Several long-term studies of course and outcome of schizophrenia report that about 25% of incident and prevalent cases show good clinical and social recovery (Reference BleulerBleuler, 1978; Reference CiompiCiompi, 1980; Reference Shepherd, Watt and FaloonShepherd et al, 1989; Reference Jablensky, Sartorius and ErnbergJablensky et al, 1992; Reference Hegarty, Baldessarini and TohenHegarty et al, 1994; Reference Wiersma, Nienhuis and SloffWiersma et al, 1998; Reference Harrison, Hopper and CraigHarrison et al, 2001; Reference WarnerWarner, 2004).
The same studies have found about 20–25% of the cases experience poor outcome (e.g. chronic psychosis, a deteriorating course or suicide).
The course of early illness has been found to be a strong predictor of the course's long-term pattern (Reference Wiersma, Nienhuis and SloffWiersma et al, 1998; Reference Harrison, Hopper and CraigHarrison et al, 2001), and the concept of a ‘critical period’ has been developed (Reference Birchwood, Todd and JacksonBirchwood et al, 1998). As early illness course is an important factor for the long-term course, intervention during this critical period is considered important (Reference McGlashan and JohannessenMcGlashan & Johannessen, 1996; Reference Birchwood, Todd and JacksonBirchwood et al, 1998). The question is whether intervention can alter the early illness course, and lead to a lower morbidity plateau and a less disabling type of course. Evidence from trials with chronic and mixed populations suggests that there is a positive effect of psychosocial interventions on clinical and social outcomes, but no large randomised clinical trial has been conducted for first-episode psychosis.
This paper focuses on prevention of poor early outcome. We hypothesised that integrated treatment compared with standard treatment would reduce the proportion of patients with a poor clinical and social outcome after 1 year.
METHOD
Sample
The sample comprised patients who fulfilled the following inclusion criteria: age 18–45 years; clinical diagnoses of schizophrenia, schizotypal disorder, persistent delusional disorder, acute and transient psychotic disorder, schizoaffective disorder, induced delusional disorder, or unspecified non-organic psychosis according to ICD–10 research criteria, based on Schedules for Clinical Assessment in Neuropsychiatry, SCAN version 2.0 and 2.1 (World Health Organization, 1993, 1998); no antipsychotic medication exceeding 12 weeks of continuous medication; absence of mental retardation and organic mental disorder; no psychotic condition solely due to acute intoxication or a withdrawal state; and written informed consent.
A total of 547 patients were included consecutively from January 1998 to December 2000. All patients were randomly allocated to integrated psychiatric treatment or standard treatment. The Copenhagen Trial Unit carried out randomisation using computer-generated random lists of patients, whereas in Aarhus, after the initial assessment was finished, the researchers contacted a secretary, who then drew a lot from among five red and five white lots from a black box.
Assessments
At inclusion, all patients were comprehensively assessed using standardised rating instruments, including SCAN 2.0 and 2.1 (World Health Organization, 1993, 1998), the Scale for Assessment of Positive Symptoms (SAPS; Reference AndreasenAndreasen, 1984), the Scale for Assessment of Negative Symptoms (SANS; Reference AndreasenAndreasen, 1983) and Global Assessment of Functioning (GAF; American Psychiatric Association, 1994). SANS and SAPS are six-point scales with the categories none, questionable, mild, moderate, marked and severe. The global scale scores can be summed up in three dimensions: positive, negative and disorganised dimension (Andreassen et al, 1995). Duration of untreated psychosis was assessed with the Interview for Retrospective Assessment of Onset of Schizophrenia (IRAOS; Reference Häfner, Riecher-Rossler and HambrechtHäfner et al, 1992). In addition, information was collected concerning socio-demographic factors (e.g. education, accommodation and employment status). The same assessment instruments were used at 1-year follow-up, supplemented with copies of medical records from the preceding year, whenever available.
Researchers with no responsibility for treatment carried out all follow-up interviews. Treatment allocation was not concealed from the researchers.
Interrater reliability
All researchers were trained how to implement the SCAN interview at the World Health Organization collaborating centre and trained in SAPS with live interviews. The intraclass correlation coefficient (ICC) for P.J. and M.A. measuring SAPS global scores was 0.63 (number of cases=8). L.P., J.O., G.K., T.C. and A.T. carried out 14 SANS and 12 SAPS reliability interviews together. The ICC was 0.54 for the negative dimension and 0.88 for the positive dimension.
Treatment groups
Integrated treatment
Three teams were established and trained, two in Copenhagen and one in Aarhus. Each patient was offered integrated treatment for a period of 2 years. The integrated treatment is described below.
An enriched Assertive Community Treatment model inspired by Stein & Test (Reference Stein and Test1980) was used. A multidisciplinary team including the following: psychiatrist, psychologist, psychiatric nurse, occupational therapist and social worker, provided the integrated treatment. The case-load was 1:10. A primary team member was designated for each patient and was then responsible for maintaining contact and coordinating the treatment within the team and across different treatment and support facilities. The patients were visited in their homes or other locations in their community, or they were seen at the office according to the patients’ preference. When hospitalised, the patient was visited weekly at the hospital. During in-patient treatment, treatment responsibility was transferred to the hospital. The office hours of the OPUS team were Monday to Friday from 08.00 h to 17.00 h. All team workers had a cell telephone with an answering function, so that patients could leave a message outside office hours and be sure that the team would respond the next morning. A crisis plan was developed for each patient. The patients were encouraged to take responsibility for their own affairs as soon as possible during the process of recovery. If the patient was reluctant to continue treatment, the team tried to motivate the patient and stayed in contact.
The team offered medication according to the low-dose recommendations for patients with first-episode psychosis using atypical antipsychotic drugs as first choice. Careful attention was paid to the patient's adherence to treatment, using psychoeducational methods.
Patients with impaired social skills assessed by the Disability Assessment Schedule (DAS; Reference Holmes, Shah and WingHolmes et al, 1982) were offered social skills training with focus on medication, coping with symptoms, conversation, problem-solving and conflict-solving skills in a group with a maximum of six patients and two therapists. The patients who were unable to work in a group were offered individual training. Patients who did not need social skills training received individual psychoeducation.
Psychoeducational family treatment modelled on McFarlane's manual of multiple-family groups was offered to patients and their family members or friends (Reference McFarlane, Lukens and LinkMcFarlane et al, 1995).
Standard treatment
The standard treatment consisted of the standard mental health service routines in Copenhagen and Aarhus. Case-loads varied between 1:20 and 1:30. Contacts were usually made in the local treatment centre. The antipsychotic medication was based on the same principles as the integrated treatment.
Data analysis
Pearson χ2 was used as appropriate to test for statistically significant differences between treatment groups at baseline. Level of significance was 0.05. With logistical regression analysis, the odds ratio (OR) for treatment effect was calculated with the baseline value of the scale included as a covariate. All statistical analysis was performed using the Statistical Package for the Social Sciences, version 11.0 for Windows. Outcome measures were analysed according to intention-to-treat principles.
Using the formula described by Pocock (Reference Pocock1996), we found that 262 patients in each treatment condition were necessary in order to detect a difference at 1-year follow-up between 10 and 20% in less frequent outcome measures, with a significance level of 0.05 and 90% power.
Representation
The number of patients included in the project corresponded to 90% in Aarhus and 63% in Copenhagen of all patients registered as having a first-episode psychosis in the schizophrenia spectrum in the same catchment areas. In both centres, patients included in the trial were significantly younger compared with those in the register, and significantly more were diagnosed with schizophrenia.
Outcome measures
We defined ‘poor outcome’ for all the variables included in the analysis: any global score of SAPS or SANS over 3; substance misuse or dependence present; GAF–S (Symptom) and GAF–D (Disability) scores below 30; being homeless or living in sheltered or supervised accommodation; no work and not in education during the preceding year. Finally, death was included as a poor outcome.
In addition, we constructed a global measure: ‘any poor outcome’: any psychotic or negative global scores of SANS or SAPS ‘marked’ or ‘severe’, or substance misuse or dependence present, or GAF scores under 30, or homeless or living in sheltered or supervised accommodation, or no work and not in education. This was based on the assumption that poor outcome in any of the variables is considered disabling.
RESULTS
Main baseline characteristics of the cohort are shown in Table 1. No statistical differences were found between treatment groups at baseline in either clinical or socio-demographic characteristics.
Table I Clinical, demographic and socio-economic baseline characteristics of 547 patients with first-episode psychosis included in the OPUS trial by entry group1
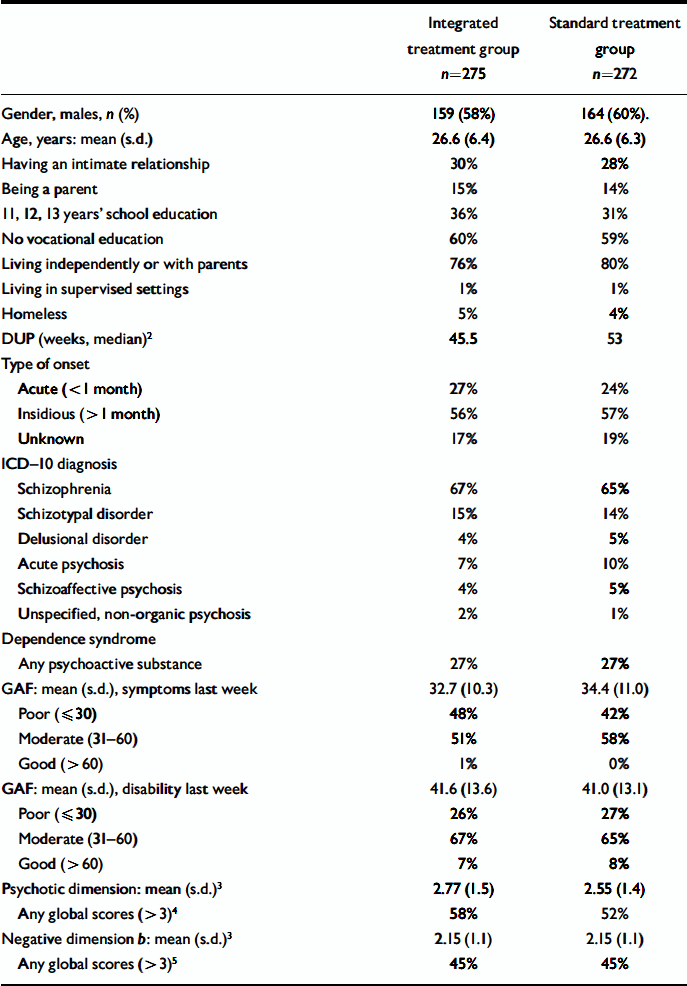
| Integrated treatment group n=275 | Standard treatment group n=272 | |
|---|---|---|
| Gender, males, n (%) | 159 (58%) | 164 (60%). |
| Age, years: mean (s.d.) | 26.6 (6.4) | 26.6 (6.3) |
| Having an intimate relationship | 30% | 28% |
| Being a parent | 15% | 14% |
| 11, 12, 13 years’ school education | 36% | 31% |
| No vocational education | 60% | 59% |
| Living independently or with parents | 76% | 80% |
| Living in supervised settings | 1% | 1% |
| Homeless | 5% | 4% |
| DUP (weeks, median)2 | 45.5 | 53 |
| Type of onset | ||
| Acute (<1 month) | 27% | 24% |
| Insidious (>1 month) | 56% | 57% |
| Unknown | 17% | 19% |
| ICD—10 diagnosis | ||
| Schizophrenia | 67% | 65% |
| Schizotypal disorder | 15% | 14% |
| Delusional disorder | 4% | 5% |
| Acute psychosis | 7% | 10% |
| Schizoaffective psychosis | 4% | 5% |
| Unspecified, non-organic psychosis | 2% | 1% |
| Dependence syndrome | ||
| Any psychoactive substance | 27% | 27% |
| GAF: mean (s.d.), symptoms last week | 32.7 (10.3) | 34.4 (11.0) |
| Poor (≤ 30) | 48% | 42% |
| Moderate (31-60) | 51% | 58% |
| Good (> 60) | 1% | 0% |
| GAF: mean (s.d.), disability last week | 41.6 (13.6) | 41.0 (13.1) |
| Poor (≤ 30) | 26% | 27% |
| Moderate (31-60) | 67% | 65% |
| Good (> 60) | 7% | 8% |
| Psychotic dimension: mean (s.d.)3 | 2.77 (1.5) | 2.55 (1.4) |
| Any global scores (> 3)4 | 58% | 52% |
| Negative dimension b: mean (s.d.)3 | 2.15 (1.1) | 2.15 (1.1) |
| Any global scores (> 3)5 | 45% | 45% |
Figure 1 shows the flow of patients during the first year of follow-up. A total of 419 patients were interviewed at 1-year follow-up. More patients in the integrated treatment group (83% v. 71% in the standard treatment group) participated in the follow-up interview. Analysis has shown that no clinical or basic socio-demographic baseline characteristics were associated with participation after 1 year except that more interviewed patients in both groups had 11–13 years of education compared with those not interviewed.
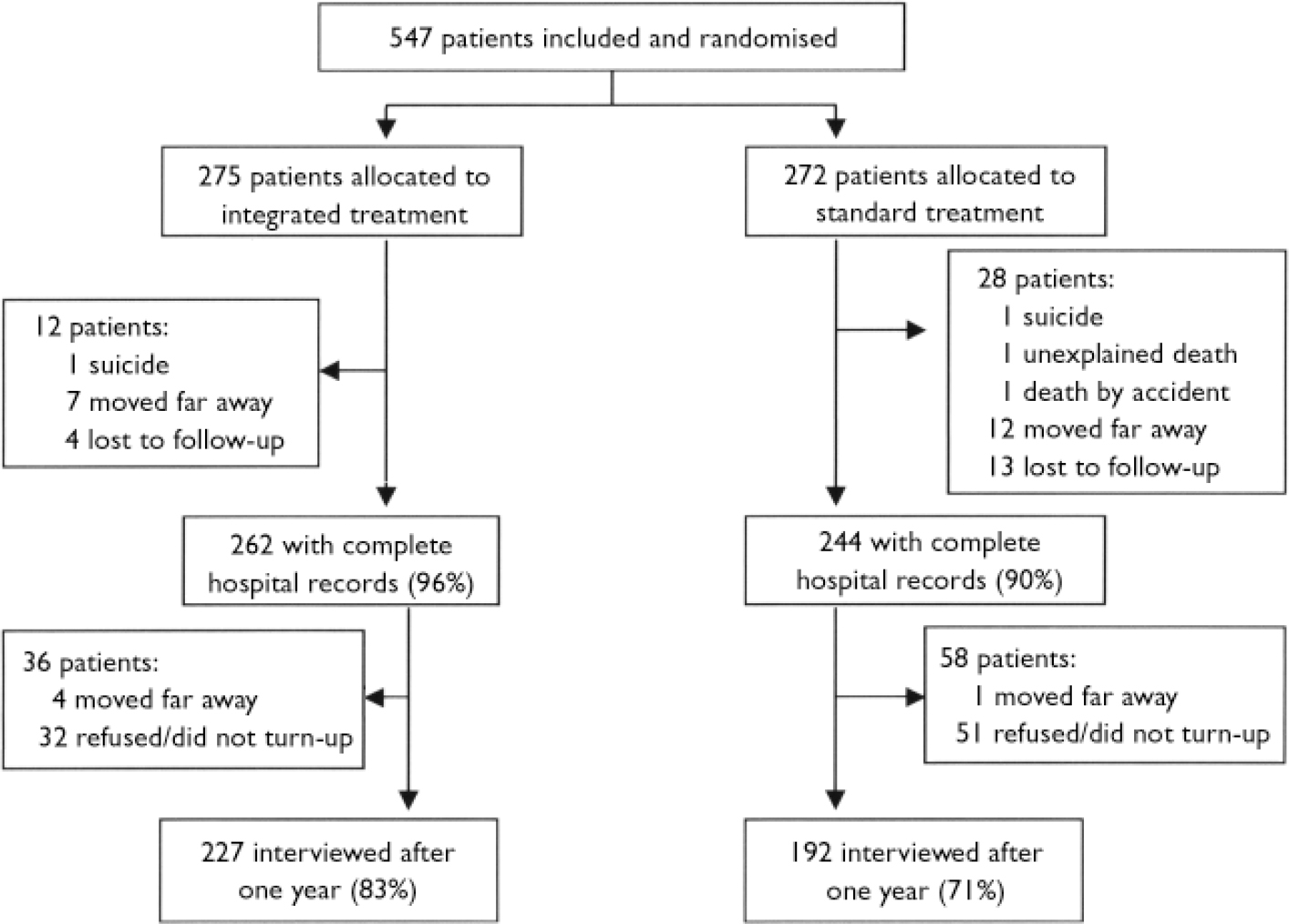
Fig. 1 Flowchart of patients in the OPUS trial during the first year of follow-up.
Table 2 summarises outcome variables after 1 year regarding poor outcome. The number of patients varies for the different variables because all relevant information concerning socio-demographic and psychopathological status at 1 year was recorded from medical records, if possible, for patients who were not re-interviewed. ‘Any poor outcome’ is based on patients attending the 1-year interview. There were significant differences favouring integrated treatment in the proportion of patients with high psychotic global scores (OR=0.35, 95% CI 0.2–0.6, P=0.001) and high negatives global scores (OR=0.49, 95% CI 0.3–0.8, P=0.002). Significantly fewer patients receiving integrated treatment had GAF–S scores below 30 (OR=0.55 95% CI 0.3–1.0, P=0.04), and fewer were homeless or living in sheltered accommodation (OR=0.53, 95% CI 0.3–0.9, P=0.02) or had no work (OR=0.31, 95% CI 0.2–0.5, P=0.01). Significantly fewer patients presented comorbidity for drug or alcohol misuse or dependence in the intervention group at 1-year follow-up (OR=0.54, 95% CI 0.3–0.9, P=0.03). Three patients in the standard group and one patient in the integrated treatment had died.
Table 2 Symptoms and social disability at 1-year follow-up: integrated treatment (IT) and standard treatment (ST)
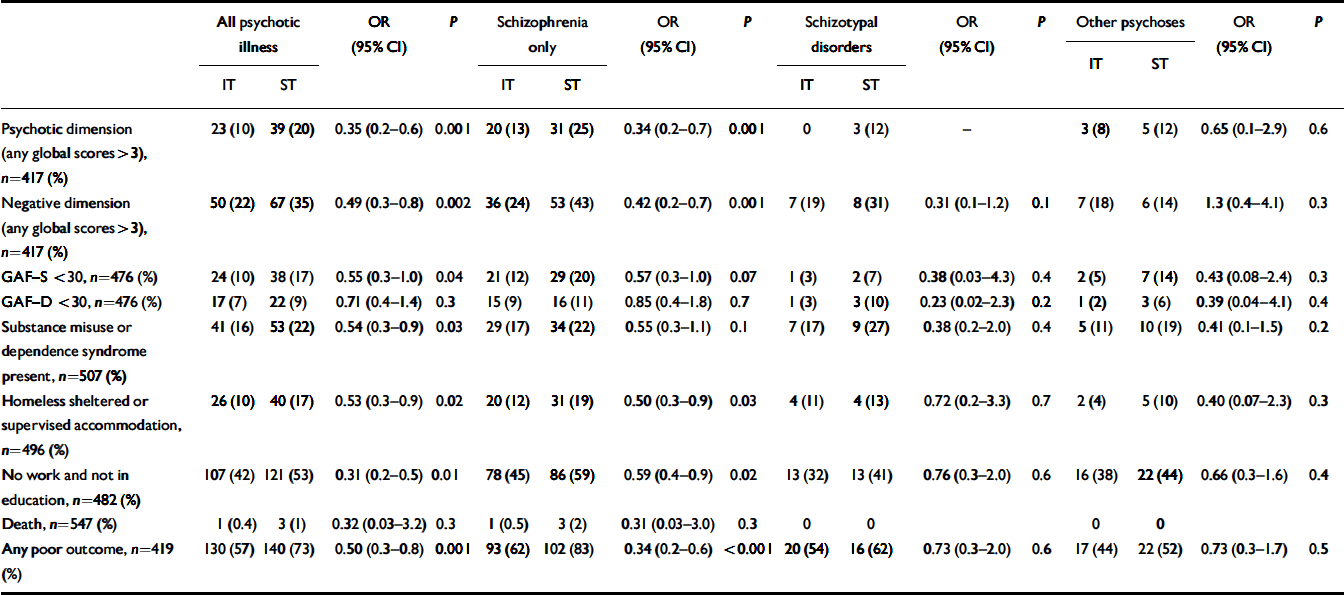
| All psychotic illness | OR (95% Cl) | P | Schizophrenia only | OR (95% Cl) | P | Schizotypal disorders | OR (95% Cl) | P | Other psychoses | OR (95% Cl) | P | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IT | ST | IT | ST | IT | ST | IT | ST | |||||||||
| Psychotic dimension (any global scores>3), n=417 (%) | 23 (10) | 39 (20) | 0.35 (0.2-0.6) | 0.001 | 20 (13) | 31 (25) | 0.34 (0.2-0.7) | 0.001 | 0 | 3 (12) | — | 3 (8) | 5 (12) | 0.65 (0.1-2.9) | 0.6 | |
| Negative dimension (any global scores>3), n=417 (%) | 50 (22) | 67 (35) | 0.49 (0.3-0.8) | 0.002 | 36 (24) | 53 (43) | 0.42 (0.2-0.7) | 0.001 | 7 (19) | 8 (31) | 0.31 (0.1-1.2) | 0.1 | 7 (18) | 6 (14) | 1.3 (0.4-4.1) | 0.3 |
| GAF—S <30, n=476 (%) | 24 (10) | 38 (17) | 0.55 (0.3-1.0) | 0.04 | 21 (12) | 29 (20) | 0.57 (0.3-1.0) | 0.07 | 1 (3) | 2 (7) | 0.38 (0.03-4.3) | 0.4 | 2 (5) | 7 (14) | 0.43 (0.08-2.4) | 0.3 |
| GAF—D <30, n=476 (%) | 17 (7) | 22 (9) | 0.71 (0.4-1.4) | 0.3 | 15 (9) | 16 (11) | 0.85 (0.4-1.8) | 0.7 | 1 (3) | 3 (10) | 0.23 (0.02-2.3) | 0.2 | 1 (2) | 3 (6) | 0.39 (0.04-4.1) | 0.4 |
| Substance misuse or dependence syndrome present, n=507 (%) | 41 (16) | 53 (22) | 0.54 (0.3-0.9) | 0.03 | 29 (17) | 34 (22) | 0.55 (0.3-1.1) | 0.1 | 7 (17) | 9 (27) | 0.38 (0.2-2.0) | 0.4 | 5 (11) | 10 (19) | 0.41 (0.1-1.5) | 0.2 |
| Homeless sheltered or supervised accommodation, n=496 (%) | 26 (10) | 40 (17) | 0.53 (0.3-0.9) | 0.02 | 20 (12) | 31 (19) | 0.50 (0.3-0.9) | 0.03 | 4 (11) | 4 (13) | 0.72 (0.2-3.3) | 0.7 | 2 (4) | 5 (10) | 0.40 (0.07-2.3) | 0.3 |
| No work and not in education, n=482 (%) | 107 (42) | 121 (53) | 0.31 (0.2-0.5) | 0.01 | 78 (45) | 86 (59) | 0.59 (0.4-0.9) | 0.02 | 13 (32) | 13 (41) | 0.76 (0.3-2.0) | 0.6 | 16 (38) | 22 (44) | 0.66 (0.3-1.6) | 0.4 |
| Death, n=547 (%) | 1 (0.4) | 3 (1) | 0.32 (0.03-3.2) | 0.3 | 1 (0.5) | 3 (2) | 0.31 (0.03-3.0) | 0.3 | 0 | 0 | 0 | 0 | ||||
| Any poor outcome, n=419 (%) | 130 (57) | 140 (73) | 0.50 (0.3-0.8) | 0.001 | 93 (62) | 102 (83) | 0.34 (0.2-0.6) | <0.001 | 20 (54) | 16 (62) | 0.73 (0.3-2.0) | 0.6 | 17 (44) | 22 (52) | 0.73 (0.3-1.7) | 0.5 |
In general, 64% of all the patients had ‘any poor outcome’ after 1 year. There was a significant beneficial effect of integrated treatment v. standard treatment on ‘any poor outcome’ (OR=0.50, 95% CI 0.3–0.8, P=0.001).
Identical analyses were carried out in diagnostic subgroups. Positive effects of integrated treatment were found among patients with schizophrenia on psychotic global scores (OR=0.34, 95% CI 0.2–0.7, P=0.001), on negative global scores (OR=0.42, 95% CI 0.2–0.7, P=0.001), on being homeless or living in a supervised environment (OR=0.50, 95% CI 0.3–0.9, P=0.03), and on work and education (OR=0.59, 95% CI 0.4–0.9, P=0.02).
There was a significant effect of integrated treatment compared with standard treatment among patients with schizophrenia on ‘any poor outcome’ (OR=0.34, 95% CI 0.2–0.6, P<0.001).
No significant differences were found in other diagnostic groups.
DISCUSSION
To our knowledge, the present study is the first randomised controlled trial of integrated treatment v. standard treatment for first-episode psychosis. The findings suggest that integrated treatment improved the early illness course, and that this was most marked in patients with schizophrenia. High levels of psychotic and negative symptom scores were less prevalent in patients receiving integrated treatment after 1 year. Better adherence to antipsychotic medication in the integrated treatment group could explain these results. However, no significant differences were found between treatment groups in antipsychotic medication (Reference Thorup, Petersen and JeppesenThorup et al, 2005). Social outcome data were also better for integrated treatment; and for the global measure ‘any poor outcome’, the significantly better effect of integrated treatment on poor outcome is replicated. Thus, the findings supported the hypothesis that patients on standard treatment had poorer outcome.
There are some limitations to the study. The randomisation procedure used in Aarhus is not optimal because it does not offer the same protection against unmasking as the computerised model used in Copenhagen. However, it does not seem to affect the results.
Blinding of the assessors to treatment allocation would have been optimal, but this was not judged to be possible in this kind of trial. There is a potential risk of bias due to skewed attrition. We have found a higher proportion living independently at 1 year among participants compared with non-participants in the 1-year follow-up interview, and the finding that patients with higher education were more likely to attend follow-up interviews might indicate that the group not participating had a worse outcome. This could bias the analyses toward the integrated treatment. This is especially important when focus is on poor outcome, as it might be expected that patients with the poorest outcome would be the ones to drop out of the trial. Short-term course is found to be an important predictor of long-term outcome. The results of the present study suggest that integrated treatment at this early stage of the illness course might have an effect in preventing patients from experiencing the poorest outcome, and thus perhaps affect the long-term course of the illness. This emphasises the importance of intervention in the early period.
Clinical Implications and Limitations
CLINICAL IMPLICATIONS
-
▪ Integrated treatment improved the early illness course.
-
▪ High levels of psychotic and negative symptoms were less prevalent in patients receiving integrated treatment after 1 year.
-
▪ Patients with substance misuse benefited from integrated treatment.
LIMITATIONS
-
▪ Treatment allocation was not concealed from the assessors.
-
▪ There was a suboptimal randomisation procedure in Aarhus.
-
▪ There was a potential risk of bias due to skewed attrition.
Acknowledgements
Funded by the Danish Ministry of Health (jr.nr. 96-0770-71), the Danish Ministry of Social Affairs, the University of Copenhagen, the Copenhagen Hospital Corporation, the Danish Medical Research Council (jr.nr. 9601612 and 9900734), and Slagtermester Wørzners Fond. The staff at Copenhagen Trial Unit planned the randomisation procedure in Copenhagen. Professor Philip Hougaard supervised the statistical analyses.






eLetters
No eLetters have been published for this article.