., Reference Lahey, D’Onofrio and Waldman2009; McGue et al., Reference McGue, Osler and Christensen2010; Rutter, Reference Rutter2009), the mechanisms by which these factors combine to influence health outcomes (GE interplay) are poorly understood.
Recent research has identified alternative models of GE interplay important to understanding health and disease (Boardman et al., Reference Boardman, Daw and Freese2013; Reiss et al., Reference Reiss, Leve and Neiderhiser2013; Shanahan & Boardman, Reference Shanahan, Boardman, Elder and Giele2009; Shanahan & Hofer, Reference Shanahan and Hofer2005). Although these models recognize that individuals inherit differential sensitivity to the environment, they differ in their environmental focus (disease-triggering effects of toxic environments vs. health-promoting benefits of favorable environments) and the expected genetic contribution to disease (maximized in adverse environments, in favorable environments or at both extremes). The differences between models of GE interplay have implications beyond resolving an academic dispute. Environmental improvements would be expected to reduce or eliminate genetically based health disparities under some models (e.g., diathesis-stress) but expand them under others (e.g., social distinction) or have a mixed impact (e.g., differential susceptibility) (Boardman et al., Reference Boardman, Daw and Freese2013; Reiss et al., Reference Reiss, Leve and Neiderhiser2013; Shanahan & Boardman, Reference Shanahan, Boardman, Elder and Giele2009; Shanahan & Hofer, Reference Shanahan and Hofer2005). Understanding whether socially enriched environments compensate for genetic vulnerability or whether they preferentially promote good health among genetically selected individuals, for example, is essential for both translating research into effective prevention strategies and anticipating consequences of social policies.
A second substantive emphasis of the IGEMS consortium is cognitive functioning in adulthood. In particular, Alzheimer’s disease and related dementias (ADRDs), along with mild cognitive impairment, present a major public health challenge due to the large numbers of people affected and lack of a clear path to prevention or cure (Katzman, Reference Katzman2004; Livingston et al., Reference Livingston, Sommerlad, Orgeta, Costafreda, Huntley, Ames and Mukadam2017; Wang et al., Reference Wang, MacDonald, Dekhtyar and Fratiglioni2017). While it is generally recognized that Alzheimer’s disease (except for rare dominantly inherited forms) is caused by multiple genetic and environmental factors, it remains unclear how these factors contribute to the disease, whether they function independently or through interactions with each other (Gatz et al., Reference Gatz, Mortimer, Fratiglioni, Johannson, Berg, Andel and Pedersen2007), the ages at which these factors have the greatest impact (Wang et al., Reference Wang, MacDonald, Dekhtyar and Fratiglioni2017), and whether these factors affect men and women equally (Mielke et al., Reference Mielke, Vemuri and Rocca2014). Moreover, it remains to be determined whether many of these factors represent modifiable targets appropriate for intervention, or actually reflect pre-existing genetic vulnerability to ADRD and its risk factors.
IGEMS Studies
From an original consortium of 8 twin studies (Pedersen et al., Reference Pedersen, Christensen, Dahl, Finkel, Franz, Gatz and Reynolds2013), IGEMS has expanded to include 18 studies from 5 countries, representing the strongest available longitudinal twin studies of adulthood and aging in the world. The total sample size is now 76,233, including both members of 10,266 monozygotic (MZ) pairs and 17,288 dizygotic (DZ) pairs, of which 5063 pairs are opposite-sex DZ. The summary below outlines the sampling principles for each study. Numbers of pairs and age ranges at intake are provided in Table 1, as well as the number of waves and length of follow-up, where appropriate. Total Ns refer to individuals and include members of incomplete pairs. Note that updates for several of the studies are included in this issue.
Table 1. Number of twins in each study included in IGEMS, by age at intake
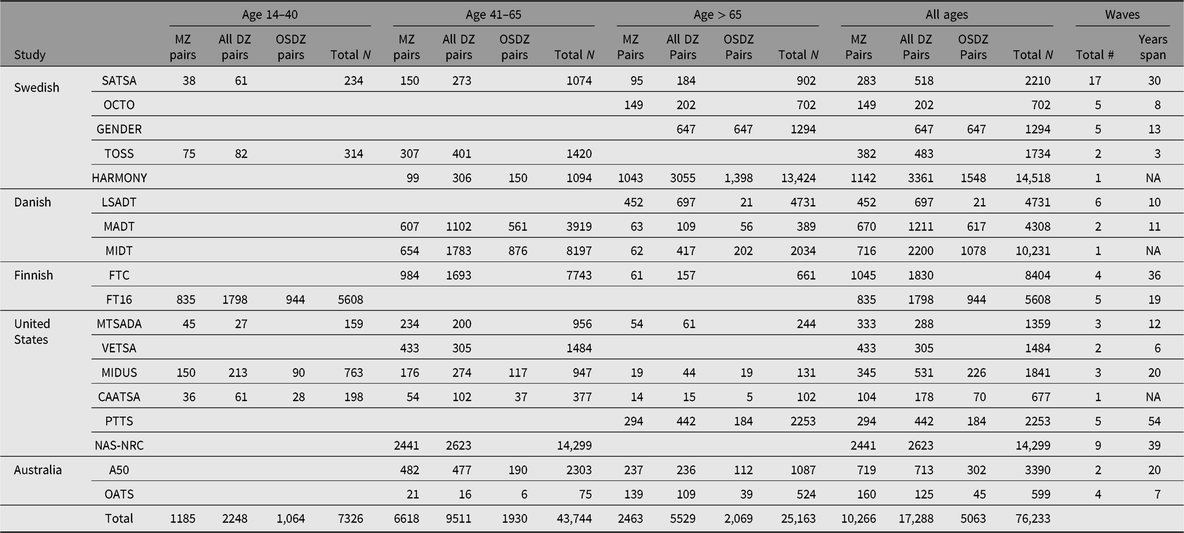
Note: MZ, monozygotic; DZ, dizygotic; OSDZ, opposite sex dizygotic pairs. Total N refers to individuals from both complete and incomplete pairs. Some individuals may have participated in more than one study, for example, in A50 and OATS. The totals in the bottom row count each pair or individual once.
Sweden
Swedish studies are drawn from the population-based Swedish Twin Registry. The Swedish Adoption/Twin Study of Aging (SATSA) began in 1984 (Finkel & Pedersen, Reference Finkel and Pedersen2004). The base population comprises all pairs of twins from the registry who indicated that they had been separated before the age of 11 and reared apart, and a sample of twins reared together matched on the basis of gender, date and county of birth. The OCTO-Twin Study (Origins of Variance in the Old-Old) included twin pairs who were over the age of 80 at baseline in 1991 (McClearn et al., Reference McClearn, Johansson, Berg, Pedersen, Ahern, Petrill and Plomin1997). Aging in Women and Men: A Longitudinal Study of Gender Differences in Health Behaviour and Health among Elderly (GENDER) is a study of opposite-sex twin pairs born between 1906 and 1925 (Gold et al., Reference Gold, Malmberg, McClearn, Pedersen and Berg2002). The Twin and Offspring Study in Sweden (TOSS) includes pairs of same-sex twins and their offspring (Neiderhiser & Lichtenstein, Reference Neiderhiser and Lichtenstein2008). The Study of Dementia in Swedish Twins (HARMONY) was conducted between 1998 and 2004. HARMONY screened all twins age 65 and over in the Screening Across the Lifespan of Twins (SALT) effort (Lichtenstein et al., Reference Lichtenstein, Sullivan, Cnattingius, Gatz, Johansson, Carlstrom and Pedersen2006) and clinically assessed those who screened positive or whose cotwin screened positive for cognitive impairment (Gatz et al., Reference Gatz, Fratiglioni, Johansson, Berg, Mortimer, Reynolds and Pedersen2005).
Denmark
The Longitudinal Study of Aging Danish Twins (LSADT) began in 1995 with the assessment of members of like-sex twin pairs born in Denmark prior to 1920; twins were recruited regardless of whether their cotwin was alive (Christensen et al., Reference Christensen, Holm, McGue, Corder and Vaupel1999). The study of Middle-Aged Danish Twins (MADTs) includes twins ranging in age from 46 to 68 years at the original assessment (Osler et al., Reference Osler, McGue, Lund and Christensen2008). The MId-aged Danish Twin (MIDT) study includes twins representing members of the Danish Twin Registry for the birth years 1931 through 1969 not already participating in MADT.
Finland
The older Finnish Twin Cohort (FTC) study spans four decades; it was initiated in 1975 by contacting all same-sex Finnish twin pairs born before 1958 with both cotwins alive in 1975 (Kaprio & Koskenvuo, Reference Kaprio and Koskenvuo2002). FinnTwin16 (FT16) is a cohort of younger twins born between 1975 and 1979. Waves 4 and 5, when the study participants were in their 20s and 30s, are included in IGEMS (Kaprio et al., Reference Kaprio, Pulkkinen and Rose2002).
United States
Each US study consists of an independent sample. The Minnesota Twin Study of Adult Development and Aging (MTSADA) is a population-based sample drawn from state birth records (Finkel & McGue, Reference Finkel and McGue1993; McGue et al., Reference McGue, Hirsch and Lykken1993). The Vietnam Era Twin Study of Aging (VETSA) is a community-dwelling sample of male–male twin pairs, all of whom served in some branch of US military service sometime between 1965 and 1975 (Kremen et al., Reference Kremen, Franz and Lyons2013). Midlife in the United States (MIDUS) is a national telephone/mail survey originally carried out in 1995–1996 that included specific recruitment methods for twins (South & Krueger, Reference South and Krueger2012). The Carolina African-American Twin Study of Aging (CAATSA) used public records to identify all living African-American twins in the State of North Carolina born between 1920 and 1970 (Whitfield, Reference Whitfield2013). The Project Talent Twin and Sibling Study (PTTS) includes 4481 twins and triplets plus 522 of their siblings, drawn from Project Talent, a longitudinal study begun in 1960 with a nationally representative sample of US high school students born 1942–1946 (Flanagan, Reference Flanagan1962). Follow-up surveys were conducted in young adulthood (ages 19, 23 and 29), at 1, 5 and 11 years following the year of expected high school graduation, and then in 2014 (aged 68–72) and 2019 (aged 73–77). The PTTS tracked 96.4% of the original PT twins (Prescott et al., Reference Prescott, Achorn, Kaiser, Mitchell, McArdle and Lapham2013). The National Academy of Sciences-National Research Council (NAS-NRC) Twin Registry consists of white male twin pairs born in the years 1917–1927, both of whom served in the armed forces, mostly during World War II (Page, Reference Page2006).
Australia
The Australian Over 50s study (A50) is based on a questionnaire mailed between 1993 and 1995 to Australian twins aged 50–95 (Hopper, Reference Hopper2002; Hopper et al., Reference Hopper, Foley, White and Pollaers2013). The Older Australian Twins Study (OATS) incorporates in-person assessments every two years of twins age 65 and older in the three eastern states of Australia: New South Wales, Victoria and Queensland (Sachdev et al., Reference Sachdev, Lammel, Trollor, Lee, Wright, Ames and Schofield2009).
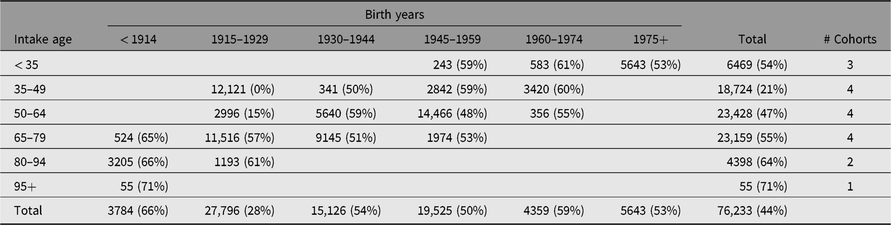
The range in study years and intake ages across the 18 IGEMS studies results in unique coverage of cohorts and historical periods. As shown in Table 2, the IGEMS sample permits sequential comparisons of sex and SES effects across six cohorts.
Table 2. Total number of individuals (% female) in each birth year range (cohort) by age at intake
IGEMS Measures
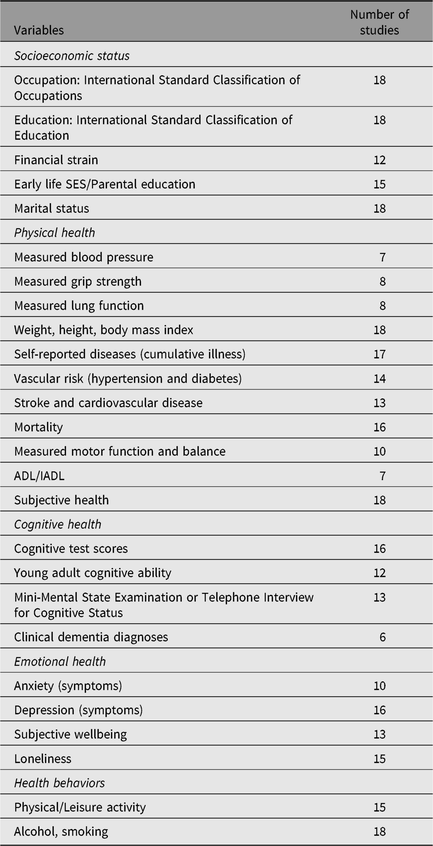
Measures used in IGEMS analyses include aging-relevant outcomes in three broad domains: physical health and functional ability (e.g., self-reported diseases, subjective health, body mass index (BMI), grip strength, motor function, activities of daily living), psychological wellbeing (e.g., depressive symptoms, anxiety symptoms, subjective wellbeing, loneliness) and cognitive health (i.e., scores on cognitive tests; dementia). Predictors and covariates include health behaviors (e.g., smoking, alcohol, physical activity, cognitively engaging leisure activity), social resources and indicators of SES. Table 3 presents a list of some of the primary phenotypes assessed and the number of IGEMS studies that include each variable.
Table 3. Number of the 18 IGEMS studies with key variables
Because participating studies differed in how similar constructs were assessed, IGEMS gives emphasis to harmonization of relevant phenotypes and outcomes. Creating scores that are common across studies enables pooling data across samples, in order to increase power. Score harmonization requires overlapping item content across studies as well as across time for longitudinal hypotheses. For some measures, it was straightforward to create a common metric, for example, BMI, lung function, and blood pressure. For harmonizing education and occupation, we have recoded all studies to the International Standard Classification of Education (UNESCO, 1997) and the International Standard Classification of Occupations (Ganzeboom et al., Reference Ganzeboom, De Graaf, Treiman and De Leeuw1992) as an international standard. Where a common metric was not already available, overlapping item content and response formats were identified and item response theory or factor-analytic techniques were implemented to create harmonized scores across studies. Where there were no common items across studies, IGEMS has collected separate samples that were administered the different measures used in different IGEMS studies to measure a given construct, with those results used to establish ‘crosswalks’ between the different scales (Gatz, Reynolds et al., Reference Gatz, Reynolds, Finkel, Hahn, Zhou and Zavala2015).
Across the 18 IGEMS studies, there is genome-wide genotype information available from 22,834 subjects, including MZ cotwins of genotyped individuals (Table 4), which will be available for analysis with appropriate correction for clustering. In Sweden, there are also genotypes available from an additional 14,498 individuals who participated in SALT substudies known as TwinGene and SALTY (Scanning Across the Lifespan of Twins — Young; Magnusson et al., Reference Magnusson, Almqvist, Rahman, Ganna, Viktorin, Walum and Larsson2013), but under age 65 when screened in SALT. Thus, the total number of individuals with genome-wide genotyping available to us is 37,332. PRSs have or will be computed for cardiovascular disease, lipids, type II diabetes, Alzheimer’s disease, neuroticism, major depression and depressive symptoms, smoking and alcohol behaviors, wellbeing and educational attainment. We will compute new PRSs as new GWAS training sets become available.
Table 4. Genotype data available in participating IGEMS twin studies
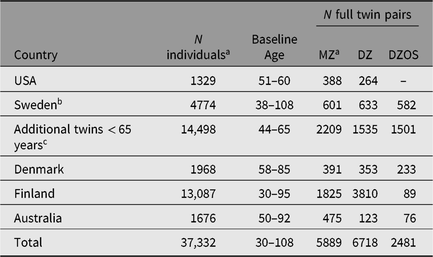
Note: aIncluding imputed MZ cotwins; bFrom SATSA, GENDER, HARMONY, TwinGene > 65 years; cFrom TwinGene < 65 years and SALTY.
Approaches
IGEMS harnesses the full analytic potential of twin designs to address issues of GE interplay as well as risk and protective factors for aging-related outcomes. Methods using MZ within-pair differences allow us to test for the presence of GE without having a specific measured early environment. With a MZ within-pair approach, we established evidence of gene × environment for BMI, depressive symptoms, a physical illness index, several cognitive domains (Reynolds et al., Reference Reynolds, Gatz, Christensen, Kaprio, Korhonen, Kremen and Pedersen2016) and longitudinal grip strength trajectories (Petersen et al., Reference Petersen, Pedersen, Rantanen, Kremen, Johnson, Panizzon and Reynolds2016). Results also suggested that the apolipoprotein E gene (APOE) may act as a ‘variability gene’ for symptoms of depression and spatial reasoning, but not for other cognitive measures or BMI, with greater intrapair differences for non-ε4 carriers. For grip-strength trajectories, a buffering effect for ε2 carriers emerged, with lower sensitivity to environments and better-maintained performance.
Cotwin control methods also provide the opportunity to strengthen causal inferences and test whether associations between early life exposures and late life outcomes are due to confounding by common familial (genetic and/or shared rearing) influences. For example, Mosing et al. (Reference Mosing, Cnattingius, Gatz, Neiderhiser and Pedersen2016) found that of a number of birth characteristics, low birth weight was associated with poorer self-rated health in adulthood when evaluated with a generalized estimating equation adjusting for the twin structure. However, as these associations were attenuated in a cotwin control analyses (first in all pairs, then only in MZ pairs), there is evidence that the association is in part due to familial influences. In subsequent analyses of birth characteristics and cognitive impairment and dementia, we found evidence that low birth weight and small head circumference are risk factors for dementia. Further, head circumference was also significantly associated with age-related cognitive impairment (Mosing et al., Reference Mosing, Lundholm, Cnattingius, Gatz and Pedersen2018). Here, within-pair analyses of identical twins suggested that the observed associations between birth characteristics and cognitive decline are likely not due to underlying familial etiology.
To quantify interplay, we have applied biometric moderation models (Purcell, Reference Purcell2002; van der Sluis et al., Reference van der Sluis, Dolan, Neale and Posthuma2008). We have examined GE interactions in relation to cognitive performance (Pahlen et al., Reference Pahlen, Hamdi, Dahl Aslan, Horwitz, Panizzon, Zavala and McGue2018; Zavala et al., Reference Zavala, Beam, Finch, Gatz, Johnson, Kremen and Reynolds2018), depression (Petkus et al., Reference Petkus, Beam, Johnson, Kaprio, Korhonen, McGue and Gatz2017), subjective health (Franz et al., Reference Franz, Finkel, Panizzon, Spoon, Christensen, Gatz and Pedersen2017) and an index of physical illness (Gatz, Petkus et al., Reference Gatz, Petkus, Franz, Kaprio and Christensen2015). For most phenotypes, unique environmental variance was greater at older ages, presumably reflecting the accumulating importance of individual differences in environmental context with age. However, there was a nonuniform pattern for genetic factors over age, in combination with SES or sex moderation. In SES moderation analyses of cognition, for verbal ability and for perceptual speed (Zavala et al., Reference Zavala, Beam, Finch, Gatz, Johnson, Kremen and Reynolds2018), genetic variance was diminished in those with higher SES, perhaps reflective of a buffering effect on normative aging processes particularly for speed; whereas for short-term/working memory and spatial performance, genetic variance was amplified with higher SES, suggesting stable experiences in enriched (high SES) environments may support genetic variation.
Because IGEMS has genome-wide genetic data, we are able to create PRS scores and incorporate these into our models of GE interplay. In this case, the PRS scores will be entered as a moderator, together with other indicators of the environment, such as SES. The interaction between PRS and SES in regression models predicting health outcomes of interest will inform whether those at high genetic risk for a health outcome are more or less susceptible to, for example, health-promoting benefits of favorable environments.
Country-level SES indicators are available from various online sources. These data provide historical measures of social and economic conditions from the mid-1800s to the early 2000s for each of the five IGEMS countries. Variables include average years of education, educational inequality, gross domestic product per capita (GDP), Gini coefficient of income inequality, public social spending and top 1% income share. As a demonstration of the use of country-level SES indicators, we examined harmonized depressive symptom scores across five countries and a wide range of birth cohorts from 1890 through 1970. We used Top 1% (share of wealth held by top 1% of residents) to index country-level inequality when participants were aged 10 (World Inequality Database, 2017). Controlling for age when the depressive symptom measure was completed, gender, and country-level GDP, adult depressive symptom scores were higher among those exposed to greater inequality as youths. Using a modified twin correlation model, we found greater genetic effects on depressive symptoms with exposure to greater inequality (Gatz et al., Reference Gatz, Finch, Beam and Thomas2018).
Summary
The IGEMS consortium harnesses a combination of twin designs and multiple studies representing different cohorts and contexts. The accomplishments of the consortium demonstrate the feasibility of this type of collaboration in addressing GE interplay with respect to important age-related outcomes.
Acknowledgments
Members of the IGEMS Consortium include Karolinska Institutet: Nancy Pedersen, Miriam Mosing, Malin Ericsson; Jönköping University: Anna Dahl Aslan, Ida Karlsson; University of Southern California: Margaret Gatz, Brian Finch, Kyla Thomas, Christopher Beam, Susan Luczak, Carol Prescott, Em Arpawong, Catalina Zavala, Andrew Petkus; University of Southern Denmark: Kaare Christensen, Marianne Nygaard, Mette Wod; University of Minnesota: Matt McGue, Robert Krueger; University of California, Riverside: Chandra Reynolds, Elizabeth Munoz, Shandell Pahlen, Dianna Phillips; Indiana University Southeast: Deborah Finkel; University of California, San Diego: William Kremen, Carol Franz, Matthew Panizzon, Jeremy Elman, Daniel Gustavson; Edinburgh University: Wendy Johnson; The Pennsylvania State University: Jenae Neiderhiser; Boston University: Michael Lyons; Wayne State University: Keith Whitfield; University of Helsinki: Jaakko Kaprio, Elina Sillanpää, Eero Vuoksimaa; University of New South Wales: Perminder Sachdev, Vibeke Catts, Marie Kondo, Teresa Lee, Karen Mather, Anbu Thalamuthu, Simone Reppermund; QIMR Berghofer: Nicholas G. Martin; American Institutes for Research: Susan Lapham, Kelly Peters; National Academies of Sciences, Engineering and Medicine: David Butler; Duke University: Brenda Plassman. We thank Patricia St. Clair and Ellen Walters for their work on data management.
Financial support
IGEMS is supported by the National Institutes of Health Grants No. R01 AG037985, R56 AG037985, R01 AG059329, R01 AG060470, RF1 AG058068. SATSA was supported by grants R01 AG04563, R01 AG10175, the John D. and Catherine T. MacArthur Foundation Research Network on Successful Aging, the Swedish Council For Working Life and Social Research (FAS) (97:0147:1B, 2009-0795) and Swedish Research Council (825-2007-7460, 825-2009-6141). OCTO-Twin was supported by grant R01 AG08861. Gender was supported by the MacArthur Foundation Research Network on Successful Aging, The Axel and Margaret Ax:son Johnson’s Foundation, The Swedish Council for Social Research and the Swedish Foundation for Health Care Sciences and Allergy Research. TOSS was supported by grant R01 MH54610 from the National Institutes of Health. The Danish Twin Registry is supported by grants from The National Program for Research Infrastructure 2007 from the Danish Agency for Science and Innovation, the Velux Foundation and the US National Institute of Health (P01 AG08761). The Minnesota Twin Study of Adult Development and Aging was supported by NIA grant R01 AG06886. VETSA was supported by National Institute of Health grants NIA R01 AG018384, R01 AG018386, R01 AG022381 and R01 AG022982, and, in part, with resources of the VA San Diego Center of Excellence for Stress and Mental Health. The Cooperative Studies Program of the Office of Research & Development of the United States Department of Veterans Affairs has provided financial support for the development and maintenance of the Vietnam Era Twin (VET) Registry. Data collection and analyses in the Finnish Twin Cohort and Finntwin16 have been supported by ENGAGE — European Network for Genetic and Genomic Epidemiology, FP7-HEALTH-F4-2007, grant agreement number 201413, National Institute of Alcohol Abuse and Alcoholism (grants AA-12502, AA-00145 and AA-09203), the Academy of Finland Center of Excellence in Complex Disease Genetics (grant numbers: 213506, 129680) and the Academy of Finland (grants 100499, 205585, 118555, 141054, 265240, 263278, 264146, 308248 and 312073). This MIDUS study was supported by the John D. and Catherine T. MacArthur Foundation Research Network on Successful Midlife Development and by National Institute on Aging Grant AG20166. Funding for the Australian Over-50’s twin study was supported by Mr. George Landers of Chania, Crete. We acknowledge the contribution of the OATS research team (https://cheba.unsw.edu.au/project/older-australian-twins-study) to this study. The OATS study has been funded by a National Health & Medical Research Council (NHMRC) and Australian Research Council (ARC) Strategic Award Grant of the Ageing Well, Ageing Productively Program (ID No. 401162) and NHMRC Project Grants (ID 1045325 and 1085606). OATS participant recruitment was facilitated through Twins Research Australia, a national resource in part supported by a Centre for Research Excellence Grant (ID: 1079102), from the National Health and Medical Research Council. We thank the participants for their time and generosity in contributing to this research. The Carolina African American Twin Study of Aging (CAATSA) was funded by NIA grant R01 AG13662. The Project Talent Twin Study has been supported by National Institute of Health grants R01 AG043656 and R01 AG056163, and development funds from American Institutes of Research. Funding for archiving the NAS-NRC Twin Registry data was provided by NIH Grant No. R21 AG039572. The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the NIA/NIH, or the VA.
Author note
IGEMS data are not publicly available given the variety of data agreements and regulations governing the different studies and countries. However, many of the individual studies participating in IGEMS do have ways to access their data, and many of the datasets may be accessed through National Archive of Computerized Data on Aging (NACDA).






