Meta-analysis of trials of cognitive-behavioural therapy (CBT) indicates successful treatment of persistent symptoms in chronic schizophrenia (Reference Gould, Mueser and BoltonGould et al, 2001; Reference Pilling, Bebbington and KuipersPilling et al, 2002). In acutely ill patients, Drury et al (Reference Drury, Birchwood and Cochrane1996a ,Reference Drury, Birchwood and Cochrane b ) reported that CBT significantly reduced recovery time by 25-50% and decreased the proportion of patients with residual symptoms to 5% in the treatment group compared with 56% in the control group. Differences between the CBT and control patient groups were not apparent 5 years later (Reference Drury, Birchwood and CochraneDrury et al, 2000). Haddock et al (Reference Haddock, Tarrier and Morrison1999a ) found that CBT provided during an acute phase of illness resulted in non-significant reductions in rates of relapse and re-hospitalisation over 2 years compared with supportive counselling.
Positive symptoms in patients with first-episode schizophrenia respond well to treatment initially, but relapses are frequent (Robinson et al, Reference Robinson, Woerner and Alvir1999a ,Reference Robinson, Woerner and Alvir b ). The Study of Cognitive Reality Alignment Therapy in Early Schizophrenia (SoCRATES) was designed to test whether CBT speeded recovery and subsequently protected against the persistence of symptoms and relapse after a first or early acute onset of the disorder (Reference Lewis, Tarrier and HaddockLewis et al, 2002). Cognitive-behavioural therapy provided directly after admission in addition to treatment as usual was compared with supportive counselling plus treatment as usual, and treatment as usual alone. The study was analysed in two phases. The first investigated whether CBT would significantly speed recovery compared with the two control groups; significant decreases in symptoms were evident over the first 7 weeks with a significant benefit for CBT over treatment as usual at 5 weeks (Reference Lewis, Tarrier and HaddockLewis et al, 2002). The second, follow-up phase of the study is reported here.
METHOD
Study design
This was a multicentre, prospective, rater-masked, randomised controlled trial with an 18-month follow-up period. Eligible patients were randomly allocated to one of three treatments: CBT and treatment as usual; supportive counselling and treatment as usual; and treatment as usual alone. The report of phase 1 of the study presented the recovery data over the first 70 days (Reference Lewis, Tarrier and HaddockLewis et al, 2002). In phase 2, patients were followed up and assessed through psychiatric interview and examination of hospital records and case notes, 18 months after randomisation. It was predicted that the group who received CBT would show decreased levels of symptoms at 18 months, and reduced rates of relapse, readmission to hospital, shorter duration of admission and longer survival times before relapse, compared with the two other groups.
Participants, recruitment and assignment
Participants were recruited over 26 months from the 11 mental health units serving three geographically defined catchment areas in England: Manchester/Salford, Liverpool and North Nottinghamshire; these areas had a combined population of 2.3 million. Inclusion criteria were:
-
(a) either first or second admission (within 2 years of a first admission) to an in-patient or day-patient unit for treatment of psychosis;
-
(b) DSM-IV criteria for schizophrenia, schizophreniform disorder, schizoaffective disorder, delusional disorder or psychosis not otherwise specified (American Psychiatric Association, 1994);
-
(c) positive psychotic symptoms for 4 weeks or more;
-
(d) score of 4 or more (moderate to extreme) on the Positive and Negative Syndrome Scale (PANSS; Reference Kay, Opler and LindenmayerKay et al, 1987) target item either for delusions (P1) or hallucinations (P3);
-
(e) neither substance misuse nor organic disorder judged to be the major cause of psychotic symptoms.
Patients legally detained in hospital were eligible.
Frequent contact by telephone and in person was maintained with the in-patient and day-patient facilities to identify new admissions. Potentially eligible patients were screened within 14 working days of hospital admission by a research psychiatrist. Following the patient's written consent, baseline assessments were made and demographic data recorded. Diagnoses at baseline were confirmed by raters at the 12-week and 18-month follow-up assessments. Independent, concealed randomisation of individuals with minimisation was performed at each centre by a trial administrator. Stratification was undertaken with the following variables: first or second admission, in-patient or day-patient admission, male or female, with the first-episode cases further stratified for duration of symptoms of more or less than 6 months.
Assessment and measures
Two ratings of symptoms at baseline and follow-up were used as primary outcome measures: the PANSS total and sub-scale scores, and the Psychotic Symptom Rating Scales (PSYRATS; Reference Haddock, McCarron and TarrierHaddock et al, 1999b ). The PSYRATS were developed to measure dimensions of delusional beliefs (Delusions Scale) and auditory hallucinations (Auditory Hallucination Scale) and have been shown to have good reliability and validity with sensitivity to change (Reference Haddock, McCarron and TarrierHaddock et al, 1999b ). Good reliability between the three psychiatric raters was established using videotaped interviews; intraclass correlation coefficients (ICC) for PANSS scores were 0.85-0.88 for positive symptoms, 0.65-0.73 for negative symptoms, 0.70-0.83 for general symptoms and 0.72-0.83 for total scores.
Patients were assessed 18 months after randomisation. After the follow-up period had elapsed, the hospital charts and case notes of all study participants were obtained and examined for evidence of relapse, using a method devised in a previous study which was found to have satisfactory reliability and validity (Reference Barrowclough, Tarrier and LewisBarrowclough et al, 1999). Relapses were defined as an exacerbation of psychotic symptoms lasting longer than 1 week and leading to a change in patient management, as recorded by hospital charts (increases in medication, admission, shift to more supported accommodation, or more community nurse or social worker input in response to clinical deterioration). Interrater reliability for whether a relapse had occurred (κ=0.72) and time to relapse (ICC=0.69) were acceptable. Readmission data were obtained from hospital information systems.
Medication
Antipsychotic medication was assessed in three ways. Total dosage in chlorpromazine equivalents (mg per day) was recorded for both typical and atypical drugs. Compliance was measured using the four-point scale devised by Barrowclough et al (Reference Barrowclough, Tarrier and Lewis1999), which classifies compliance from ‘very poor’ to ‘very good’; this scale has good interrater reliability (ρ=0.76-0.95). Scores on the Drugs Attitude Inventory (DAI; Reference Hogan, Award and EastwoodHogan et al, 1983) were recorded at 3 and 18 months. Scores on the DAI have been shown to be strongly associated with subsequent medication compliance.
Therapeutic interventions
The interventions were carried out independently of the assessors, who were kept unaware of treatment allocation. Direct family interventions were not undertaken. Procedures to standardise routine clinical care, including drug treatment, were not used.
The interventions were devised by the principal investigators and were based on past treatment studies (Reference Kingdon and TurkingtonKingdon & Turkington, 1991; Reference Tarrier, Yusupoff and KinneyTarrier et al, 1998; Reference Haddock, Tarrier and MorrisonHaddock et al, 1999a ). The feasibility of the planned intervention for patients in an early, acute stage of schizophrenia was tested and confirmed in a pilot study of a separate sample of 35 patients (Reference Haddock, Tarrier and MorrisonHaddock et al, 1999a ). The CBT was manual-based and conducted by one of five therapists trained in CBT for psychosis, supervised by experienced cognitive-behavioural therapists. The research therapists were each based at one of the three centres and worked exclusively in that centre. Two therapists were based in Liverpool, two in Manchester/Salford and one in North Nottinghamshire. All therapists were eligible for accreditation as cognitive-behavioural therapists by the British Association for Behavioural and Cognitive Psychotherapies. Three were clinical psychologists and two nurse therapists. At the commencement of the study the therapists were trained in the interventions and throughout the study they received separate expert and peer supervision on a regular basis to maintain treatment quality.
The delivery was designed to provide 15-20 hours of treatment within 5 weeks of admission, plus ‘booster’ sessions at 2 weeks, and 1, 2 and 3 months after the initial treatment. Details of the treatment are given by Haddock et al (Reference Haddock, Tarrier and Morrison1999a ). Interventions particularly addressed delusions and hallucinations, generating alternative hypotheses for abnormal beliefs and hallucinations, identifying precipitating and alleviating factors and reducing associated distress, and teaching coping strategies.
Supportive counselling was used, as in a previous study (Reference Tarrier, Yusupoff and KinneyTarrier et al, 1998), as a comparison intervention to control for non-specific elements of therapist exposure. It was therefore delivered in the same 5-week format with four booster sessions, with the aim of providing the same amount of therapist contact time as in the CBT arm. The supportive counselling was also manual-based and was supervised by an experienced counsellor. The same five research therapists administered both interventions, according to randomisation.
Interventions were started within 3 days of randomisation. Following their discharge from hospital care, patients continued their treatment in hospital settings, in family practitioner surgeries and in their own homes. All treatment sessions, for both therapeutic interventions, were audiotaped where consent was given. A random selection of 50 of these tapes were viewed by two independent raters (masked) who were asked to classify them as CBT or supportive counselling sessions, and to rate the quality of therapy on the Cognitive Therapy Scale for Psychosis (CTS-Psy; Reference Haddock, Devane and BradshawHaddock et al, 2001).
Treatment exposure and fidelity
In terms of exposure to treatment, the mean number of therapy sessions was similar in the CBT group (mean 16.1 sessions, 95% CI 15.2-17.1) and the supportive counselling group (mean 15.7 sessions, 95% CI 14.7-16.7). The CBT group did, however, receive more total therapy time (mean 8.6 h, 95% CI 7.6-9.63) than the supportive counselling group (mean 7.1 h, 95% CI 6.3-7.9). Four patients allocated to CBT and six allocated to supportive counselling did not attend any treatment session. For the rating of treatment fidelity, agreement between the two independent masked raters was good (CTS-Psy, ICC=0.93). Quality of CBT was assessed as good, with the ‘cognitive-behavioural techniques’ sub-scale score of the CTS-Psy confirming the specificity of cognitive-behavioural techniques to the CBT group (mean sub-scale score 20.7; 95% CI 18.2-23.3) and their absence in the supportive counselling group (mean sub-scale score 2.7, 95% CI 1.9-3.6). Raters correctly classified 49 of the 50 tapes in terms of the appropriate therapy.
Concealment of allocation
Randomisation was done by the centre administrator independently of both the assessors and the therapists. At each centre the administrator entered the patient's details into the minimisation programme for that centre, independently of the therapists, and then informed the therapist of the allocation. The administrator was aware only of the code number, name and date of birth and stratification criteria for each patient. Extensive steps were taken to maintain the masking of raters, by methods successfully used in a previous study (Reference Tarrier, Yusupoff and KinneyTarrier et al, 1998). Therapists and assessors were not permitted to communicate details about individual patients to each other; separate offices and administrative procedures were provided for assessors and therapists; data storage and management were kept separate and secure; clinical staff were instructed not to divulge details of therapist contacts to the assessors; and the patients were instructed not to discuss details of their treatment with assessors. Assessors were asked to record any loss of masking to treatment allocation; this occurred on four occasions only. At the end of the study the assessors were asked to guess the treatment allocation.
Analysis
Power analysis indicated that group sizes of 118 would detect a reduction of relapse rates from 40% to 20% at 18 months using two-tailed tests with conventional levels of statistical significance and 90% power. All analyses were carried out using Stata version 6.0 (StataCorp, 2001). Analysis of outcomes was performed on an intention-to-treat basis (i.e. as randomised) with all available participant data in the analysis. Estimates of treatment effects on the symptom scores were obtained through the use of a regression (analysis of covariance, ANCOVA) model after allowing for relevant baseline score, time of assessment, centre, patient gender, prior hospitalisation, first or second episode, and duration of untreated psychosis as covariates. Treatment contrasts were constructed, first to test whether the outcome for CBT differed from that for supportive counselling, and second to compare both treatments together with treatment as usual (if the first test were to show different outcomes for CBT and supportive counselling, then the second test would be replaced by separate comparisons of CBT and supportive counselling with treatment as usual). The sensitivity of the results to patterns of attrition (missing follow-up data) was examined using inverse probability weighting as described by Heyting et al (Reference Heyting, Tolboom and Essers1992) and Everitt & Pickles (Reference Everitt and Pickles1999). A supplementary series of analyses then tested for centre-treatment-group interactions, and where they were statistically significant, this was followed by estimation of the treatment effects for each of the three centres separately. Where the data appeared highly skewed (the auditory hallucination scores in particular), the robustness of the findings was checked using multiple logistic regression (the dependent variable being the presence or absence of hallucinations). Analysis of relapse and readmission to hospital was carried out using survival models.
RESULTS
Participant recruitment and follow-up
Initial patient identification resulted in 433 patients being screened at interview. Of these, 370 met the eligibility criteria for study entry and 315 gave written consent to participate in the study, after 10 were judged incapable of giving informed consent and a further 45 declined (Fig. 1). Patients were recruited at a median of 6 days after hospital admission and all were randomised within 3 days of consent. There was no significant difference between consenting and non-consenting patients in age, gender, ethnicity, first v. second admission and day-patient v. in-patient treatment (Reference Lewis, Tarrier and HaddockLewis et al, 2002). One person was randomised twice by error (once at first and once at second admission); the second randomisation was disregarded in the analysis. Six patients were excluded from the study and the analysis after randomisation, when it became clear on assessment at week 1 that they met diagnostic exclusion criteria (3 organic psychosis, 2 bipolar disorder, 1 factitious psychosis).

Fig. 1 Study flow diagram.
Of the total sample, 38% were detained under the Mental Health Act 1983 during the 70-day period, reflecting the fact that these people were severely ill. Baseline characteristics of the three treatment groups are given in Table 1.
Table 1 Baseline characteristics of the participants in the three treatment groups
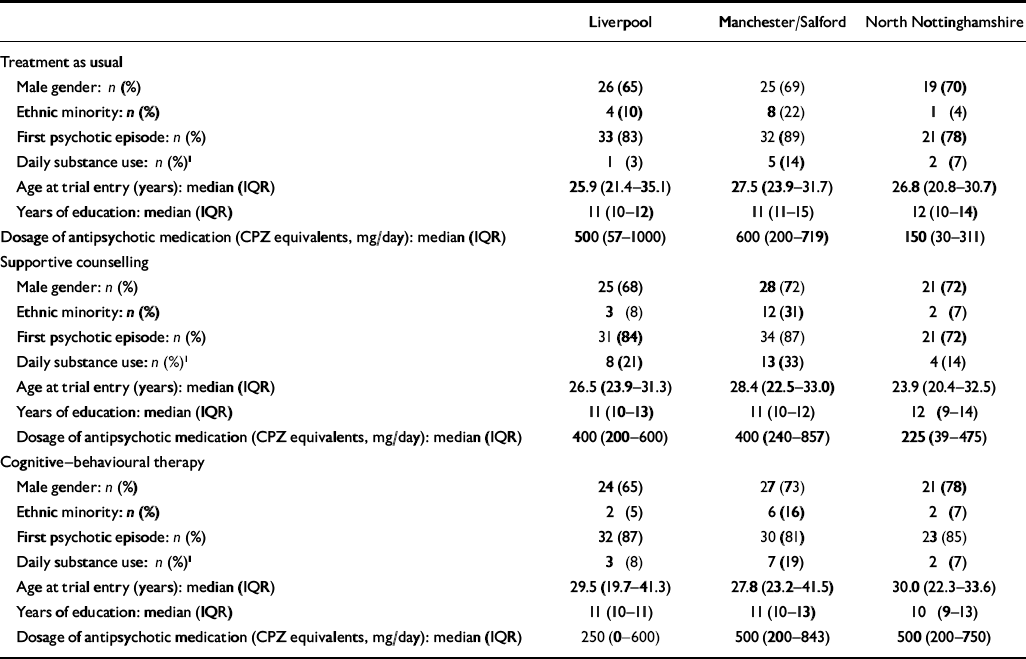
| Liverpool | Manchester/Salford | North Nottinghamshire | |
|---|---|---|---|
| Treatment as usual | |||
| Male gender: n (%) | 26 (65) | 25 (69) | 19 (70) |
| Ethnic minority: n (%) | 4 (10) | 8 (22) | 1 (4) |
| First psychotic episode: n (%) | 33 (83) | 32 (89) | 21 (78) |
| Daily substance use: n (%)1 | 1 (3) | 5 (14) | 2 (7) |
| Age at trial entry (years): median (IQR) | 25.9 (21.4–35.1) | 27.5 (23.9–31.7) | 26.8 (20.8–30.7) |
| Years of education: median (IQR) | 11 (10–12) | 11 (11–15) | 12 (10–14) |
| Dosage of antipsychotic medication (CPZ equivalents, mg/day): median (IQR) | 500 (57–1000) | 600 (200–719) | 150 (30–311) |
| Supportive counselling | |||
| Male gender: n (%) | 25 (68) | 28 (72) | 21 (72) |
| Ethnic minority: n (%) | 3 (8) | 12 (31) | 2 (7) |
| First psychotic episode: n (%) | 31 (84) | 34 (87) | 21 (72) |
| Daily substance use: n (%)1 | 8 (21) | 13 (33) | 4 (14) |
| Age at trial entry (years): median (IQR) | 26.5 (23.9–31.3) | 28.4 (22.5–33.0) | 23.9 (20.4–32.5) |
| Years of education: median (IQR) | 11 (10–13) | 11 (10–12) | 12 (9–14) |
| Dosage of antipsychotic medication (CPZ equivalents, mg/day): median (IQR) | 400 (200–600) | 400 (240–857) | 225 (39–475) |
| Cognitive–behavioural therapy | |||
| Male gender: n (%) | 24 (65) | 27 (73) | 21 (78) |
| Ethnic minority: n (%) | 2 (5) | 6 (16) | 2 (7) |
| First psychotic episode: n (%) | 32 (87) | 30 (81) | 23 (85) |
| Daily substance use: n (%)1 | 3 (8) | 7 (19) | 2 (7) |
| Age at trial entry (years): median (IQR) | 29.5 (19.7–41.3) | 27.8 (23.2–41.5) | 30.0 (22.3–33.6) |
| Years of education: median (IQR) | 11 (10–11) | 11 (10–13) | 10 (9–13) |
| Dosage of antipsychotic medication (CPZ equivalents, mg/day): median (IQR) | 250 (0–600) | 500 (200–843) | 500 (200–750) |
The final sample consisted of 309 participants, of whom 101 were allocated to CBT, 106 to supportive counselling and 102 to treatment as usual. At the 18-month follow-up assessment, 225 patients (73% of those randomised) were interviewed: of these, 75 had received CBT, 79 supportive counselling and 71 treatment as usual. Of those patients unavailable, 7 had died during the follow-up period: 3 definite suicides (2 from the supportive counselling group and 1 from the CBT group), 1 probable suicide (CBT group), 1 probably accidental death (supportive counselling group), and 2 from natural causes (both from the CBT group). Four patients withdrew consent, and 73 were lost to follow-up. Participant hospital data were available for assessment of re-hospitalisation in 307 (99% of the sample) and case notes for assessment of relapse in 295 (95%). The median number of days until assessment (with interquartile range) was, for each group: CBT, 575 (IQR 557-616), supportive counselling, 571 (557-617) and treatment as usual, 582 (554-640).
Assessor masking
Assessors’ guesses at treatment allocation were no better than chance (χ2=2.69, d.f.=4, P=0.61).
Analysis of outcome
Symptom status
The data from the baseline and 18-month follow-up assessment are given in Table 2. Comparisons were made between the treatment groups of scores on the PANSS total, positive, negative and general sub-scales, and of the delusion and auditory hallucination total scores of the PSYRATS, by means of separate ANCOVAs with relevant baseline score, time to assessment, centre, gender, hospitalisation, first or second episode and duration of untreated psychosis as covariates (Table 3). Supplementary comparisons were made between treatment groups and centres (Table 4). Receipt of treatment (either CBT or supportive counselling) significantly improved outcome (compared with treatment as usual) as measured by the PANSS total score and by all three of the PANSS sub-scales (positive, negative and general). There was, however, no difference for delusions and auditory hallucinations on the PSYRATS. There was no significant difference between CBT and supportive counselling, although there was a trend towards significance for CBT to have a superior effect on hallucinations. There was no significant difference between the treatment groups in terms of antipsychotic medication (type of medication, use of clozapine (n=9), and dosage).
Table 2 Summary of clinical assessments at baseline and at 18-month follow-up

| CBT | Supportive counselling | Treatment as usual | |||||||
|---|---|---|---|---|---|---|---|---|---|
| n | Baseline mean (s.d.) | 18 months mean (s.d.) | n | Baseline mean (s.d.) | 18 months mean (s.d.) | n | Baseline mean (s.d.) | 18 months mean (s.d.) | |
| Liverpool | |||||||||
| PANSS score | |||||||||
| Total | 26 | 81.5 (16.9) | 53.7 (13.3) | 23 | 80.6 (13.3) | 53.0 (14.6) | 23 | 83.4 (11.1) | 69.5 (13.6) |
| Positive | 26 | 21.8 (3.5) | 12.0 (4.3) | 23 | 21.9 (3.0) | 13.3 (4.8) | 23 | 23.2 (3.5) | 16.3 (6.5) |
| Negative | 26 | 17.4 (6.8) | 12.8 (4.3) | 23 | 17.5 (5.8) | 11.8 (4.0) | 23 | 17.9 (5.6) | 17.6 (5.8) |
| General | 26 | 42.3 (9.5) | 28.9 (6.9) | 23 | 41.2 (7.8) | 27.9 (7.4) | 23 | 42.3 (6.8) | 35.7 (6.3) |
| Delusions (total) | 24 | 17.8 (5.5) | 3.5 (6.0) | 23 | 18.2 (4.8) | 5.9 (7.3) | 19 | 17.0 (3.0) | 8.6 (8.6) |
| Auditory hallucinations (total)1 | 11 | 29.3 (8.3) | 0.0 (0.0) | 11 | 33.1 (4.1) | 9.5 (14.4) | 10 | 27.6 (7.6) | 0.0 (0.0) |
| Manchester/Salford | |||||||||
| PANSS score | |||||||||
| Total | 25 | 98.7 (12.0) | 71.2 (15.8) | 30 | 101.7 (17.3) | 76.6 (21.7) | 26 | 94.8 (16.3) | 73.2 (21.9) |
| Positive | 25 | 27.2 (4.0) | 14.8 (4.1) | 30 | 26.7 (4.3) | 16.2 (6.2) | 27 | 24.8 (4.7) | 17.2 (7.1) |
| Negative | 25 | 21.9 (5.9) | 18.0 (5.9) | 30 | 22.4 (7.4) | 18.9 (5.9) | 27 | 20.1 (6.9) | 17.4 (7.1) |
| General | 25 | 49.7 (7.3) | 38.3 (10.8) | 30 | 52.6 (9.4) | 41.5 (12.2) | 27 | 49.9 (7.7) | 39.1 (11.2) |
| Delusions (total) | 25 | 17.2 (3.7) | 8.9 (6.9) | 27 | 17.2 (6.0) | 9.2 (8.1) | 25 | 16.1 (5.5) | 7.9 (7.3) |
| Auditory hallucinations (total)1 | 15 | 28.5 (8.1) | 7.1 (12.5) | 20 | 28.5 (8.1) | 14.2 (16.2) | 12 | 30.9 (5.3) | 7.0 (12.9) |
| North Nottinghamshire | |||||||||
| PANSS score | |||||||||
| Total | 24 | 78.4 (11.8) | 51.5 (7.5) | 26 | 83.0 (10.6) | 51.4 (9.6) | 21 | 85.8 (13.7) | 54.5 (10.1) |
| Positive | 24 | 20.8 (3.4) | 10.5 (2.5) | 26 | 20.8 (3.4) | 10.2 (2.5) | 21 | 22.6 (4.4) | 11.6 (3.6) |
| Negative | 24 | 17.3 (4.5) | 12.2 (2.9) | 26 | 19.1 (3.7) | 12.3 (3.5) | 21 | 19.0 (4.4) | 12.4 (3.3) |
| General | 24 | 40.3 (6.1) | 28.8 (3.8) | 26 | 43.0 (6.3) | 28.9 (4.7) | 21 | 44.3 (7.3) | 30.5 (5.1) |
| Delusions total | 23 | 17.9 (4.4) | 5.5 (5.3) | 25 | 17.9 (4.3) | 6.4 (5.2) | 19 | 18.2 (3.2) | 7.9 (5.8) |
| Auditory hallucinations total1 | 14 | 28.9 (5.2) | 4.4 (8.9) | 15 | 26.4 (6.0) | 2.7 (7.3) | 17 | 28.8 (5.5) | 8.4 (10.9) |
Table 3 Estimated treatment effects

| Outcome | Receipt of treatment1 | |||
|---|---|---|---|---|
| (CBT/SC v.TAU) | CBT v. SC | |||
| Estimate (s.e.) | P | Estimate (s.e.) | P | |
| PANSS | ||||
| Total score | -6.22 (2.17) | 0.005 | -0.85 (2.42) | 0.725 |
| Positive | -2.36 (0.70) | 0.001 | -0.80 (0.78) | 0.309 |
| Negative | -1.51 (0.72) | 0.034 | 0.21 (0.80) | 0.797 |
| General | -2.57 (1.18) | 0.030 | -0.32 (1.32) | 0.811 |
| Delusions | -1.67 (1.04) | 0.111 | -1.04 (1.14) | 0.357 |
| Auditory hallucinations2 | 0.50 (2.80) | 0.827 | -4.36 (2.52) | 0.086 |
Table 4 Separate analyses of treatment effects for each centre

| Receipt of treatment1 | ||||
|---|---|---|---|---|
| SC v. TAU | CBT v. TAU | |||
| Estimate (s.e.) | P | Estimate (s.e.) | P | |
| PANSS total score | ||||
| Centre L | -16.51 (4.09) | <0.001 | -15.62 (3.93) | <0.001 |
| Centre M | 0.31 (5.19) | 0.952 | -4.63 (5.36) | 0.390 |
| Centre N | -3.36 (2.59) | 0.200 | -1.02 (2.59) | 0.696 |
| (Global test of treatment-centre interaction: F (4,209)=2.87; P=0.024) | ||||
| PANSS positive sub-scale | ||||
| Centre L | -2.77 (1.60) | 0.088 | -4.20 (1.55) | 0.009 |
| Centre M | -1.36 (1.60) | 0.405 | -2.96 (1.69) | 0.084 |
| Centre N | -1.47 (0.83) | 0.082 | -0.67 (0.81) | 0.414 |
| (Global test of treatment-centre interaction: F (4,210)=0.76; P=0.551) | ||||
| PANSS negative sub-scale | ||||
| Centre L | -5.89 (1.34) | <0.001 | -4.58 (1.29) | 0.001 |
| Centre M | 0.28 (1.60) | 0.173 | -0.22 (1.66) | 0.895 |
| Centre N | -0.47 (0.97) | 0.632 | -0.04 (0.96) | 0.965 |
| (Global test of treatment-centre interaction: F (4,210)=3.40; P=0.010) | ||||
| PANSS general sub-scale | ||||
| Centre L | -8.00 (1.99) | <0.001 | -6.97 (1.92) | <0.001 |
| Centre M | 1.21 (2.90) | 0.678 | -1.20 (3.00) | 0.691 |
| Centre N | -1.49 (1.37) | 0.281 | -0.96 (1.36) | 0.485 |
| (Global test of treatment-centre interaction: F (4,210)=2.52; P=0.042) | ||||
| Delusions | ||||
| Centre L | -3.37 (2.26) | 0.141 | -5.84 (2.18) | 0.010 |
| Centre M | 1.15 (2.11) | 0.589 | 0.50 (2.15) | 0.817 |
| Centre N | -2.29 (1.74) | 0.193 | -2.60 (1.65) | 0.120 |
| (Global test of treatment-centre interaction: F (4,195)=1.17; P=0.326) | ||||
| Auditory hallucinations | ||||
| Centre L | 7.41 (4.22) | 0.092 | -0.40 (4.19) | 0.926 |
| Centre M | 8.24 (5.42) | 0.137 | 1.78 (5.79) | 0.760 |
| Centre N | -5.02 (3.60) | 0.170 | -3.89 (0.274) | 0.274 |
| (Global test of treatment-centre interaction: F (4,110)=1.82; P=0.129) | ||||
The association of missing 18-month PANSS data (85 cases out of the 309 with baseline measurements) with baseline measures and treatment variables was examined. There were significant effects of centre (P=0.002) and number of sessions of therapy (P=0.022). There was no overall difference between the genders in loss to follow-up, but men were significantly more likely to withdraw from the study in one of the centres (P=0.023). A logistic model was constructed to predict missing 18-month outcome data. The explanatory variables were number of sessions, centre, gender, and a centre-gender interaction. The reciprocal of the predicted probability of having a non-missing outcome was then used as an inverse probability weight in a repeat of the main outcome analyses for PANSS positive, negative, general and total scores. The results were essentially the same as those given in Table 3.
There were significant centre-treatment interactions for PANSS total score and negative and general sub-scales. Separate analyses by centre (Table 4) indicated strong treatment effects at the Liverpool centre, with CBT and supportive counselling appearing to be superior to treatment as usual on nearly all measures, but no significant result from either of the other two centres. However, with the possible exception of hallucinations, there appeared to be no difference between centres in the outcomes after CBT and supportive counselling.
Re-hospitalisation and relapse
Data were available for 307 (99%) participants for hospital admission and 285 (92%) for relapse. Rates of rehospitalisation were: CBT, 33/101 (33%); supportive counselling, 31/106 (29%); treatment as usual, 37/102 (36%). Rates of relapse were: CBT, 53/97 (54.6%); supportive counselling, 50/96 (52.1%); treatment as usual, 47/92 (51.1%). There was no significant difference in number or survival times (Cox's regression) for either re-hospitalisation or relapse between the groups.
Medication
Data on medication dosage were available for 171 patients (78% of the sample) who completed the 18-month follow-up. The medians and range of daily medication dosage (in chlorpromazine equivalents) were as follows: CBT, 500 mg (0-1250); supportive counselling, 400 mg (0-1700); treatment as usual, 342.9 mg (0-1800). Medication compliance data were available for 103 patients, or 80% of both the Manchester and Liverpool sample who completed follow-up. Generally medication compliance was good, with median scores and ranges on the Barrowclough compliance scale (Reference Barrowclough, Tarrier and LewisBarrowclough et al, 1999) as follows: CBT, 3 (1-4); supportive counselling, 4 (1-4); treatment as usual, 3 (1-4). However, 20 patients (19% of those assessed) were classified as very poor on compliance. Drugs Attitude Inventory data were available from 133 (68%) of the 195 patients from the Manchester and Nottinghamshire centres, but data were not available from the Liverpool centre. There was no significant difference between treatment groups on any of the medication measures. Medication dosage, compliance and DAI had no effect on therapy-group scores for PANSS total scores. Thus, medication variables did not appear to influence treatment-group differences.
DISCUSSION
The results indicate that psychological treatment is beneficial in terms of symptom profile at follow-up but offers no advantage in terms of relapse or re-hospitalisation. The results emerging from the follow-up phase of this study closely resemble those of the acute phase and of a similarly designed trial involving patients with chronic schizophrenia experiencing residual symptoms (Reference Tarrier, Kinney and McCarthyTarrier et al, 2000). Treatment-group differences could not be explained by differences in medication dosage or compliance.
CBT v. supportive counselling
Contrary to the study hypothesis, there is little general difference between the effects of CBT and supportive counselling, although both appear to be superior to treatment as usual in terms of symptoms. We supposed that supportive counselling would have minimal effects above those of usual treatment, and that the specific techniques used in CBT would be the active agents. In the acute phase there was significantly faster recovery in the CBT group, with a further trend that supportive counselling appeared to slow resolution in auditory hallucinations, so that usual treatment alone resulted in faster resolution than usual treatment in combination with supportive counselling. Evidence from a trial of therapy in patients with chronic schizophrenia also indicated that, unlike delusions, hallucinations responded poorly to counselling (Reference Tarrier, Kinney and McCarthyTarrier et al, 2001). There are trends in the current data that reflect this, although centre effects and the smaller number of patients experiencing hallucinations at follow-up (n=84) limit the power of the analysis.
Direct comparison with other published trials is difficult, as populations and therapeutic aims differ. For example, in a trial studying patients with chronic schizophrenia a form of supportive counselling - befriending - resulted in equivalent improvement to that achieved with CBT during treatment, but at follow-up the benefit had declined in the supportive group (Reference Sensky, Turkington and KingdonSensky et al, 2000). We therefore expected a clear superiority of CBT over supportive counselling at follow-up, but this was not evident. It may be that the form of counselling practised in this and an earlier trial (Reference Tarrier, Yusupoff and KinneyTarrier et al, 1998) is more powerful than befriending, or that the treatment of acute disorder in our trial does not allow comparison with the treatment of chronic disorder; but since supportive counselling is unstructured, it is difficult to understand why it performs as well as it does, although this has been speculated upon elsewhere (Reference Tarrier, Kinney and McCarthyTarrier et al, 2000). However, there appear to be certain symptoms, specifically auditory hallucinations, for which supportive counselling is not beneficial.
In the only comparable study involving the treatment of acutely ill patients, Drury et al (Reference Drury, Birchwood and Cochrane1996a ) reported a significantly greater decline in positive symptoms in patients who had received CBT compared with those who had received a recreation and support control treatment. However, there are a number of differences between that and the one reported here. In the former study, CBT included individual and group cognitive therapy, as well as ‘family engagement’ and a structured activity programme. The latter included interpersonal and self-care skills. Thus, the content and the duration were significantly different from our study, in which CBT was individual and relatively brief. Finally, the study by Drury et al suffered methodological flaws that might have resulted in an optimistic estimate of effect size.
Relapse rates
Relapse rates overall were high. Previous studies suggest that for patients with first episodes of schizophrenia, relapses accumulate with time. Robinson et al (Reference Robinson, Woerner and Alvir1999a ) reported that approximately 16% of patients had relapsed by the end of 12 months and 54% by the end of 24 months, with the vast majority (82%) experiencing a relapse within 5 years. Contrary to the study hypothesis, there was no reduction in relapse rates in patients who received CBT; in fact, the rates across the three groups were remarkably similar. In the pilot study there had been marked (although non-significant) differences in re-significant) lapse rates between the CBT group (44%) and the supportive counselling group (73%) as well as longer times to relapse and fewer days in hospital (Reference Haddock, Tarrier and MorrisonHaddock et al, 1999a ).
In retrospect, it might have been over-optimistic to expect approximately 8 hours of therapy during the first 5 weeks of an acute episode to have significant benefits 18 months later. Moreover, the mean duration of treatment (8 h) was markedly less than the 15-20 hours proposed. This shorter treatment duration resulted from logistical problems and high levels of patient disturbance. Successful treatment requires patients to perceive their environment in a different way from that when they are highly symptomatic. To successfully sustain this it may well be necessary to persist with CBT for a much longer period, as would be expected with a maintenance drug treatment. Furthermore, the hospital environment in which the treatment was provided was very different from the community environment in which patients lived for most of the follow-up period, both in stimulus characteristics and social context. Thus, generalisation of effect might have been small and exposure to stressful interpersonal environments probable; for example, patients might have returned to families with high levels of expressed emotion, with its associated risk of exacerbation (Reference Butzlaff and HooleyButzlaff & Hooley, 1998).
Centre effects
There were significant interactions between centre and treatment group at the 18-month assessment for the PANSS total score and the PANSS negative and general sub-scales. In each of these cases the pattern was the same. Supplementary analyses comparing supportive counselling with treatment as usual, and CBT with treatment as usual, showed a highly significant benefit for both psychological treatments over usual treatment alone in the Liverpool centre. In the other two centres the differences between groups for these variables were not significant.
The differences between centres could have been generated by therapists, raters, services or patients, and deciding with certainty which factors were most important is impossible in so few centres. Examining the data from the treatment as usual group for the three centres, the North Nottinghamshire centre had the lowest mean PANSS scores at baseline and at 18 months, reflecting lower threshold criteria for hospital admission, which might have led to more successful routine treatment and reduced the scope for psychological intervention to provide further benefit. Fewer patients relapsed or were readmitted in this centre. In contrast, Manchester/Salford had the greatest deprivation and the highest PANSS scores at baseline, and these more severely ill patients were more likely to be readmitted and re-hospitalised. Patients in this centre might have been unable to realise the benefits of therapy because the burden of symptoms, psychosocial pressures and deprivation was too great. Inferences about the relative effectiveness of the therapists in the different centres are impossible to make with any certainty, given the complexity of the overall situation. However, it is notable that the therapist in the Nottinghamshire centre, owing to an unexpectedly high rate of recruitment in that centre and the wide geographic dispersal of the patients, recorded the lowest overall contact time.
Optimum psychosocial management
In summary, these follow-up results of patients with early schizophrenia are in agreement with those found in the follow-up of patients with chronic disease receiving psychological treatment (Reference Tarrier, Kinney and McCarthyTarrier et al, 2000). Patients receiving either CBT or supportive counselling in combination with usual treatment demonstrated better symptomatic recovery but no significant reduction in relapse compared with those receiving usual treatment alone. Family interventions have consistently been shown to reduce relapse rates in studies of both efficacy (Reference Tarrier, Barrowclough and PorcedduTarrier et al, 1994; Reference Pitschel-Waltz, Leucht and BaumlPitschel-Waltz et al, 2001) and effectiveness (Reference Barrowclough, Tarrier and LewisBarrowclough et al, 1999; Reference Sellwood, Barrowclough and TarrierSellwood et al, 2001). We suggest that the optimum psychosocial management of early schizophrenia would include a combination of CBT and family intervention. Such a combination has recently been shown to reduce psychotic symptoms and relapse in dual diagnosis patients (comorbid schizophrenia and substance misuse) (Reference Barrowclough, Haddock and TarrierBarrowclough et al, 2001).
Clinical Implications and Limitations
CLINICAL IMPLICATIONS
-
▪ ▪ Cognitive-behavioural therapy (CBT) and supportive counselling delivered during an acute admission have beneficial effects on psychotic symptoms at18 months follow-up compared with treatment as usual.
-
▪ ▪ Auditory hallucinations respond better to CBT than to supportive counselling.
-
▪ ▪ Individual psychological therapy, unlike family interventions, appears to have little effect on relapse rates.
LIMITATIONS
-
▪ ▪ There was a significant centre effect.
-
▪ ▪ Supportive counselling, used as a non-specific control treatment, had a more powerful effect than anticipated, but was an unstructured intervention.
-
▪ ▪ The duration of psychological treatments was briefer than had been planned.
Acknowledgements
We thank members of the independent trial steering committee: Professor E. Paykel (Cambridge; Chair), Professor Peter Tyrer and Professor Til Wykes, and. the independent data monitoring and ethics committee: Professor Peter Diggle (Lancaster; Chair), Professor David Clark and Professor Steven Hirsch. The. trial was funded by the UK Medical Research Council (41%); Northwest England. Regional NHSE Office (27%); Trent Regional NHSE Office (7%); and the following. Health Authorities: Manchester (8%); Salford and Trafford (2%); Liverpool (3%); Sefton (3%); St Helens and Knowsley (3%); North Nottinghamshire (6%).








eLetters
No eLetters have been published for this article.