INTRODUCTION
Norovirus (NoV) is the most commonly reported viral cause of foodborne outbreaks in the European Union, affecting 3784 people in 111 outbreaks in 2007 [1]. Food can be contaminated during its production by washing or growing in faecally contaminated water, or through preparation by an infected food handler. Oysters can accumulate NoV in their tissues [Reference Tian2] and are often implicated in foodborne NoV gastroenteritis outbreaks as they are traditionally consumed raw or lightly cooked [Reference Ang3].
Worldwide, the majority of NoV outbreaks have been associated with NoV genogroup II (GII) genotypes, in particular genotype 4 (GII.4) [Reference Siebenga4]. Most food handler-related outbreaks or person-to-person outbreaks are associated with a single NoV genotype [Reference Noda, Fukuda and Nishio5, Reference Matthews6]. In contrast, multiple NoV GI and GII genotypes are frequently identified in shellfish-related outbreaks, being detected in the faeces of infected patients and implicated shellfish [Reference Noda, Fukuda and Nishio5, Reference Le Guyader, Atmar and Le Pendu7]. Furthermore, a significantly higher attack rate has been reported in oyster-related outbreaks than in food handler-associated outbreaks [Reference Noda, Fukuda and Nishio5].
In Europe the sanitary quality of shellfish harvest areas is assessed by E. coli monitoring under regulation (EC) No. 854/2004. Harvest areas are classified into categories on the basis of E. coli concentrations and the level of treatment required prior to consumption. Despite these controls, oysters compliant with EU regulations have been implicated in outbreaks of NoV illness [Reference Le Guyader8, Reference Doré9]. Recently, a standardized real-time quantitative reverse transcription polymerase chain reaction (RT-qPCR) for the detection of NoV and hepatitis A in shellfish has been developed by the European Committee for Standardization (CEN) Working Group (TC275/WG6/TAG4) [Reference Lees10]. Although the standardized RT-qPCR method has been considered suitable for the detection and quantification of NoV in oysters by the European Food Safety Authority, guidance or regulatory limits for NoV concentrations in oysters have yet to be established [11].
In this study, we investigated two NoV oyster-related outbreaks that occurred during the winters of 2010 and 2012 in Ireland. We used the newly standardized RT-qPCR method to determine the concentrations of NoV in the implicated oysters. In addition, sequencing procedures were used on oyster and faecal samples to identify the causative NoV GI and GII genotypes.
METHODS
Outbreak descriptions and sampling
Outbreak 1 (2010)
From January to March 2010, 334 cases of NoV infection were linked to the consumption of raw oysters originating from several harvest areas in Europe [Reference Westrell12]. About 70 of these cases originating at restaurants throughout Ireland and the UK were linked to a single commercial harvest area (harvest area 1) in Ireland. The implicated oysters were harvested before 5 February from commercial harvest area 1 in Ireland. The area was classified as a category A production area, which means that the oysters could be sold directly for consumption. However, as an additional precaution, oysters were also depurated prior to sale [Reference Doré9]. Following reports of NoV gastroenteritis, oyster samples were collected and analysed from two sites (sites 1 and 2) within harvest area 1 on 14 February. The remains of oysters directly linked to illness were received and analysed from two Irish restaurants (A and B) on 18 February. A stool sample from an infected consumer that had an oyster meal in restaurant B on 5 February was analysed by the National Virus Reference Laboratory (NVRL) on 17 February and sequence data was available for further phylogenetic analysis.
Outbreak 2 (2012)
On 11 January 2012, the competent authorities in Ireland were informed that 18 people had reported gastroenteritis symptoms following consumption of oysters served as a portion of up to six oysters each per person at restaurant C. These oysters originated from a harvest area in the west of Ireland (harvest area 2). An oyster sample obtained directly from the batch of oysters causing illness and served at restaurant C was collected and analysed on 11 January for NoV. In addition to the restaurant oysters, an oyster sample was collected from the implicated harvest area (harvest area 2) on 12 January. Harvest area 2 was classified as a category B harvest area and depuration was routinely undertaken by the producer prior to dispatch. E. coli concentrations detected in oysters from restaurant C and harvest area 2 were 20 and <20 most probable number 100/g, respectively. Four stool samples were collected from infected consumers and were analysed for NoV.
Sample preparation and RNA extraction
Upon arrival at the laboratory, ten oysters (Crassostrea gigas) from each sample were opened and the digestive tissue (DT) dissected out. To prepare the shellfish extract, 2 g oyster DT was weighed to which 2 ml of 100 μ/ml proteinase K solution (30 U/mg; Sigma-Aldrich, USA) was added [Reference Flannery13]. Mengo virus strain MC0 was used as an internal positive control (IPC) virus. Viral RNA was extracted using NucliSENS® miniMAG® platform and NucliSENS® magnetic extraction reagents (bioMérieux, France) according to the manufacturer's protocol.
For each stool sample, 1 ml PBS (Oxoid, UK) was added to a 2-ml tube containing between 73 and 218 mg faecal material (neat) and vortexed vigorously. Then, 100 μl of the re-suspended faecal material (neat) was transferred into a fresh tube containing 900 μl of PBS (10−1) and serial dilutions were prepared up to 10−5. Virus RNA was extracted from 500 μl of each dilution using NucliSENS magnetic extraction reagents (bioMérieux) and eluted into 100 μl elution buffer.
Quantification of NoV using one-step RT-qPCR
A previously described RT-qPCR was performed using RNA Ultrasense™ one-step quantitative RT-PCR system (Invitrogen, USA) on an AB7500 real-time PCR instrument (Applied Biosystems, USA) [Reference Rajko-Nenow14]. For NoV GI analysis, forward primer QNIF4 [Reference Da Silva15], reverse primer NV1LCR, and probe NVGG1p [Reference Svraka16] were used, and for NoV GII, forward primer QNIF2 [Reference Loisy17], reverse primer COG2R [Reference Kageyama18], and probe QNIFS [Reference Loisy17] were used. Primers Mengo209, Mengo110 and probe Mengo147 used were the same as those described by Pintò et al. [Reference Pintò, Costafreda and Bosch19] to analyse the IPC virus. A log dilution series (range 1 × 101 to 1 × 105 copies/μl) of plasmids carrying the GI and GII target sequences were included in duplicate on each RT-qPCR run and the number of NoV RNA genome copies/μl was determined. The limit of quantification (LOQ) and the limit of detection (LOD) for NoV GI and GII was calculated as 100 and 20 genome copies/g DT, respectively. All oyster samples were assessed for both RT-PCR inhibition using external control RNA and extraction efficiency using IPC.
For quantification of stool samples, five dilutions of each (from 10−1 to 10−5) were analysed on separate RT-qPCR runs to avoid cross-contamination and the geometric mean of dilutions (>LOD) were calculated and expressed as genome copies/g faeces. The LOD for NoV GI and GII was calculated as 20 genome copies/ml.
NoV molecular characterization and genotyping
Reverse transcription (RT) was performed using the High Capacity cDNA Reverse Transcription kit (Applied Biosystems) according to the manufacturer's protocol. The nested PCR was performed as described previously [Reference Rajko-Nenow20]. Briefly, sample cDNA (5 μl) was added to a first round of PCR mixture, containing primers COG1F, G1-SKR for NoV GI and COG2F, G2-SKR for NoV GII [Reference Kageyama18, Reference Kojima21]. The primers used in a second round of PCR were GISKF, GISKR for NoV GI and GIISKF, GIISKR for NoV GII [Reference Kojima21]. The nested RT-PCR products were cloned into pCR® 4-TOPO® vector according to the protocol for TOPO TA Cloning® kit (Invitrogen). About five clones were randomly selected for DNA sequencing with BigDye Terminator v. 3.1 Ready Reaction kit (Applied Biosystems) and analysed on ABI Prism 310 genetic analyser (Applied Biosystems).
Sequence alignment and phylogenetic analysis
Nucleotide sequences were aligned using the ClustalW algorithm of MegAlign software (DNAstar Inc., USA) and compared to reference strains retrieved from GenBank (http://www.ncbi.nlm.nih.gov/genbank/). Percent identity was calculated for each pair of sequences in the nucleotide alignment. The length of NoV alignment was 285 bp for NoV GI (5354-5645 of Norwalk virus M87661) and 294 bp for NoV GII (5085-5353 of Lordsdale virus X86557). Phylogenetic trees were computed using the maximum-likelihood method with PAUP* software version 4.0 [Reference Swofford22]. Using the hierarchal likelihood ratio test in Modeltest [Reference Posada and Crandall23], the F81+G and GTR+I+G models of evolution were selected for NoV GI and NoV GII phylogenetic analysis, respectively. The reliability of the generated tree was estimated by bootstrap analysis of 1000 replicates of the dataset using PAUP*. NoV genotypes were assigned based on the clustering in the phylogenetic tree (>70% bootstrap support).
The Genbank nucleotide sequence accession numbers for all the sequence analysed during this study are KC954402–KC954472.
RESULTS
Outbreak 1 (2010)
Concentrations of NoV GI and GII in the oyster sample (REST79) collected from restaurant A were <LOQ and 2350 genome copies/g DT, respectively (Table 1). Similar concentrations of NoV GI and GII were detected in oysters (REST80) from restaurant B; <LOD and 2040 genome copies/g DT, respectively. NoV GI concentrations in the two oyster samples collected from sites 1 and 2 following notification of the outbreak, were 195 and <LOD genome copies/g DT, respectively. NoV GII concentrations detected in sites 1 and 2 were 2890 and 1920 genome copies/g DT, respectively (Table 1). For all oyster samples, NoV GII concentrations were at least tenfold greater than NoV GI concentrations.
Table 1. Norovirus genogroup I (GI) and genogroup II (GII) concentrations and genotypes detected in outbreak samples

n.t., Not typed as nested RT-PCR amplification was unsuccessful.
* Sample tested by National Virus Reference Laboratory in Dublin.
† Real-time RT-qPCR results are expressed in genome copies/g faeces for stool samples and in genome copies/g digestive tissue for oyster samples.
Although NoV GI was below the LOD of the RT-qPCR assay, in two oyster samples (REST80 and HAR70, Table 1) NoV GI was amplified for genotyping by the nested RT-PCR assay and NoV GI.4 genotype was detected. A single NoV GII.3 genotype was detected in the oyster sample (REST79) collected from restaurant A, whereas four NoV genotypes (GI.4, GII.3, GII.4 New Orleans 2009, GII.13) were detected in the oysters (REST80) from restaurant B (Figs 1 and 2). Multiple NoV genotypes were also detected in oysters from site 1 (GI.4, GII.3, GII.13) and site 2 (GI.4, GII.12, GII.13) in harvest area 1.
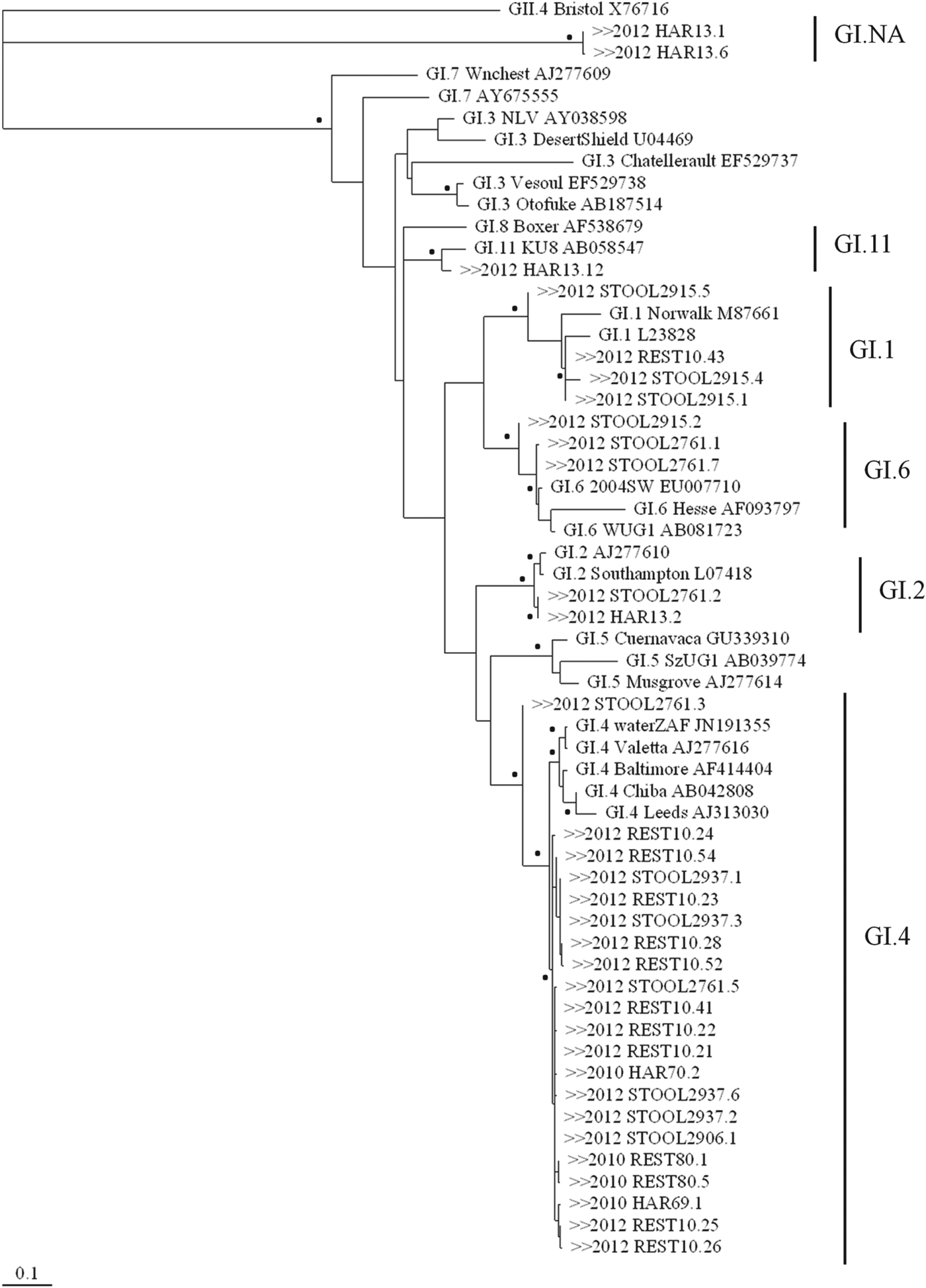
Fig. 1. Maximum-likelihood tree based on capsid N/S domain (285 bp) of the NoV GI sequence alignment. Bootstrap analysis was performed for 1000 replicates of the dataset and values of >70% are indicated by the black dots beside the appropriate branch. NoV GI sequences detected during outbreaks 1 and 2 are preceded by ‘>>2010’ and ‘>>2012’, respectively. The lower scale represents genetic distances in nucleotide substitutions per site.
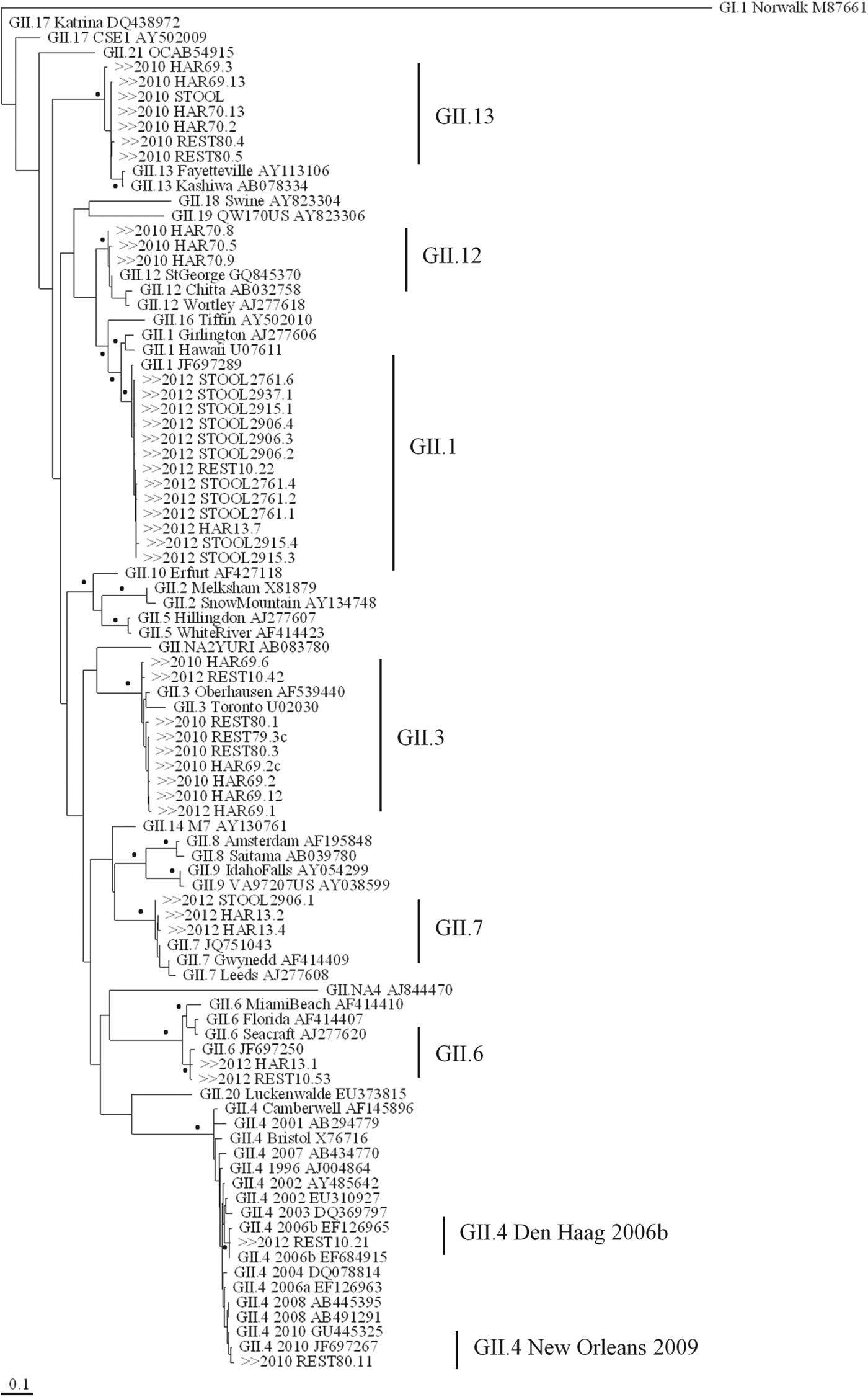
Fig. 2. Maximum-likelihood tree based on capsid N/S domain (294 bp) of the NoV GII sequence alignment. Bootstrap analysis was performed for 1000 replicates of the dataset and values of >70% are indicated by the black dots beside the appropriate branch. NoV GII sequences detected during outbreaks 1 and 2 are preceded by ‘>>2010’ and ‘>>2012’, respectively. The lower scale represents genetic distances in nucleotide substitutions per site.
A single NoV GII.13 genotype was detected by direct sequencing in the stool sample by the NVRL (Table 1). This NoV GII.13 sequence (STOOL) had 100% identity to a GII.13 sequence detected in harvest area 1, site 2 (HAR70.2) and 99·7% identity to sequences REST80.5 (restaurant B) and HAR69·13 (harvest area 1, site 1).
Outbreak 2 (2012)
The NoV GII concentration in the oyster sample from restaurant C (REST10) was 2380 genome copies/g DT, whereas the NoV GI concentration was <LOD of the RT-qPCR assay (Table 1). NoV GI and GII concentrations of 1790 and 4000 genome copies/g DT, respectively, were detected in the oyster sample from harvest area 2 (HAR13).
In the oysters that were consumed at restaurant C (REST10) and directly linked to illness, six NoV genotypes were detected; GI.1, GI.4, GII.1, GII.3, GII.4 Den Haag 2006b, and GII.6 (Figs 1 and 2). NoV GI.4 was detected in 74% (n = 14/19) and NoV GI.1 in 26% (n = 5/19) of the NoV GI clones in this sample. For NoV GII, the most frequently detected genotype was GII.4 Den Haag 2006b (41%, n = 9/22) followed by GII.3 (27%, n = 6/22), GII.1 (23%, n = 5/22), and GII.6 (9%, n = 2/22). Multiple NoV genotypes (GI.2, GI.11, GII.1, GII.6, GII.7) were also detected in oyster samples (HAR13) from harvest area 2, including an unidentified NoV strain that was designated GI.NA (Table 1).
Four stool samples were cloned prior to sequence analysis and multiple genotypes were detected in samples; 2937, 2915, 2906 and 2761 (Table 1). All stool samples contained NoV GII and concentrations ranged from 2·2 × 108 to 5·66 × 109 genome copies/g faeces, while only one stool sample (2906) had similarly high NoV GI concentrations (1·32 × 108 genome copies/g faeces). NoV GII.1 was the only genotype detected in all stool samples. NoV GII.1 sequences 2761·1, 2906·3, 2915·1, 2915·3 and 2937·1 detected in the stool samples shared 99·7% identity to a NoV GII.1 sequence found in the oyster sample from restaurant C (REST10.22) and harvest area 2 (HAR13.7). In addition to NoV GII.1, GII.7 genotype was also identified in the faeces of one patient (2906).
NoV GI concentrations of 1·32 × 108 and 3·46 × 105 genome copies/g faeces were detected in stool samples 2906 and 2937, respectively. In these stool samples, NoV GI.4 was the only GI genotype identified and sequences 2906·1 and 2937·2 shared 100% identity with the GI.4 sequence detected in the oyster sample from restaurant C (REST10.21). In contrast, the concentration of NoV GI detected in stool sample 2761 that contained three GI genotypes (GI.4, GI.2, GI.6) was 2·45 × 103 genome copies/g faeces. In stool sample 2915, concentrations of NoV GI were not detected using the RT-qPCR assay, yet were positive using the nested RT-PCR assay and NoV GI.1 and GI.6 were identified. NoV GI.6 was identified in two of the stool samples despite not being detected in the oyster samples collected from the restaurant or harvest area.
DISCUSSION
We analysed samples from two oyster-related outbreaks of NoV gastroenteritis that occurred in Ireland in 2010 and 2012. The implicated oysters were harvested from production areas designated as category A (outbreak 1) and category B (outbreak 2) under EU Regulation 854/2004 [24]. According to EU regulations, category A oysters are suitable for direct human consumption, whereas category B oysters require post-harvest treatment prior to consumption. Oysters harvested from both areas were routinely depurated prior to sale. Despite this, depurated oysters from both harvest areas contained total NoV (GI+GII) concentrations >1000 genome copies/g DT. This is consistent with a previous study that demonstrated that oysters containing a total NoV concentration >500 genome copies/g DT had a significantly increased risk of causing illness outbreaks when consumed than oysters containing concentrations <500 genome copies/g [Reference Lowther25]. Concentrations of NoV GI were close to the LOQ of the RT-qPCR in almost all oyster samples during the two outbreaks. One exception was the oyster sample collected on 12 January from harvest area 2 following notification of the outbreak. This sample contained 1790 genome copies/g DT and showed a different NoV genotype profile compared to the oysters that were previously harvested and were implicated in illness in consumers. It is likely that harvest area 2 underwent further contamination with NoV during the period 2–12 January 2012. In particular, on 4 January 2012 a significant rainfall (16·4 mm reported by Met Éireann, http://www.met.ie) event occurred, which may have caused the additional NoV contamination from the nearby wastewater treatment plant ~1 km from the oyster-growing area.
Oysters causing illness in the two outbreaks investigated here contained multiple strains of NoV GI and GII. In outbreak 1 a single NoV GII.13 strain was detected in the stool sample. However, as only direct sequencing was performed, it is possible that other NoV strains were present in this faecal sample. Baker et al. attempted direct sequencing of oysters associated with the wider outbreak and also originating from harvest area 1, but phylogenetic analysis was incomplete due to the presence of several NoV strains [Reference Baker26]. During outbreak 2, nested PCR products were cloned prior to sequencing and showed that all patients were infected with a mix of NoV GI and GII genotypes. Oysters served at restaurant C were contaminated with multiple NoV genotypes (GI.1, GI.4, GII.1, GII.3, GII.6, GII.4 Den Haag 2006b). However, not all of these genotypes were present in each of the individual stool samples. NoV GII.1 was present in all four, GI.4 in three and GI.1 in only one stool sample. Some NoV genotypes (GI.2, GI.6, GII.7) were shed by patients, but were not detected in the restaurant oysters (REST10). As asymptomatic NoV infections have been reported in 12% of the healthy population [Reference Phillips27], it is possible that the detection of these genotypes in faeces could not be attributed to the consumption of oysters. It appears that NoV GII.1 genotype was the most virulent genotype as it was detected in all stool samples along with high concentrations of NoV GII in faeces (>108 copies/g) determined by RT-qPCR. It has been demonstrated previously that different genotypes of NoV appear to cause differences in severity of disease [Reference Friesema28]. NoV GII.4 strains in particular were found to be associated with more frequent vomiting and higher attack rates in individuals.
The NoV GII.1 genotype detected in oysters and faecal samples during outbreak 2 shared high identity with the NoV GII.g/GII.1 recombinant (JF697289) [Reference Mathijs29] that caused a number of NoV outbreaks across Europe during 2010 and 2011 [Reference Van Beek30]. It is possible that NoV GII.1 was a newly circulating strain in Ireland at the time of outbreak 2. If so, it is likely that the population was highly susceptible to this particular strain due to lack of existing immunity. In addition, high concentration of NoV GII in the oysters, presumably including a correspondingly high GII.1 concentration, could contribute to the effective shellfish-borne spread of GII.1. During both investigated outbreaks, oysters directly linked to illness were contaminated with multiple NoV genotypes, including GII.3. However, this genotype was not detected in any of the stool samples which is in contrast with a previous study that demonstrated that NoV GII.3 had a greater attack rate in oyster-related outbreaks than any other genotype [Reference Noda, Fukuda and Nishio5]. This may be indicative of pre-existing immunity within the oyster consumers for NoV GII.3 during these outbreaks.
During outbreak 2, NoV GI was not detected in the restaurant oysters by the RT-qPCR assay, but was detected using the nested PCR and indicates that NoV GI was present at very low concentrations. Despite this, three out of the four consumers had NoV GI.4 in their faeces. In two of these three stool samples NoV GI.4 was the only NoV GI strain present and these contained NoV GI concentrations at 105 and 108 genome copies/g which could be indicative of an active infection. Comparable NoV GI.4 concentrations in faeces have been previously reported during a waterborne outbreak affecting more than 2400 people in Sweden [Reference Nenonen31]. These findings concur with a recent study that estimated a median infectious dose for NoV GI as low as 7·5 genome copies/oyster [Reference Thebault32]. It is possible that there may be considerable differences between the infectious doses of NoV genotypes and suggests that caution is required when ascribing the risk associated with oyster consumption based on total NoV concentrations. However, given the relatively low concentration and sporadic occurrence of NoV GI detected in stool samples it remains unclear from our investigations what role NoV GI.4 detected during this outbreak played in causing illness. In addition, a part of the NoV genome (~300 bp) encoding for the N/S domain was sequenced; therefore, it cannot be excluded that the 100% identity shared between the NoV GI.4 sequence detected in the stool and the oyster samples could significantly differ in other parts of the NoV genome, including the P2 domain.
Despite the continued occurrence of oyster-associated NoV outbreaks, viral standards for shellfish do not exist in EU food legislation and it is clear that E. coli monitoring of shellfish is unsuitable to indicate the risk posed by NoV-contaminated shellfish. In this study, two NoV-related outbreaks were caused by consumption of highly contaminated oysters, containing multiple NoV strains and total NoV concentrations >1000 genome copies/g DT. It has been demonstrated previously that such highly contaminated oysters can pose a significant risk to human health [Reference Lowther25]. While multiple NoV genotypes were present in oysters, not all appeared to be successful in causing NoV infection in consumers as judged in this study by the phylogenetic analysis. This suggests that factors such as pre-existing population immunity and differences in the infectious dose may impact on the ability NoV strains present in oysters to cause illness. This may have implications when considering setting acceptable concentrations for NoV standards in oysters.
DECLARATION OF INTEREST
None.





