Sleep and mental health are inextricably linked, with associations between abnormal sleep and depression,Reference Riemann, Berger and Voderholzer1 bipolar disorder,Reference Harvey2 schizophrenia,Reference Sarkar, Katshu, Nizamie and Praharaj3 autism spectrum disorders,Reference Cohen, Conduit, Lockley, Rajaratnam and Cornish4 attention-deficit hyperactivity disorder (ADHD)Reference Owens5 and relapse rates in addiction.Reference Brower and Perron6 Adequate duration and timing of sleep are essential for maintaining good physical and mental health.Reference Gopalakrishnan, Ji and Cirelli7–Reference Soldatos9 Sleep disturbance has been associated with a two- to threefold increase in risk of psychiatric in-patient admissionReference Walker, Talavera, Nomamiukor, Madubata, Alfano and Vujanovic10 and of suicide.Reference Ribeiro, Pease, Gutierrez, Silva, Bernert and Rudd11 The psychiatric in-patient setting in itself can be detrimental to sleep quality.Reference Doǧan, Ertekin and Doǧan12 As many as two-thirds of in-patients on psychiatric wards experience insomniaReference Talih, Ajaltouni, Ghandour, Abu-Mohammad and Kobeissy13 and poor sleep is a predictor for psychiatric readmission.Reference Koffel, Thuras, Chakravorty, Germain and Khawaja14
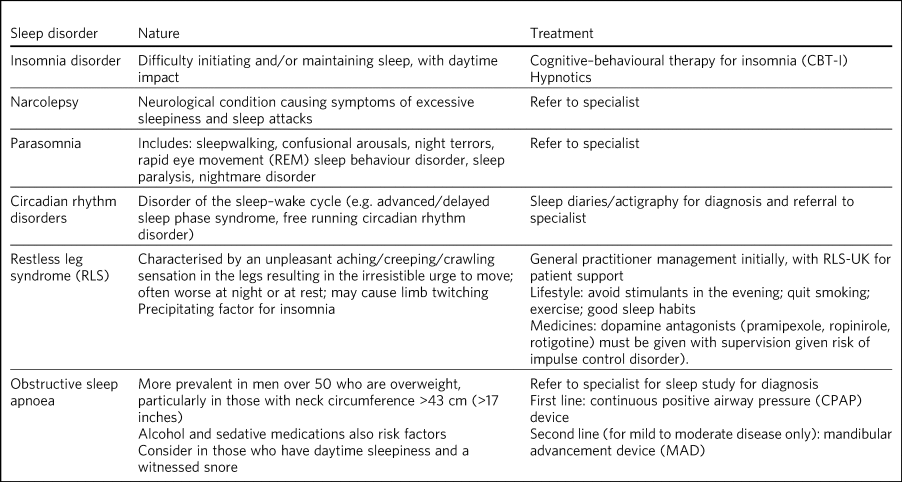
If left untreated, sleep disorders (Table 1) such as insomnia, narcolepsy, circadian rhythm disorders, parasomnias and restless legs syndrome can have deleterious effects on cardiovascular, metabolic and mental health, and some are linked to an increase in mortality.Reference Nedeltcheva and Scheer15–Reference Partinen, Jamieson and Guilleminault17 ‘Poor sleep’ is not in its own right a diagnosis.Reference Thorpy and Chokroverty18 This evidence–practice gap contributes to delays in diagnosis and treatment,Reference Perry, Patil and Presley-Cantrell19 an overprescribing of hypnotic medication,Reference Janhsen, Roser and Hoffmann20 has a negative impact on recovery from psychiatric disorders and can cause misattribution of symptoms.Reference Ford and Kamerow21 Teaching on distinct sleep disorders and effective behavioural interventions for insomnia is generally lacking in UK undergraduate medical curricula.Reference Romiszewski, May, Homan, Norris, Miller and Zeman22 In practice, an increase in prescribing rates, mortality and drug spend indicates a culture of inappropriate prescribing, suggesting that there is also a paucity of training about sleep disorders at graduate level.Reference O'Regan23
Table 1 Overview of specific sleep disorders
Hypnotics are widely prescribed for adult psychiatric in-patients suffering from poor sleep.Reference Hallahan, Murray and McDonald24 They can be effective and necessary for acute insomnia,Reference Guina and Merrill25 but if insomnia persists the benefits of hypnotics may wane while the risk of side-effects increases.Reference Ancoli-Israel, Richardson, Mangano, Jenkins, Hall and Jones26 Hypnotics have the potential to worsen outcomes for sleep apnoea, anxiety, cognitive function and depression,Reference Li, Bai, Lee, Mao, Chen and Tu27 and pose a significant risk of dependence and harm with long-term use.Reference Baldwin, Aitchison, Bateson, Curran, Davies and Leonard28 Hypnotics are commonly overprescribed:Reference Weymann, Gladstone, Smolina and Morgan29 a 2015 survey of English general practice populations described inappropriate hypnotic prescribing as a public health problem, estimating that over a quarter of a million people take hypnotic medication beyond licensed timescales.Reference Davies, Rae and Montagu30
Hypnotic medication should only be considered after appropriate assessment and after, or in combination with, psychological therapies such as cognitive–behavioural therapy for insomnia (CBTi).Reference Wilson, Anderson, Baldwin, Dijk, Espie and Espie31 The deprescribing (cessation of inappropriate medication)Reference Reeve, Gnjidic, Long and Hilmer32 of hypnotic medication is considered problematic.Reference Ng, Le Couteur and Hilmer33 A lack of institutional structures, inadequate resources, and the attitudes and practice of more senior clinicians have been cited as barriers to this process.Reference Kuntz, Kouch, Christian, Peterson and Gruss34
At present, compliance with evidence-based care in sleep management is suboptimal.Reference Guina and Merrill35 Increasingly, pharmacological agents are chosen over non-pharmacological treatments.Reference Moloney36 Disturbances to sleep are multifactorial in nature.Reference Stores37 Although causal components can be tied to the patient's psychiatric diagnosis, underlying physical health condition or a distinct sleep disorder, there is a need for improved resourcing and greater awareness of assessment and treatment strategies to aid practitioners in managing insomniaReference Sirdifield, Anthierens, Creupelandt, Chipchase, Christiaens and Siriwardena38 and, more broadly, improving sleep on in-patient units. Barriers to implementation include: workload/time pressures, a lack of resources, a lack of authority to change practice, a workplace culture resistant to change and a lack of support from other staff members.Reference Wallis39 There are modifiable behavioural factors that can markedly improve sleep.Reference Brower40 In in-patient settings, staff-related factors include reducing ward noise and overnight nursing observations, optimising the patient's bedroom for sleep and increasing staff awareness of sleep management.Reference Horne, Hay, Watson and Anderson41 Patient-related factors include reducing caffeine, alcohol and nicotine consumption,Reference Garcia and Salloum42 setting a routine wake up time, introducing a wind-down period prior to attempting sleep and avoiding prolonged periods in bed while awake.Reference Christodulu and Durand43 Strategic factors include educational meetings, printed educational materials, local opinion leaders, revising professional roles and practice facilitation. Multifaceted, tailored strategies may be more effective at creating sustained change in practice.Reference Lau, Stevenson, Ong, Dziedzic, Treweek and Eldridge44
This study examines whether improving awareness of sleep, insomnia and behavioural sleep interventions among clinicians supports the deprescribing of hypnotics. The impact of a simple, online educational package on the prescribing patterns on in-patient mental health units was evaluated.
Method
Design
A co-design approachReference Jessup, Osborne, Buchbinder and Beauchamp45 was employed to develop a package of educational interventions designed to be generalisable to practitioners across all care settings. The package, part of the Smarter Sleep educational materials (available from https://www.cntw.nhs.uk/resource-library/smarter-sleep/), comprised
• an overview video: a 9 min overview of normal sleep, specific sleep disorders, hypnotics and psychological therapies for insomnia
• a poster: to prompt nursing staff towards simple sleep interventions instead of hypnotic administration
• a handbook: a detailed review of sleep, causes of sleep disturbance and basic principles of CBTi.
These interventions were introduced to wards, and emails were sent each month as reminders that the interventions were active. Acute and rehabilitation wards for both male and female patients on one hospital site were eligible to participate. Wards on other sites within the trust (other than the control ward) were excluded.
Development of the intervention
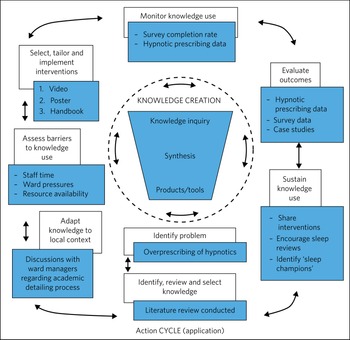
The development, embedding and evaluation of these interventions can be mapped to the knowledge-to-action (KTA) frameworkReference Graham, Logan, Harrison, Straus, Tetroe and Caswell46 (Fig. 1). This framework involves two phases – knowledge creation and the action cycle – which are interlinked and feed dynamically between each other.

Fig. 1 The knowledge-to-action (KTA) framework (based on Graham et alReference Graham, Logan, Harrison, Straus, Tetroe and Caswell46).
Knowledge creation
Represented by a ‘knowledge funnel’, this consists of knowledge inquiry, synthesis and products/tools. This process encapsulates primary literature (knowledge inquiry) and secondary literature (synthesis), which are condensed down to produce ‘third-generation knowledge’ in the form of products/tools (decision aids, guidelines and pathways).
The action cycle
This part of the KTA framework represents the application of the knowledge creation process. It involves identifying the problem (the overprescribing of hypnotics) and then selecting the knowledge required for the intervention. We did this through literature review.Reference Richardson, Paterson, Rathbone, Blagburn and Husband47,Reference Paterson, Richardson, Rathbone, Blagburn and Husband48 Knowledge was then adapted to the local context through discussions with ward managers, and barriers to knowledge use were identified (time, ward pressures, resource availability). The interventions were tailored and implemented in the form of a video, poster and handbook, and knowledge use was monitored through the survey completion data and hypnotic prescribing rate data. Outcomes were evaluated using these data and case studies from implementation of the interventions. Knowledge use is being sustained through the embedding of interventions into everyday practice, and the hosting of the resources on the trust website (https://www.cntw.nhs.uk/smartersleep).
Hypnotic prescribing and administration rates
A controlled before-and-after approach was taken to assess the impact of the interventions. Point prevalence data were collected via retrospective drug chart analysis over 2 full months (September 2019 and December 2019), with 2 months between for interventions to become embedded. These data consisted of a count of the number of active hypnotic prescriptions written on the drug charts of each ward during these periods. Data were also collected on the number of hypnotic administrations (i.e. individual doses) that took place on each pilot ward over this same periods. All doses in these time periods were counted, even if a patient was prescribed multiple hypnotic agents. These outcomes were relevant in evaluating the impact of the interventions on practice.
For example, seven doses administered against a prescription of ‘zopiclone 7.5 mg at night when required’ would count as one active prescription and seven total administrations. Data were analysed using descriptive statistics.
Pre- and post-intervention survey
The survey was undertaken entirely online. A pre-intervention survey was emailed to all participants prior to watching the video, along with a link to the video and electronic copies of the resources. This was followed by a post-intervention survey 10–12 weeks later (Table 2). The survey assessed baseline knowledge and attitudes towards current sleep management (pre-intervention survey) and then measured any changes that had taken place in the knowledge and attitudes of individuals over the intervention period (post-intervention survey). These outcomes were relevant in assessing whether the interventions had an impact on staff knowledge and behaviours, even if this impact was not enough to change prescribing and administration rates on the wards.
Table 2 Comparison of the questions in the pre- and post-intervention surveys

Participants
The package of educational interventions was designed by a stakeholder group consisting of hospital pharmacists (n = 8), primary care pharmacists (n = 4), a neurologist with specialist interest in sleep (n = 1) and consultant psychiatrists (n = 3). Stakeholders were recruited through local networks and selected for their knowledge and experience in insomnia, hypnotics or local practice. A patient and public involvement group was also consulted to consider which messages they would want to ensure were communicated to healthcare staff caring for them. Extensive consultation in the form of draft videos and resources shared via email ensured that these interventions would be generalisable across all care settings. A draft video was also shown at a regional pharmacy conference and feedback was collected on the content and quality, to be integrated into the final version.
The resulting interventions were introduced on in-patient wards (n = 7) in one hospital and compared against a control ward. A geographically distant hospital within the same trust was selected for the control ward, to mitigate the risk of migration of practice if staff cross-covered pilot and control wards.
Ethics
All participants gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Newcastle University Faculty of Medical Sciences. The project was also registered with the research department at Cumbria, Northumberland, Tyne and Wear NHS Foundation Trust.
Standards
The SQUIRE statement was used as a scaffold in the writing of this paper.Reference Pinnock, Barwick, Carpenter, Eldridge, Grandes and Griffiths49
All wards received the same level of resources for implementation of the intervention. Variation in levels of staff engagement with the interventions may have led to variation between wards, but the interventions themselves were standardised across all pilot wards. The control (comparator) ward had no interventions delivered, but staff were informed that data would be collected to allow for comparison, and interventions made available after the evaluation phase had been completed.
Results
Hypnotic prescribing and administration rates
A decrease in the number of administrations occurred on all wards except one, which was a psychiatric intensive care unit (PICU) treating patients with more challenging behaviours (Fig. 2). Overall, there was a mean reduction of 41% in the number of hypnotic administrations per patient across the pilot site. This compares with the control site, which showed a 37% increase in the administrations of hypnotics per patient on the control ward (Table 3).
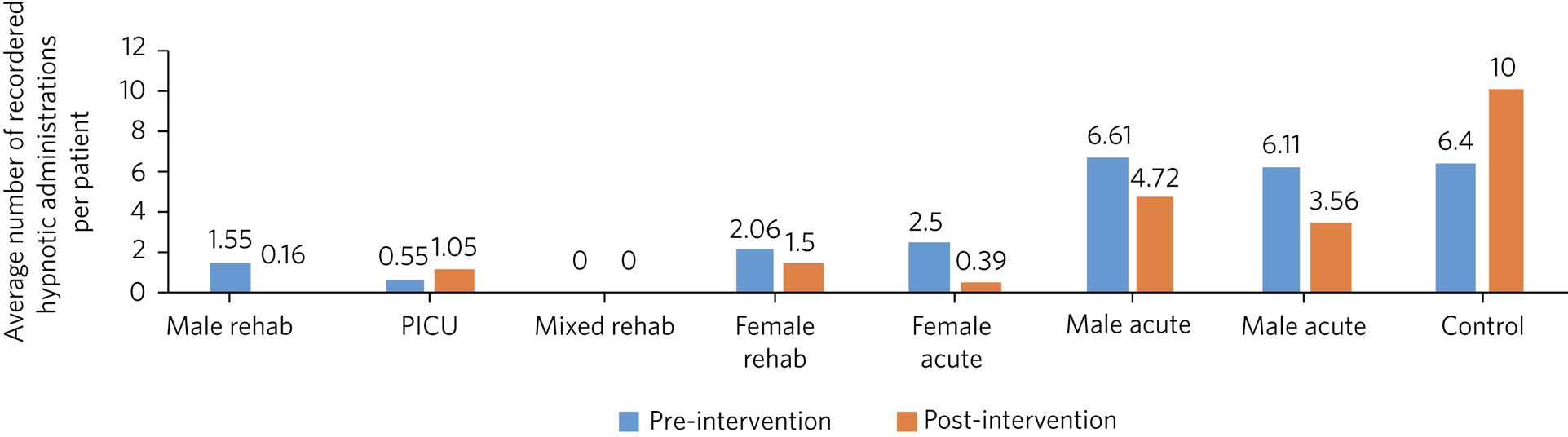
Fig. 2 Average number of recorded administrations of hypnotics per patient pre- and post-educational interventions across eight mental health in-patient wards. Rehab, rehabilitation; PICU, psychiatric intensive care unit.
Table 3 Patients on hypnotics and administration rate per patient pre- and post-educational intervention to reduce hypnotic prescribing
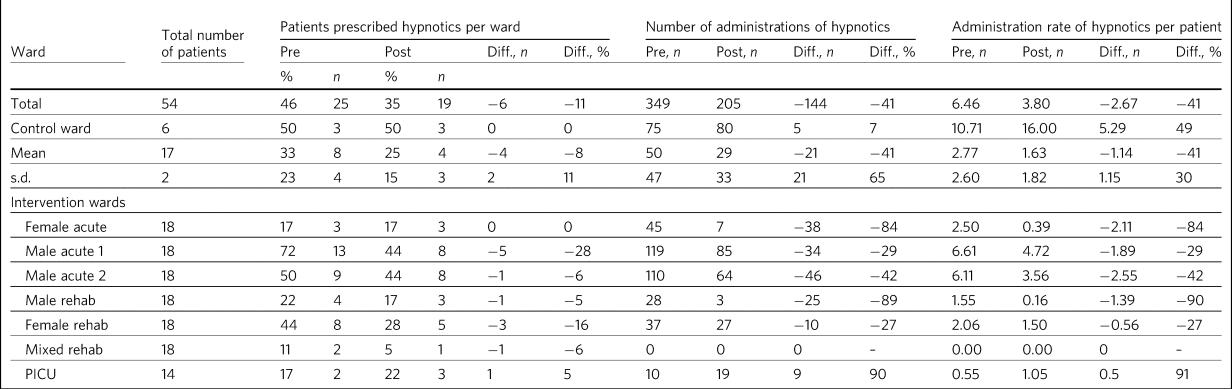
Diff., difference; PICU, psychiatric intensive care unit.
A decrease in the percentage of patients prescribed hypnotics occurred on 71% of the pilot wards (Fig. 3). The PICU and female acute wards did not show a decrease, however. Overall, there was a mean reduction of 24% in the percentage of patients prescribed hypnotics on the pilot site. This compares with the control ward (a mixed-gender rehabilitation ward), which showed no change.
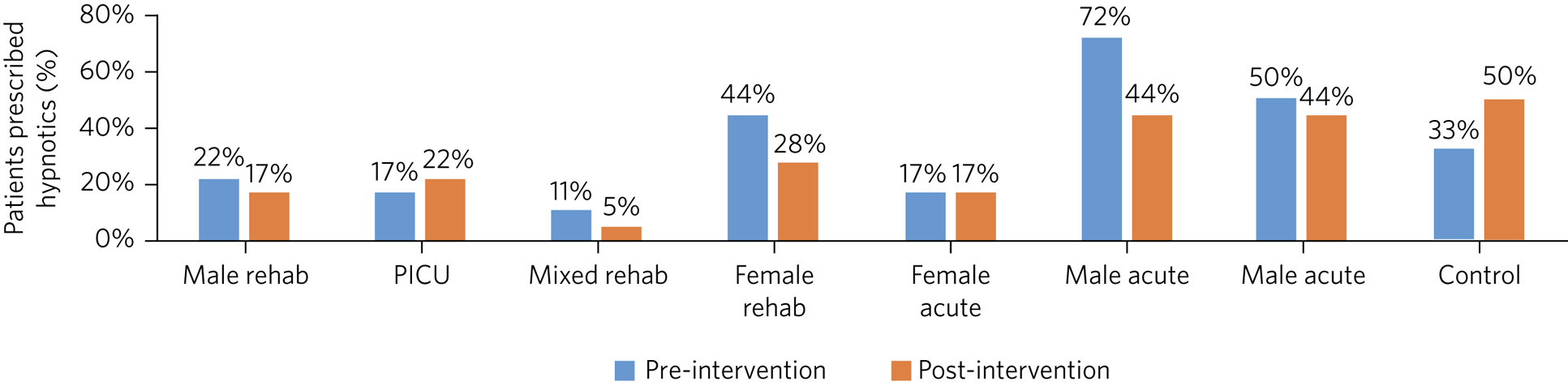
Fig. 3 Percentage of patients prescribed hypnotics per ward pre- and post-educational interventions across eight mental health in-patients wards. Rehab, rehabilitation; PICU, psychiatric intensive care unit.
Pre- and post-intervention survey
A total of 251 staff were invited to participate in the survey. Non-participation reasons included a lack of time, leave of absence and self-perceived adequate pre-existing knowledge in this subject area. Survey response data were collected on the pre-intervention questionnaire (n = 85) and post-intervention questionnaire (n = 45) (total n = 131). Total respondents for both surveys comprised: nurses (pre-intervention n = 27; post-intervention n = 13); junior doctors (pre, 7; post, 1); specialty trainees (pre, 6; post, 3); consultants (pre, 3; post, 2); pharmacists (pre, 12; post, 12); pharmacy technicians (pre, 6; post, 4); psychologists (pre, 7; post, 2); and ‘other’ (pre, 17; post, 8). Two respondents did not complete the question about job role.
Before and after the intervention, respondents were asked to rate their confidence (on a Likert scale from 1, ‘not confident’ to 5, ‘extremely confident’) in using medication versus psychological strategies to help patients sleep. An increase was seen in confidence scores for using medication and for using psychological strategies to help patients sleep (Table 4).
Table 4 Self-rated confidence scoresa before and after educational intervention
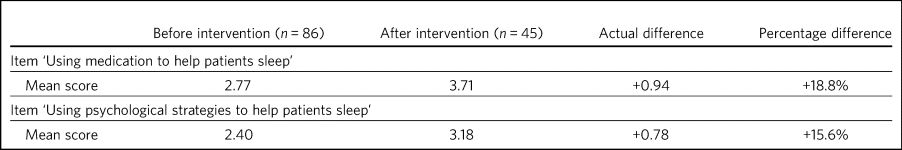
a. Rated on a scale of 0–5, where 0 indicates no confidence and 5 indicates very confident.
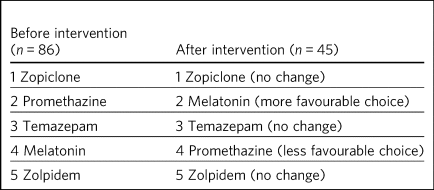
The intervention prompted a change in the order in which hypnotics were considered (Table 5) for sleep management. Melatonin rose in ranking, whereas promethazine fell.
Table 5 Average rank order of hypnotics for prescribing, first choice to last choice, before and after educational intervention
When asked pre-intervention to comment on their rationale behind the ranking decisions they had made, 87% of respondents mentioned that their ranking of the listed medications was based on what they had ‘seen in practice’ or ‘heard of the most’, i.e. choices were being informed by existing practice on the wards that staff worked. Only 9% of respondents mentioned anything about knowledge of drugs in their response, and no respondents made reference to the evidence base, cost-effectiveness or risk–benefit. For example,
‘I picked the medication that I had heard of used within the service I work. Patients are more frequently prescribed promethazine and zopiclone, promethazine is usually offered first. Melatonin I believe is non formulary so is used less regularly. Temazepam is used rarely. I have never heard of zolpidem before.’ (Foundation Year 1 doctor)
Asked the same question post-intervention, only 13% of respondents still mentioned in their responses that their choices were being informed by experience of practice, whereas 76% mentioned the evidence base, cost-effectiveness and/or risk–benefit in their responses. For example,
‘Melatonin is licensed in my client group for insomnia. Evidence base supports the use of Z-drugs and benzodiazepines for short term use in insomnia and Z-drugs have a slightly more favourable side-effect profile in terms of long-term dependency. There is little evidence to support the use of sedating antihistamines for insomnia.’ (Advanced Pharmacist Practitioner)
Discussion
Hypnotic prescribing and administration rates
Among staff working on the in-patient units studied, knowledge about the mechanism of action of hypnotic medications and different sleep disorders was limited and informed by previous practice, not the evidence base. After a short educational video about sleep disorders and hypnotics, the hypnotic prescribing rates decreased by 24% and the administration rates fell by 41% compared with the control ward. There was also a difference in which medications were used as hypnotics, with promethazine use being reduced. Staff described a better understanding of sleep and the different available therapies for sleep disorders after the intervention.
An increase in hypnotics issued was seen on the PICU despite the implementation of the educational interventions (Table 2). This may be due to the nature of the patients on this ward, who are more likely to have challenging behaviours and disturbed sleep and to not engage as much with psychological therapies compared with patients admitted to acute and rehabilitation wards.
Pre- and post-intervention survey
Self-rated confidence scores show a larger improvement in the mean score in relation to medication than in the mean score for psychological strategies for improving sleep. Further support and training may be needed to help staff feel confident in psychological management. Alternatively, this might suggest that, owing to the often acute nature of the in-patient setting, hypnotics are deemed to be of greater utility than psychological strategies in the care of patients struggling with sleep and/or anxiety.
The table ranking the order in which hypnotics were considered (Table 5) shows convergence of practice towards the evidence base. Zolpidem is non-formulary in this geographical area, which is likely to be the reason for it being lowest ranked. Promethazine has fallen in ranking, which is likely to be because the educational package highlighted its weak evidence base and high side-effect profile. Melatonin rose in ranking, possibly due to its favourable side-effect profile and the fact that it is an endogenous hormone involved in regulation of the circadian rhythm. It does, however, have a short half-life and so its use in sleep disorders may be limited,Reference Quera-Salva and Claustrat50 as was highlighted in the educational package.
An increase in the evidence-based language used when rationalising the rank order of hypnotics suggests that this intervention went some way towards closing the evidence–practice gap in management of poor sleep, as end-user feedback illustrates.
End-user feedback
Doctors found these resources useful for instigating positive change in relation to hypnotic prescribing, reviewing the clinical need and reducing doses where appropriate:
‘Patients were on hypnotics for weeks and weeks and weeks and then stopped immediately at discharge. This is bad practice, and leaves a mess for both GPs [general practitioners] and the patients. These interventions created a change in culture so that the mindset for patients prescribed hypnotics is now to reduce them during the in-patient stay wherever appropriate.’ (Specialty trainee)
Nursing staff found these resources helpful in offering alternative solutions to sleep disturbances, other than administering hypnotic medication:
‘I feel like, after watching the video, we started to encourage patients to use non-pharmacological methods more. This demonstrated that patients were often showing drug seeking behaviours, as they would often then go to sleep without needing hypnotics.’ (Ward manager)
The video was viewed favourably by staff and shone a light on a field of practice that is often overlooked:
‘Excellent video, extremely important field that is greatly underestimated, but is pivotal in a patient's physical, mental and emotional wellbeing.’ (Consultant)
Generalisability
The presence of the pharmacy team on all wards enabled the proliferation of the intervention among other staff groups. Rotation of nursing staff between wards enabled migration of practice to spread engagement with the interventions across wards. Some wards (n = 2) were involved in the SleepWell Project,Reference Novak, Packer, Paterson, Roshi, Locke and Keown51 which may have motivated staff to engage with further work on sleep, but the impact of SleepWell in relation to this study is unclear. The above aspects may not exist in other settings.
Limitations
There were several limitations to the study. No data were gathered on which of the interventions was felt to be the most effective, as the intervention package was conceptualised as a whole. The evaluation period was short, owing to the limited time span available within the context of the funding. There were fewer responses to the post-intervention survey than to the pre-intervention survey, as pre-intervention responses were gathered immediately before accessing the intervention. Post-intervention responses were gathered 10–12 weeks later, to allow time for a change in practice to occur. A shorter interval may have resulted in a lower attrition rate, but seen a smaller change in prescribing and administration rates. Completing the post-intervention survey straight after the resources were accessed may have increased responses but would have allowed less time for reflection. Introducing a prize raffle for completion of the post-intervention survey may also have increased responses.
The control ward was selected because of its geographical distance from the intervention wards to prevent migration of practice. During the course of this study the ward was planned for closure, so admitted fewer and fewer patients, leading to a lower number of patients in this group.
Future work
Future work could examine the nuances of uptake and benefit between interventions. It might also be valuable to evaluate whether the reduction in inappropriate hypnotic prescriptions is sustained over time. If it is not sustained, measuring how long this change lasts and investigating barriers and enablers for maintaining a change in evidence-based practice would be worthy of future investigation. Further work could also look at the feasibility and uptake of these interventions in primary care and examine barriers and enablers to the delivery of online educational interventions in this setting.
About the authors
Alastair Paterson, Pharmacy Department, Cumbria, Northumberland, Tyne and Wear NHS Foundation Trust; and School of Pharmacy, Faculty of Medical Sciences, Newcastle University, Newcastle upon Tyne, UK. Martina Khundakar, Pharmacy Department, Cumbria, Northumberland, Tyne and Wear NHS Foundation Trust; and School of Pharmacy, Faculty of Medical Sciences, Newcastle University, Newcastle upon Tyne, UK. Anthony Young, Pharmacy Department, Cumbria, Northumberland, Tyne and Wear NHS Foundation Trust, Newcastle upon Tyne; and School of Pharmacy, Faculty of Health Sciences and Wellbeing, University of Sunderland, UK. Jonathan Ling, Faculty of Health Sciences and Wellbeing, University of Sunderland, UK. Samantha Chakraborty, Department of General Practice, Monash University, Melbourne, Australia. Adam Pattison Rathbone, School of Pharmacy, Faculty of Medical Sciences, Newcastle University; and Newcastle upon Tyne Hospitals NHS Foundation Trust, Newcastle upon Tyne, UK. Stuart Watson, Translational and Clinical Research Institute, Newcastle University; and Inpatient Services, Cumbria, Northumberland, Tyne and Wear NHS Foundation Trust, Newcastle upon Tyne, UK. Tim Donaldson, Pharmacy Department, Cumbria, Northumberland, Tyne and Wear NHS Foundation Trust, Newcastle upon Tyne, UK. Kirstie N. Anderson, Department of Neurology, Faculty of Medical Sciences, Newcastle University, Newcastle upon Tyne; and Regional Sleep Disorders Service, Newcastle upon Tyne Hospitals NHS Foundation Trust, UK.
Data availability
The data that support the findings of this study are available from the corresponding author, A.P., upon reasonable request.
Acknowledgements
A.P. thanks the Academic Health Science Network for the North East and North Cumbria (AHSN NENC) for the funding of this project; the pharmacy department, Cumbria, Northumberland, Tyne and Wear NHS Foundation Trust, for their support in releasing staff time to conduct this work; the Newcastle University VOICE patient and public involvement group for their advice in this project; and the Regional Sleep Service, Newcastle upon Tyne Hospitals, for arranging for the professional production of the video resource.
Author contributions
Conceptualisation: A.P., M.K., A.Y., A.P.R., S.W. and K.N.A.; data curation: A.P.; formal analysis, A.Y.; funding acquisition: A.P., S.W., T.D. and K.N.A.; Methodology: A.P., A.Y., J.L., S.C. and K.N.A.; project administration, A.P.; resources, A.P.R. and K.N.A.; supervision, M.K., J.L., S.C., A.P.R., S.W., T.D. and K.N.A.; writing – original draft, A.P. and K.N.A.; writing – review and editing, M.K., A.Y., J.L., S.C., A.P.R., S.W., T.D. and K.N.A.
Funding
This work was supported by the Academic Health Science Network for the North East and North Cumbria (AHSN NENC), project number MO3. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Declaration of interest
None.











eLetters
No eLetters have been published for this article.