INTRODUCTION
Despite a marked decline in the incidence of invasive pneumococcal disease (IPD) following universal childhood vaccination with heptavalent pneumococcal conjugate vaccine (PCV7), Streptococcus pneumoniae (SP) remains the most common cause of invasive bacterial disease in children. Isolates from IPD are now predominantly non-PCV7 serotypes, although some IPD from PCV7 serotypes continues to exist [Reference Pilishvili1, Reference Hsu2]. In contrast, non-PCV7 serotypes have essentially fully replaced PCV7 serotypes as the dominant carriage serotypes in the nasopharynx (NP) [Reference Huang3]. In fact, current carriage of non-PCV7 serotypes is at the same level as PCV7 serotype carriage prior to PCV7 vaccine. Since mucosal entry remains the initial step in the pathogenesis of IPD [Reference Gray, Converse and Dillon4], understanding risk and protective factors for SP NP mucosal carriage remains critical, particularly as new changes occur in the era of universal 13-valent pneumococcal conjugate vaccination (PCV13) [5].
The complete replacement of pneumococcal serotypes in the NP has raised questions about whether factors previously shown to confer risk or protection for carriage have changed. We have previously reported that individual risk factors of young age, childcare attendance, concurrent upper respiratory tract infection, and having young siblings remain associated with increased carriage despite the new dominance of non-PCV7 strains [Reference Huang3]. Similarly, recent antibiotic use remains associated with decreased odds of carriage [Reference Huang3, Reference Pebody6].
In this study we sought to assess whether community-level factors, also associated with carriage in previous studies, continued to predict carriage of non-PCV7 strains. We previously reported that in addition to individual risk factors, living in communities with crowded households and lower incomes independently predicted carriage of PCV7 serotypes, particularly in children who were not in group childcare [Reference Huang7, Reference Huang, Finkelstein and Lipsitch8] Here, we examine whether these previously identified community-level factors remain predictive of pneumococcal NP carriage in the PCV7 era, after complete serotype replacement with non-PCV7 strains [Reference Huang3]. We particularly examined potential differential effects of community-level factors according to participation in group childcare.
METHODS
We performed a secondary analysis of data from two studies examining NP colonization with SP (any SP, not limited to non-PCV7 serotypes) in young children aged <7 years during sick and well-child clinic visits. One study was conducted in eight Massachusetts communities outside of Boston [Reference Huang3]; and, a parallel study was conducted at a single site in urban Boston, both during the 2006–2007 winter respiratory virus season. Details of the eight Massachusetts communities outside of Boston have previously been described. The eight Massachusetts paediatric practices which participated from the eight Massachusetts communities, were selected because they were among the largest of the previously participating paediatric practices in a cluster-randomized trial of a community-level intervention to promote judicious antibiotic prescribing for children [Reference Finkelstein9, Reference Finkelstein10]. These eight communities were originally chosen on the basis of evidence that few children crossed community boundaries for paediatric care. Because these two study cohorts were drawn from very different populations served by very different paediatric practices, analysis of the cohorts was conducted separately. Data collection and microbiological processing were conducted using identical protocols as described previously [Reference Huang11]. In brief, adult caregivers consented to a survey of possible individual- and household-level predictors of pneumococcal carriage and collection of a swab of their child's NP by trained study staff. This survey included individual- and household-level questions about race/ethnicity, income and education level. Medical records were subsequently reviewed for pneumococcal vaccination history. NP samples were processed within 24 h and all pneumococcal isolates underwent serotyping as described previously [Reference Finkelstein10, Reference Huang11].
In addition to individual-level risk factors, community-level risk factors were evaluated to understand the contribution of population-level factors above and beyond individual characteristics. Previously evaluated community-level predictors [Reference Huang7] were obtained from the U.S. Census for the census tract of residence of each individual child by geocoding street addresses obtained from the medical record. U.S. Census tracts are a defined geographical area, usually with 2500–8000 persons, designed to be homogeneous with respect to population characteristics, economic status, and living conditions [12]. Census tract variables included population size, density, age and race distribution, use of childcare facilities, education, percent lacking in-unit plumbing, poverty, and community stability. All census tract variables were dichotomized around the median value for the studied cohorts, for consistent analysis between the two cohorts. Each census tract variable was also analysed as a continuous variable and using breakpoints initially established via recursive partitioning in a previous study [Reference Huang7]. Because the objective was to examine the contribution of community-level risk factors above and beyond individual- or household-level risk factors, our base models to which community-level factors were added, included individual- and household-level factors such as race/ethnicity, parental educational level (less than college graduation, or college graduation or better) and household income, regardless of whether those factors were significant after adjustment for other factors within the base model. Similarly, individual PCV7 vaccine exposure, defined as having received at least one dose of PCV7, was kept within base models because of its impact on select non-PCV7 serotypes, some of which cross-react with antibodies in PCV7 [Reference Bechini, Boccalini and Bonanni13]. In each cohort, adjusted associations between census tract variables and outcomes were evaluated by adding one census tract variable at a time to each cohort's base model, then removing it and replacing with another census tract variable. Specific stratification by childcare participation was performed in both cohorts, as had been performed previously [Reference Huang7]. All models were generalized linear mixed models that accounted for clustering by census tracts.
Two models were developed – one for the site in urban Boston and another for the eight communities outside of Boston. Analyses were performed using SAS, version 9.2 (SAS Institute Inc., USA). All study procedures were approved by the Harvard Pilgrim Health Care and Boston University Medical Center institutional review boards.
RESULTS
Individual characteristics of the two study populations are shown in Table 1. Overall, >95% of the study population had received at least one dose of PCV7. Both cohorts of children reported equivalent proportions of childcare participation and exposure to smokers within the home. Subjects in urban Boston were predominantly black, non-Hispanic (64%) and had a median household income of <US$35 000. Those from communities outside Boston were predominantly white, non-Hispanic (84%) with a higher median income (between US$50 000 and US$74 000). Fewer urban Boston children had an upper respiratory tract infection, current or recent antibiotic exposure, or siblings aged <6 years, compared to children outside Boston. Urban Boston families also reported lower levels of parental education.
Table 1. Characteristics of Massachusetts children who provided nasopharyngeal specimens, 2006–2007*

* Numbers vary because of missing data (participants excluded from analysis cohort if missing values for variables in multivariate model).
† Includes runny nose, sore throat, cough, bronchitis, ear pain/infection, sinus pain.
‡ Reflects the antibiotic status on the day of the swab, prior to being seen by the physician.
§ Doses of PCV7 completed 30 days prior to the swab date.
¶ Defined as an affirmative response to the question, ‘Does anyone in the child's home smoke cigarettes?’
Of 543 children from urban Boston, 92% received ⩾1 dose of PCV7 vaccine. Twenty-two percent (121 children) carried pneumococci, of which 93% were non-PCV7 serotypes (11% 19A, 11% 6A, 10% 35B, 9% 15 B/C, 7% 11A, 6% 15A, 6% NT). In the base model, individual-level predictors for pneumococcal carriage included young age, respiratory tract infection, and group childcare (Table 2). Race/ethnicity, recent antibiotic use, and receipt of at least one PCV7 dose were not found to be significant individual-level predictors of pneumococcal carriage.
Table 2. Base models analysing individual- and household-level factors associated with Streptococcus pneumoniae carriage, prior to addition of community-level variables
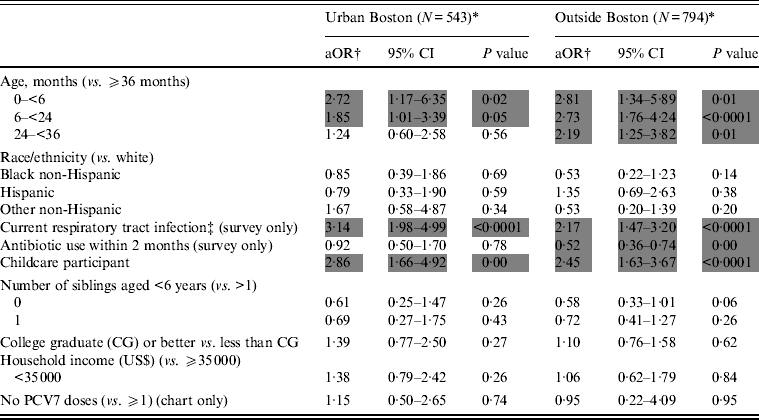
* GLIMMIX used to adjust for clustering by census tract number. Multivariable adjusted odds ratios (aOR) presented with lower and upper 95% confidence intervals (CI).
† Odds ratios adjusted for age, race/ethnicity, current respiratory tract infection, recent antibiotic use, childcare participation, education, household income, and PCV7 vaccine dose.
‡ Includes runny nose, sore throat, cough, bronchitis, ear pain/infection, sinus pain.
Of 794 children from the eight communities outside of Boston, 99% received ⩾1 dose of PCV7 vaccine. Thirty percent (241 children) carried pneumococci, of which 96% were non-PCV7 serotypes (16% 19A, 11% 15 B/C, 10% 6C, 9% 35B, 9% 35B, 9% 11A, 7% 23A, 6% 23B). Of note, overall pneumococcal carriage rates differed between urban Boston and outside Boston cohorts (22% vs. 30%, P = 0·0011). Consistent with results from the urban Boston children, base model individual-level predictors for pneumococcal carriage included young age, respiratory tract infection, and group childcare, but not race/ethnicity or PCV7 vaccine exposure (⩾1 dose of PCV7), (Table 2). However, in contrast to children from urban Boston, recent antibiotic use was additionally found to be protective against carriage.
Census tract data from the two study populations are shown in Table 3. There were marked differences in census tract characteristics between the urban Boston and outside Boston study participants, particularly in terms of population density, proportions of non-white residents, levels of adult education, levels of in-unit plumbing, and measures of poverty. However, census tracts of subjects in urban Boston and outside Boston had similar overall levels of household size, proportions of households with children aged <6 years, and proportions of households with children aged <6 years reporting both parents working.
Table 3. Census tract data for study participants
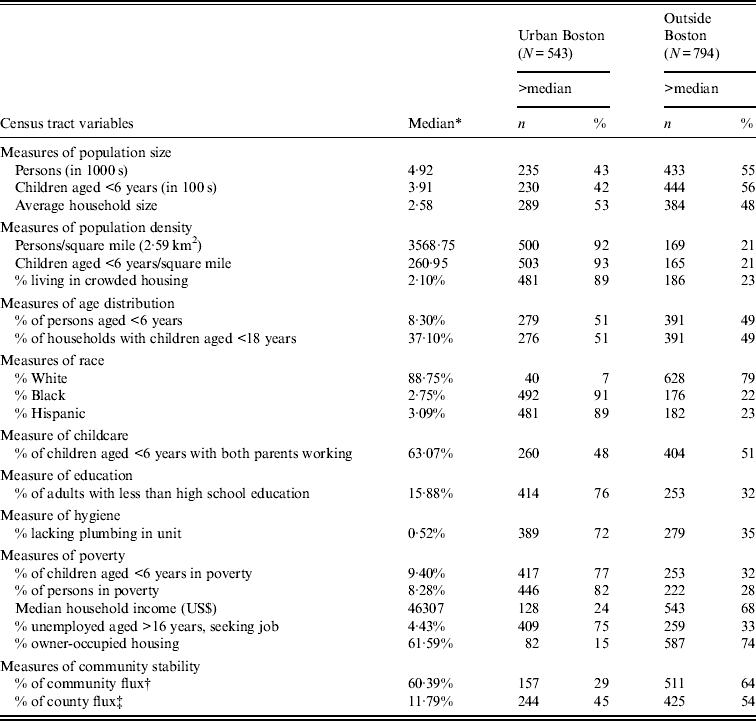
* Proportion above and below medians established by combining urban Boston and outside Boston cohorts.
† Census tract members, expressed as percentage, who relocated their homes between 1996 and 2000.
‡ Census tract members, expressed as percentage, who relocated from outside the county between 1996 and 2000.
Census tract measures did not significantly modify odds of carriage in overall models of urban Boston or outside Boston cohorts when using median values as breakpoints (Table 4), nor when using previous breakpoints from the 2004 study [Reference Huang7], nor when analysing census tract measures as continuous variables. However, a number of significant interactions were identified between group childcare exposure and census tract measures particularly in the urban Boston cohort. Therefore we further analysed community models stratified by group childcare exposure, and found differences between the two cohorts. These differences were not attributable to individual differences in socioeconomic status, as household income distribution did not differ between those in childcare and those not in childcare. In the urban Boston cohort, and only in those who attend group childcare, increased odds for pneumococcal carriage was seen in children who lived in census tracts characterized by markers of lower socioeconomic status (e.g. more adults with less than a high school education, or four out of five census tract measures of increased poverty, or more members who relocated homes). In contrast, measures of increased poverty were significantly protective against pneumococcal carriage in children who did not attend group childcare (Table 4). Living in census tracts with more adults with less than high school education appeared marginally protective against SP colonization in the outside Boston cohort, but no other census tract measures showed effect modification according to group childcare exposure.
Table 4. Analysis of census variables (above vs. below median unless otherwise noted) added one at a time to the base multivariate model* of individual-level factors associated with Streptococcus pneumoniae carriage
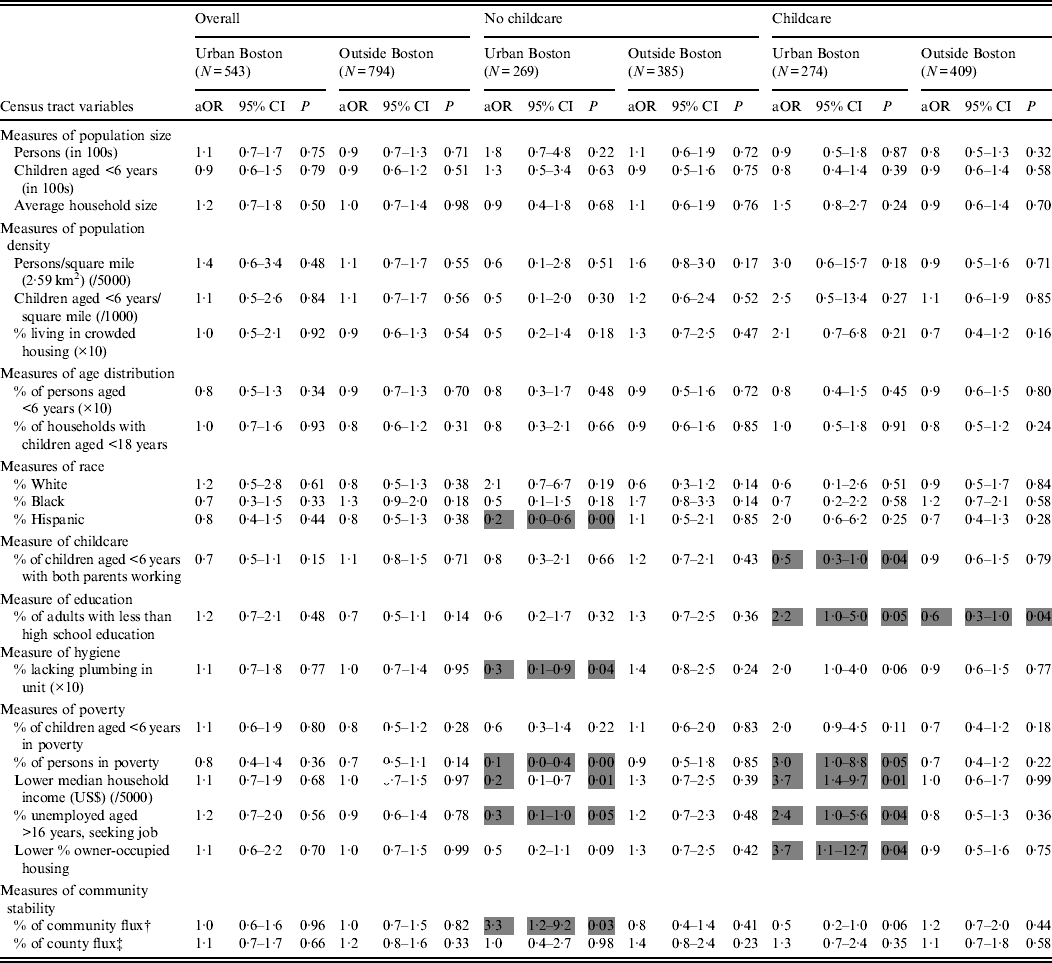
aOR, Adjusted odds ratio; CI, confidence interval.
* Base model shown in Table 2: odds ratios adjusted for age, race/ethnicity, current respiratory tract infection, recent antibiotic use, childcare participation, education, household income, and PCV7 vaccine dose.
† Census tract members, expressed as percentage, who relocated their home between 1996 and 2000.
‡ Census tract members, expressed as percentage, who relocated from outside the county between 1996 and 2000.
DISCUSSION
Community-level factors were previously shown to have independent effects on childhood NP pneumococcal carriage during the pre-PCV7 era [Reference Huang7]. Therefore we re-evaluated whether community-level factors continue to have an impact on pneumococcal carriage beyond individual factors in the era of widespread use of PCV7, after complete serotype replacement with non-PCV7 strains. Because of differential findings by childcare participation in previous analyses [Reference Huang7, Reference Huang, Finkelstein and Lipsitch8], we looked for differential effects of community-level factors in childcare participants vs. non-participants.
In the cohorts studied here where 90% of the study population had received at least one dose of PCV7, risk for carriage of primarily non-PCV7 serotypes (younger age, concomitant upper respiratory tract illness, and group childcare), appear to have remained relatively constant in our two demographically different cohorts of Massachusetts paediatric patients – those residing outside urban Boston (who tended to live in families with higher levels of parental education and income), and those in urban Boston (who tended to be non-white, with lower levels of parental education and income). Consistency of these individual-level risk factors for pneumococcal carriage was demonstrated despite noticeable differences in the overall rates of carriage and the proportion of children in each cohort who had current respiratory infection or recent antibiotic use.
Similar to what was found prior to widespread introduction of pneumococcal conjugate vaccine [Reference Huang7], no community-level measures of population size, population density, age distribution, race, childcare, education, hygiene, poverty, or stability were found to be significant predictors of pneumococcal colonization in overall models (not stratified by childcare) controlling for significant individual-level risk factors for pneumococcal colonization, as well as individual- and household-level race, education, household income, and PCV7 vaccination (Table 4).
While overall models identified no impact from community variables, we examined whether living in census tracts with larger average household size, or in socioeconomically disadvantaged communities, which had previously been associated with an excess risk (2- to 3-fold increased odds) of SP carriage in the pre-PCV7 era in childcare attendees, still continued to confer excess risk [Reference Huang7]. In this PCV7 era follow-up study, we were no longer able to demonstrate consistent linkages with childhood pneumococcal carriage in children attending or not attending group childcare in the outside Boston cohort. It is possible that widespread use of PCV7 has reduced the impact of community-level sociodemographic characteristics and essentially ‘levelled the playing field’, so to speak; alternatively, non-PCV7 serotypes now occupying the NP niche may not be as affected by community variables compared to previous serotypes.
In the urban Boston cohort the results were more puzzling. It appeared that living in socioeconomically disadvantaged census tracts was protective against non-PCV7 serotype pneumococcal carriage in those unexposed to group childcare, but in those exposed to group childcare, the opposite effect was seen (i.e. increased risk of carriage). Since there is no known mechanism that would explain this finding, we consider it only hypothesis-generating for further analyses in other cohorts. An alternative explanation is that the finding from this model, regarding community-level factors, represents a spurious association (Type 1 error) with pneumococcal carriage. It is also important to point out that both the individual- and community-level variables assessed here represent markers or proxies for biological risk factors; we do not assert a particular mechanism by which they affect carriage directly.
In conclusion, in a highly vaccinated population 6 years after universal childhood vaccination with PCV7 was begun, living in census tracts with larger households, or that are socioeconomically disadvantaged, no longer consistently confers increased odds of childhood non-PCV7 serotype pneumococcal carriage above individual or household predictors of pneumococcal carriage, even in children not participating in group childcare. The finding that some community-level variables impact carriage rates differentially (with associations seen only in some childcare exposure groups, and only in some communities) warrants further study.
ACKNOWLEDGEMENTS
We thank Lizzie Ford, Verna Moran, RN, Shannon Opel, Divya Reddy, MD, MPH, Sam Theodore, MD, and Kelly Welch for their dedication to recruitment, enrolment, and data entry for this research project. We also gratefully acknowledge the participation of nine Massachusetts paediatric practices and their many patient families, whose individual contributions made our population-based analyses possible. Support for this study came from National Institute of Allergy and Infectious Diseases (NIAID) Grant R01 AI066304 and Pfizer, Inc. Investigator-Initiated Research Grant WS1591672.
DECLARATION OF INTEREST
S. I. Pelton has received investigator-initiated research grant funding (Pfizer, Merck), and honoraria for participation on Pneumococcal Vaccine Advisory Boards (Pfizer, GSK Bio, Merck).






