Crossref Citations
This article has been cited by the following publications. This list is generated based on data provided by
Crossref.
Vidal, Olivier
Lanari, Pierre
Munoz, Manuel
Bourdelle, Franck
and
De Andrade, Vincent
2016.
Deciphering temperature, pressure and oxygen-activity conditions of chlorite formation.
Clay Minerals,
Vol. 51,
Issue. 4,
p.
615.
Berger, Alfons
Wehrens, Philip
Lanari, Pierre
Zwingmann, Horst
and
Herwegh, Marco
2017.
Microstructures, mineral chemistry and geochronology of white micas along a retrograde evolution: An example from the Aar massif (Central Alps, Switzerland).
Tectonophysics,
Vol. 721,
Issue. ,
p.
179.
Süssenberger, Annette
Pospiech, Solveig
and
Schmidt, Susanne Theodora
2018.
[MnO|SiO2,Al2O3,FeO,MgO] balanced log-ratio in chlorites: a tool for chemo-stratigraphic mapping and proxy for the depositional environment.
Clay Minerals,
Vol. 53,
Issue. 3,
p.
351.
Lempart, Małgorzata
Derkowski, Arkadiusz
Luberda-Durnaś, Katarzyna
Skiba, Michał
and
Błachowski, Artur
2018.
Dehydrogenation and dehydroxylation as drivers of the thermal decomposition of Fe-chlorites.
American Mineralogist,
Vol. 103,
Issue. 11,
p.
1837.
Masci, Lorella
Dubacq, Benoît
Verlaguet, Anne
Chopin, Christian
De Andrade, Vincent
and
Herviou, Clément
2019.
A XANES and EPMA study of Fe3+ in chlorite: Importance of oxychlorite and implications for cation site distribution and thermobarometry.
American Mineralogist,
Vol. 104,
Issue. 3,
p.
403.
Verdecchia, Sebastián O.
Collo, Gilda
Zandomeni, Priscila S.
Wunderlin, Cecilia
and
Fehrmann, Mariano
2019.
Crystallochemical indexes and geothermobarometric calculations as a multiproxy approach to P-T condition of the low-grade metamorphism: The case of the San Luis Formation, Eastern Sierras Pampeanas of Argentina.
Lithos,
Vol. 324-325,
Issue. ,
p.
385.
Ilalova, R. K.
and
Gulbin, Yu. L.
2019.
Thermometry of Nickel Bearing Chlorites from the Kolskii Massif (Northern Urals).
Geology of Ore Deposits,
Vol. 61,
Issue. 8,
p.
736.
Lanari, Pierre
and
Duesterhoeft, Erik
2019.
Modeling Metamorphic Rocks Using Equilibrium Thermodynamics and Internally Consistent Databases: Past Achievements, Problems and Perspectives.
Journal of Petrology,
Vol. 60,
Issue. 1,
p.
19.
Moëlo, Yves
Fritsch, Emmanuel
Gloaguen, Eric
and
Rouer, Olivier
2020.
Polygenic chamosite from a hydrothermalized oolitic ironstone (Saint-Aubin-des-Châteaux, Armorican Massif, France): crystal chemistry, visible–near-infrared spectroscopy (red variety) and geochemical significance.
Clay Minerals,
Vol. 55,
Issue. 1,
p.
83.
Di Rosa, Maria
Meneghini, Francesca
Marroni, Michele
Frassi, Chiara
and
Pandolfi, Luca
2020.
The coupling of high-pressure oceanic and continental units in Alpine Corsica: Evidence for syn-exhumation tectonic erosion at the roof of the plate interface.
Lithos,
Vol. 354-355,
Issue. ,
p.
105328.
Bourdelle, Franck
2021.
Low-Temperature Chlorite Geothermometry and Related Recent Analytical Advances: A Review.
Minerals,
Vol. 11,
Issue. 2,
p.
130.
Gisbert, Guillem
Tornos, Fernando
Losantos, Emma
McClenaghan, Sean
Pons, Juan Manuel
Videira, Juan Carlos
and
Brodbeck, Maurice
2022.
Vectors to ore in replacive volcanogenic massive sulphide deposits of the northern Iberian Pyrite Belt: Major and trace element mineral chemistry.
Ore Geology Reviews,
Vol. 147,
Issue. ,
p.
104963.
Li, Changhao
Shen, Ping
Zhao, Yong
Li, Pei
Zhang, Lejun
and
Pan, Hongdi
2022.
Mineral chemistry of chlorite in different geologic environments and its implications for porphyry Cu ± Au ± Mo deposits.
Ore Geology Reviews,
Vol. 149,
Issue. ,
p.
105112.
Mohanty, Sarada P.
and
Mishra, Prasanta K.
2023.
Greenalite-Chamosite composition, geothermometry and oxygen fugacity variations in pisolitic ironstone and carbonates of the Chilpi Group: implication on Paleoproterozoic seawater chemistry.
Physics and Chemistry of Minerals,
Vol. 50,
Issue. 4,
Lamm, Sarah N.
Lacroix, Brice
Marshall, C. P.
Lahfid, Abdeltif
Gasda, Patrick J.
Kempton, Pamela D.
Trincal, Vincent
Buatier, Martine
Lanari, Pierre
and
Vennemann, Torsten
2023.
Empirical Raman calibration of trioctahedral ferromagnesium chlorite minerals.
Journal of Raman Spectroscopy,
Vol. 54,
Issue. 12,
p.
1423.
Mota-Heredia, Carlos
Cuevas, Jaime
and
Fernández, Raúl
2024.
Geochemical Evolution of Mg-Bentonite Affected by the Contact of Carbon Steel and a Hydrothermal Gradient.
Applied Sciences,
Vol. 14,
Issue. 3,
p.
1259.
Dubacq, Benoît
and
Forshaw, Jacob B.
2024.
The composition of metapelitic biotite, white mica, and chlorite: a review with implications for solid-solution models.
European Journal of Mineralogy,
Vol. 36,
Issue. 4,
p.
657.
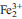 □Al]O10(OH)8 within the chlorite solid-solution domain. According to our observations, up to two Fe3+ cations might be allocated in the M2-M3 chlorite sites by the substitution AFDT, which does not involve Al. These unexpected observations were made possible by the development of μXANES techniques allowing in situ measurements of XFe3+ (Fe3+/(Fe2+ + Fe3+)) in heterogeneous chlorite. Although further studies are required to confirm the crystallographic position of Fe3+ and refine its ionic/ magnetic behaviour in chlorite, this development creates opportunities for developing new geothermometers.
□Al]O10(OH)8 within the chlorite solid-solution domain. According to our observations, up to two Fe3+ cations might be allocated in the M2-M3 chlorite sites by the substitution AFDT, which does not involve Al. These unexpected observations were made possible by the development of μXANES techniques allowing in situ measurements of XFe3+ (Fe3+/(Fe2+ + Fe3+)) in heterogeneous chlorite. Although further studies are required to confirm the crystallographic position of Fe3+ and refine its ionic/ magnetic behaviour in chlorite, this development creates opportunities for developing new geothermometers.

