Psittacosis is caused by Chlamydophila psittaci, mainly after exposure to infected birds or other animals [1, 2]. Infection usually occurs when a person inhales the aerosolized organism from dried faeces or respiratory secretions, which can occur through mouth-to-beak contact or handling of infected birds [3]. Psittacosis typically causes influenza-like symptoms but can also lead to severe pneumonia, endocarditis, myocarditis, or encephalitis [3]. The incubation period is typically 5–14 days but longer periods have been reported [3]. Since April 1999, psittacosis has been a notifiable disease in Japan.
On 23 July 2001, Bird Park X opened in Matsue City, Shimane Prefecture. Among around 1300 birds held in this park about 660 birds were housed in hothouses. Visitors could enter a hothouse that housed birds from temperate regions (W) and two other hothouses that housed birds from the tropics (T and P). Most of the birds were allowed to fly freely in W, T and P and visitors were allowed to feed birds in P. Birds not on display were maintained in the backyard, located in the same building as P. Only staff were allowed to enter the backyard and the staff building. From 31 December 2001 to 12 January 2002, five psittacosis cases (one student, three staff members and one visitor), were reported to the National Notifiable Disease Surveillance System. Bird Park X closed T and P on 14 January 2002 and closed the entire park from 16 January 2002 to May 2002. From the park's opening to its temporary closing, about 285 000 persons had visited the park.
A psittacosis case was defined as a person related to Bird Park X, who developed symptoms after 30 October 2001, with a physician's diagnosis of psittacosis, confirmed by complement fixation (CF) antibody or micro-immunofluorescence (MIF). Cases were detected by the National Notifiable Disease Surveillance System and the government of Matsue City. Laboratory confirmation by CF was defined as a ⩾fourfold change between two serum specimens obtained ⩾2 weeks apart, or a single titre ⩾32-fold. Laboratory confirmation by MIF was performed at the Laboratory of Rickettsia and Chlamydia Department of Virology I, National Institute of Infectious Diseases (R&C, NIID) and defined as follows: (1) a ⩾fourfold change in IgG or IgA between two serum specimens obtained ⩾2 weeks apart, (2) a single IgG titre ⩾512, or (3) a single IgM titre ⩾32.
We conducted a field investigation at Bird Park X in late January and early February 2002. We interviewed each case regarding the illness and exposure to birds and collected climate data and environmental samples of bird faeces, soil and water for detection of the C. psittaci gene by polymerase chain reaction (PCR). A primer pair designed to generate a Chlamydia spp.-specific region of the major outer membrane protein (MOMP) gene was used to amplify a single DNA fragment of 245–259 bp, and we confirmed C. psittaci by restriction endonuclease analysis with AluI and PvuII [Reference Yoshida4].
Serum samples from each park staff member were collected twice for antibody testing during mid and late January 2002. After receiving informed consent, antibodies for C. psittaci, Chlamydophila pneumoniae and Chlamydia trachomatis were tested for these samples using MIF for acute serologically confirmed psittacosis (SCP) infection. Cases with suspected cross-reactions with C. pneumoniae or C. trachomatis were excluded.
A questionnaire survey was conducted for staff and students regarding bird exposures, influenza-like illness after employment at Bird Park X, and prior knowledge about psittacosis. A retrospective cohort study was conducted to evaluate risk factors for SCP infection. Relative risks (RR) with 95% confidence intervals (CI) were calculated using Epi-Info, version 6 (CDC, Atlanta, GA, USA).
Seventeen cases were detected (14 female; median age 54 years, age range 20–87). There were 12 visitor, three staff and two student cases. Fourteen cases were confirmed by MIF. All 17 cases had fever and 16 had radiographic evidence of pneumonia. Symptoms included general fatigue (n=11), cough (n=8), headache (n=5), myalgia (n=4), and vomiting (n=4). No severe complications occurred. All 17, including 13 hospitalized cases, recovered with antibiotic treatment (e.g. tetracyclines). Duration of hospitalization was 5–45 days (median 15 days).
The onset dates among the visitor cases were from 16 November 2001 to 9 January 2002 (Fig.). Suspected median incubation period for the 12 visitor cases was 12 days (range 9–25 days). All 12 visitor cases had visited all the hothouses. Seven of these cases had handled birds in the park and five had fed birds. Only two cases visited the park in the afternoon. Eight visitors were exposed on 14 or 15 December 2001, despite the fact that there were fewer visitors (mean visitors about 1600 per day) on 14 December (n=174) and 15 December (n=464). These days were the coldest and windiest days since the park's opening; the air temperature was <5°C and the mean wind velocity was >12 m/s.

Fig. Epidemic curve of Bird Park X-related psittacosis cases, Shimane, 2001–2002 (n=17). □, Visitor; ![]() , student; ■, staff.
, student; ■, staff.
The onset dates among staff and students were from 8 to 20 December 2001 (Fig.). All three staff cases had worked with birds as ‘bird staff’. Two student cases started their work in mid-November or early December 2001; they had no evident contact with birds prior to employment. Bird Park X staff suspected the source of infection might be sick birds imported on 30 October 2001 from Bird Park Y in Chiba Prefecture. The imported birds were displayed in open glass enclosures with other non-imported birds. None of the imported birds had been quarantined.
The air temperature in T, P and the backyard was maintained between 20°C and 25°C with an air circulation system. During the winter, the hothouses operated as a completely closed circulation system. Late in November 2001, bird staff began using dehumidifiers in T and the backyard, and started using a high water-pressure jet spray for cleaning cages in the backyard. This washer was used during the park's morning business hours, and the area was not ventilated during this time.
Ill birds with mild symptoms were housed in the backyard, and in cages on the first floor of the staff building, where staff prepared feed for the birds. On the second floor of this building, where staff took short breaks, there was an isolation room for severely ill birds. The isolation room was not ventilated. Staff entered this building, including the isolation room, without any respiratory protection. A veterinarian had never been employed, and no records were kept regarding ill birds. The risk of C. psittaci in secretions had never been discussed with employees prior to this outbreak.
Between late January and early February 2002, 125 faecal samples were collected from the park's cages. Eleven samples (eight samples from T and three from the backyard) tested positive using PCR. These positive samples were collected from both cages containing birds imported from Bird Park Y and those containing no imported birds. Environmental samples (n=31) of soil, water and samples of air conditioner filter tested negative.
Eight of 91 staff members (who were not psittacosis cases) had SCP infection based on MIF. Two of the eight had an influenza-like illness in early December 2001 and early January 2002. The other six exhibited no symptoms. Eighty-one staff members, including the eight infected, responded to our questionnaire. Reported psittacosis cases (three staff and one student) also responded to the questionnaire. Only five (38%) of the bird staff and three (4%) of other staff members knew about psittacosis and the transmission risks before this outbreak.
In univariate analysis, gender, bird-handling in the park, being bitten by birds in the park or keeping of pet birds were not associated with SCP infection. However, relative to park staff who did not handle birds, being a bird staff member had a higher risk for SCP infection (RR 3·96, 95% CI 1·48–10·58). The association between SCP infection and entry into a particular building was also evaluated in univariate analysis during two periods, pre- and post-importation of birds from Bird Park Y on 30 October 2001 (Table). In the post-importation period, entering the staff building was associated with an almost fourfold increase in the risk of SCP infection, relative to not entering (RR 3·61, 95% CI 1·03–12·60).
Table. Relative risk and 95% confidence interval of developing acute serological infection among staff who entered a particular building compared to staff who did not
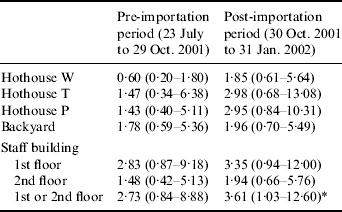
* Indicates statistical significance.
In Japan, most reported psittacosis cases before 2001 were sporadic, except for an occupational outbreak from a Siberian moose at a zoo in 2001 [2]. In the United States, a psittacosis outbreak from a shipment of pet birds affected 46 persons [Reference Moroney5]. An outbreak related to a duck farm and processing plant has been reported in Australia [Reference Hinton6]. However, outbreaks related to bird parks have not been commonly reported. To our knowledge this is the largest psittacosis outbreak reported in Japan. During the winter, the hothouses were operating under a predominately closed air circulation system. This might have raised the concentration of the pathogen, increasing the possibility of transmission. Dehumidifiers or the jet washers might have dispersed the pathogen in the closed hothouses. Ten out of 12 visitor cases were exposed in the morning, when jet washers were in use.
There were severe deficiencies in the isolation of ill birds and basic precautionary practices to prevent transmission. Employees entered areas with ill birds without any respiratory protection. It is possible that the number of cases might be underestimated among visitors, because only severe cases tend to be diagnosed. Indeed, we detected SCP cases with mild or no symptoms among the staff. Bird Park X staff suspected the source of infection might have been imported birds. While we were unable to find the definitive source of infection, entering the staff building during the post-importation period was associated with a significantly increased risk of seroconversion. However, the post-importation period was also the time of the coldest weather. We do not know whether the building's closed circulation during this period and/or the importation of birds contributed more to the outbreak.
There are important limitations in this study. First, we were unable to determine the time at which the birds became infected. In addition, distinguishing psittacosis and C. pneumonia infection is sometimes difficult using serological tests because of cross-reactions, even with MIF [Reference Telfer7]. In addition, we could not perform PCR on any human samples; we diagnosed our visitor cases as psittacosis based on symptoms, serology, and exposure to Bird Park X.
To lessen the risk of psittacosis outbreaks, we recommended adequate quarantine and isolation of birds. There should also be proper treatment of ill birds and an open circulation system in hothouses. If hothouses need to operate as a closed system, they should be fully partitioned from the viewing area with an independent ventilation system. Staff and visitors should be informed about psittacosis and its modes of transmission. We further recommend syndromic surveillance for both staff and birds in bird parks. Last, while the National Notifiable Disease Surveillance System was useful in detecting this outbreak, final confirmation of psittacosis infection by MIF was mostly limited to the R&C, NIID. The availability of current laboratory diagnostic methods for psittacosis should be improved.
Until recently, bird parks were not legally managed or regulated to prevent zoonotic diseases in Japan. After two psittacosis outbreaks in 2001, recommendations on preventing zoonoses at animal-related facilities were published in 2003, and government officials are now able to investigate or intervene at the implicated facilities according to legal guidelines.
DECLARATION OF INTEREST
None.




