Introduction
Handshake antimicrobial stewardship is a form of prospective audit and feedback characterized by the collaborative review of all antimicrobials by an infectious diseases (ID) physician and ID pharmacist followed by in-person rounds to provide feedback to prescribers. Reference MacBrayne, Williams and Levek1 This intervention has been recognized as a “leading practice” by The Joint Commission and is highlighted in the Core Elements of Hospital Antimicrobial Stewardship defined by the Centers for Disease Control (CDC). Reference Baker, Hyun, Neuhauser, Bhatt and Srinivasan2,3 Prior descriptions of this intervention in pediatric hospital settings have shown that handshake stewardship can have a sustained impact on reducing antimicrobial use without negatively impacting patient outcomes. Reference MacBrayne, Williams and Levek1 In adult patient populations, literature often focuses on rounds in the intensive care units. Reference Taggart, Leung, Muller, Matukas and Daneman4–Reference Seidelman, Turner and Wrenn6 Outside of intensive care units, there are some data supporting the use of “high intensity” or “academic detailing” strategies, similar to handshake stewardship, to reduce broad-spectrum antimicrobial use, but there are limited data on the applicability of this strategy more broadly in adult hospitals. Reference Langford, Brown, Chan and Downing7–Reference Gorsline, Miller, Bobbitt, Satyanarayana, Baljevic and Staub9
Considering workload volume and engagement of key stakeholders, our antimicrobial stewardship program implemented handshake stewardship rounds in patients admitted to medicine floors. The purpose of this study was to describe the implementation of our service and evaluate its impact on antimicrobial use.
Methods
Barnes-Jewish Hospital is a large, academic medical center (1,278 staffed beds) located in Saint Louis, Missouri that has had an antimicrobial stewardship program (ASP) since 1985. Core program components include prior authorization and 72-hour prospective audit and feedback of select antimicrobials, institution-specific guidelines for common infectious diseases, clinical decision support within the electronic health record (Epic®, Epic Systems Corporation, Verona, WI), and tracking/reporting of antimicrobial use data (additional details in Supplementary Table 1). From August 2019 to June 2021, dedicated program FTEs included 1.0 FTE of infectious diseases (ID) physician support and 2.0 FTE of ID pharmacist support. An additional 1.0 FTE ID physician support was added in July 2021 to support handshake antimicrobial stewardship.
Handshake stewardship was implemented on the thirteen medicine floors three days per week. The ∼250 medicine beds are covered by either one of twelve medicine teaching services (>150 internal medicine residents) or twelve medicine hospitalist services (>100 hospitalist physicians). Eleven units had both medicine teaching services and hospitalists providing care for patients on those floors, including three telemetry units for cardiology patients. The final two units housed patients cared for exclusively by the hospitalist service. Daily activities of the handshake stewardship service included reviewing all antimicrobials for patients admitted to a medicine service without a formal ID consultation split by the ID physician and ID pharmacist (∼1 h each). The ID physician and ID pharmacist then met to review cases and potential interventions (∼1 h). Reviews focused on general appropriateness of antimicrobial orders within the context of each individual patient. The team rounded in-person to discuss recommendations with the primary team providers (∼1.5 h). Following rounds, the ID physician and ID pharmacist documented recommendations in the pharmacy intervention system (∼0.5 h). Two days per week the handshake stewardship team reviewed medicine teaching service patients and one day per week hospitalist patients. Handshake stewardship rounds started in August 2021.
To account for unpredictable fluctuations in antibiotic use due to the COVID-19 pandemic, two years of pre- (August 2019–July 2021) and one year post- (August 2021–July 2022) implementation of the handshake stewardship service on medicine floors were evaluated. Antimicrobial use was measured in days of therapy (DOT) per 1,000 days present (DP) as reported to the CDC’s National Healthcare Safety Network Antimicrobial Use module. Data were aggregated for each month of the study periods. The antibiotic spectrum index (ASI) was calculated using an expanded and modified version developed by Gerber et al. Reference Gerber, Hersh, Kronman, Newland, Ross and Metjian10,Reference Ilges, Ritchie, Krekel, Neuner and Micek11 Assessment of intravenous versus oral antibiotic usage was performed using the digestive DOT (dDOT) over total DOT (tDOT) metric as described by Moehring et al. Reference Moehring, Davis and Dodds Ashley12 Descriptive data on types of recommendations and acceptance rate were evaluated. Patient outcomes included length of stay and in-hospital mortality. Rates of Clostridioides difficile infection were also evaluated; testing was performed by a toxin EIA assay (Alere TOX A/B II, Abbott, Lake Bluff, IL) from August 2019 to February 15, 2022 and from February 16, 2022 to July 2022 testing was performed using a GDH & Toxin A/B combination assay (C. Diff Quik Check Complete® TechLab, Blacksburg, VA) with discordant results automatically reflexed to nucleic acid amplification (Cepheid Xpert C. difficile PCR Cepheid, Sunnyvale, CA). Feedback from medical residents and hospitalists was evaluated through surveys sent via email with two reminders over three weeks in December 2021 and November 2022, respectively (detailed survey questions in Supplementary Table 2).
This study was approved by the local institutional review board.
Intervention and survey data were examined using descriptive statistics. For comparisons of pre- and post-intervention groups, categorical data were compared by the χ 2 test and continuous variables with a Mann–Whitney test. Antibiotic usage per month and location were plotted along with a cubic spline smoother (with six equally spaced knots) to identify general temporal trends. Furthermore, an interrupted time-series analysis comparing both the immediate effect of the intervention (the intercept at time 0) and change of slope in DOT/1,000 DP pre-intervention to post-intervention was performed. Data were analyzed using R version 4.2.2 (R Foundation for Statistical Computing, Vienna, Austria).
Results
The handshake stewardship service reviewed 7,410 patient charts, rounded 138 days, and made 1,558 recommendations (daily average of 54 charts and 11.3 recommendations, Table 1). The most common recommendations were to discontinue (37%), de-escalate (28%), or shorten/define duration (14%) of antimicrobials. The overall intervention acceptance rate was 80% but varied by intervention type from a high of 95% for dose optimization to a low of 58% for diagnostic-related recommendations.
Table 1. Description of handshake antimicrobial stewardship interventions

Note. MRSA, methicillin-resistant Staphylococcus aureus.
a 223/437 (51%) De-escalation recommendations included a recommendation to switch from an IV agent to a PO agent with a narrower spectrum of activity.
b Other antimicrobial stewardship recommendation types included 13 lengthen duration of therapy, 8 drug interaction or contraindication -related, 1 allergy clarification.
c Multiple agents were intervened upon in 18% of overall recommendations; percentages do not add up to 100%.
d MRSA agents included ceftaroline, daptomycin, linezolid, and intravenous vancomycin.
e Other agents included 22 clindamycin, 21 sulfamethoxazole-trimethoprim, 16 ampicillin, 14 nitrofurantoin, 11 cefazolin, 5 aztreonam, 4 oral vancomycin, 2 atovaquone, 1 baricitinib, 1 dexamethasone, 1 fidaxomicin, 1 fosfomycin, 1 hydroxychloroquine, 1 oxacillin, and 1 penicillin.
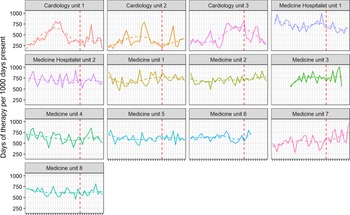
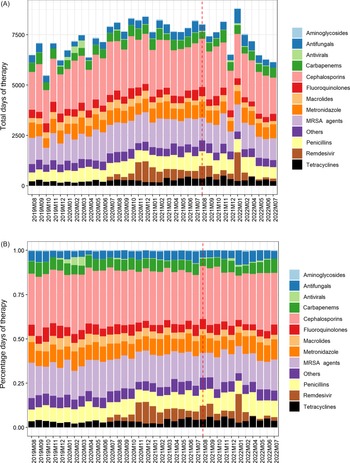
Overall, hospital-wide antimicrobial use during the pre-implementation and post-implementation phases did not significantly differ, with the monthly median being 749 DOT/1,000 DP (IQR 744–762) before and 742 (IQR 725–763) after implementation (p = 0.7). Antimicrobial use outcomes specific to the thirteen medicine floors are displayed in Table 2. On the medicine floors, there was no significant difference in antimicrobial use pre- and post- implementation of handshake stewardship with 626 DOT/1,000 DP (IQR 523–712) vs 646 (IQR 530–719), p = 0.6. Figure 1 displays the interrupted time series for the thirteen medicine units. During the twenty-four months preimplementation, there was an increasing trend (p = 0.06), and post-implementation of handshake stewardship there was a decreasing trend in antimicrobial use (p = 0.3). There was no immediate change noticeable from the intervention (p = 0.4). However, in a post hoc supplementary analysis, there was a significant difference when the intervention effect was lagged by 3 months both in terms of the delayed “immediate” effect (p = 0.03) and for the post-implementation slope effect (p = 0.03). Trends in antimicrobial use per month for each of the 13 floors and groups of antimicrobials are displayed in Figures 2 and 3. Subgroup analysis excluding remdesivir showed the same trends (Supplementary Figure 1). Evaluating specific antimicrobial groups or agents, there were some significant differences in use before and after handshake stewardship was implemented, including a significant increase in tetracyclines (17 vs. 26, p < 0.001) and amoxicillin-clavulanate (11 vs. 15, p < 0.001). Use of macrolides (24 vs. 19, p = 0.001), meropenem (24 vs. 17, p = 0.007), and vancomycin (78 vs. 70, p = 0.002) were significantly decreased. Overall antimicrobial use was not significantly different pre- and post- intervention after applying an antimicrobial spectrum index to DOT/1,000 DP (3017 ASI/1,000 DP vs. 2,970 ASI/1,000 DP, p = 0.7). There was a marginal trend toward an increase in oral antimicrobials as measured by the dDOT/tDOT metric (0.32 vs. 0.34, p = 0.06).
Table 2. Antimicrobial days of therapy per 1000 days present

Note. dDOT, digestive days of therapy; tDOT, total days of therapy. Median (interquartile range). Penicillin and derivatives include aminopenicillins, oxacillin, amoxicillin-clavulanate, ampicillin-sulbactam, and piperacillin/tazobactam.

Figure 1. Antibiotic usage by month and location (dashed); intervention start dashed red line; overall cubic spline smoother shown (six knots, light blue/dashed) with 95% confidence intervals shaded, and average consumption from the predicted distribution of the interrupted time series segmented regression model (red/solid, interrupt at August 2021).

Figure 2. Monthly antimicrobial use by individual medicine unit; nonparametric smoother shown dashed.

Figure 3. Total antibiotic usage by month and by antibiotic class, absolute (A) and percentage (B); intervention start shown as dashed red line.
In-hospital mortality during the study period was unchanged pre- and post-intervention (0.02% (95% CI 0.02–0.03%) vs. 0.03% (95% CI 0.02–0.03%), p = 0.2) (see Table 3). The number of hospital onset C. difficile infection cases increased on the medicine floors in the post intervention group (monthly median 1.5 (IQR: 0.75–3) vs. 3.5 (IQR: 2.75–6.25), p < 0.001 (see Supplementary Figure 2). Median hospital length of stay increased post-intervention to 8.2 d (IQR: 2.7, 9.1) from 7.3 (IQR: 2.5, 8.1) d pre-intervention (p < 0.001) (see Supplementary Figure 3).
Table 3. Medicine units patient outcomes

Note. Median (interquartile range); CI, confidence interval.
Response rates for the surveys sent for feedback about the handshake stewardship service were 28% (45/162) for medical residents and 43% (49/113) for hospitalists. Respondents indicated the frequency of contact with the handshake stewardship service was just right (88.5% and 88%) for medical residents and hospitalists, respectively. The preferred method of communication was in-person (66% for residents, 52% for hospitalists) followed by Epic® chat message (34% residents, 43% hospitalists). The full results of the surveys are available in the Supplementary Table 2. Residents and hospitalists responded that they felt the recommendations from handshake stewardship to be helpful always (54% and 48%), most of the time (40% and 38%), about half of the time (3% and 7%), or sometimes (3% and 7%), and no one answered the recommendations were never helpful. Free-text comments were overwhelmingly favorable.
Discussion
After one year of performing handshake stewardship on the medicine floor teams at a large teaching hospital with an existing stewardship program, there have been some important shifts in patterns of overall antibiotic use. While there was not a significant decrease in overall antibiotic use immediately following the implementation of the intervention, there has been a reversal of a trend of overall increasing antibiotic use that was statistically significant when the intervention effect was lagged by 3 months. Additionally, there were significant decreases in the use of specific broad-spectrum agents such as vancomycin and meropenem. Another important shift in usage patterns observed was the trend toward an increasing preference for oral agents as opposed to intravenous agents with the dDOT/tDOT metric. This may suggest that there were shifts toward more appropriate antibiotic use.
Unlike prior descriptions of handshake stewardship services, we did not find a statistically significant change in overall antibiotic use, which could be attributed to several factors. First, the size of the hospital and number of providers impacted the reach of our service and could have diffused the effects of any changes in antibiotic use. A high intensity stewardship service implemented in five internal medicine units at a 400-bed community hospital in Toronto demonstrated a reduction in overall antibiotic use from 483 to 442 defined daily doses/1,000 patient days. Reference Langford, Brown, Chan and Downing7 Our study had 13 medicine units and >200 different providers some of whom were only on service a limited number of times over the year of the intervention thus impacting the reach of our intervention. Second, prior to this intervention, there was already an active antimicrobial stewardship program in place, and our starting antibiotic usage was slightly below the national average. Reference Goodman, Cosgrove and Pineles13 Third, there is a significant proportion of antibiotic use driven by ID consult services, and handshake stewardship did not routinely review these antimicrobial regimens. The antibiotic use data was at the unit level; therefore, patients with ID consults were unable to be excluded from this outcome measurement. Fourth, the frequency of handshake stewardship rounds was only twice a week for medicine teaching services and once a week for hospitalist services. The Children’s Hospital of Colorado team, who pioneered handshake stewardship, started rounding three days per week but increased to five days per week after the first year and demonstrated a sustained decrease in overall antimicrobial use hospital wide after five years. Reference MacBrayne, Williams and Levek1 The less frequent interaction in our study may have reduced the effect of the intervention. However, our acceptance rates compared favorably to prior studies describing this intervention. Seidelman et al. reported acceptance rates of 77.5% at one week, Reference Seidelman, Turner and Wrenn6 whereas Hurst et al. reported acceptance rates of 86% at the end of the workday. Reference Hurst, Child and Parker14
Safety measures in stewardship studies are crude, but we saw no change in in-hospital mortality in the pre- and post-intervention period. We did see an unexpected increase in both length-of-stay and C. difficile infection rates in the studied period. The former likely reflects the overall secular trend of increasing lengths of stay that has been observed nationally since the onset of the COVID-19 pandemic, while the latter likely reflects a significant change in the C. difficile testing protocol implemented in 2022 (see supplementary discussion). 15
Stewardship interventions are difficult to generalize across different healthcare settings due to disparities in resources, tracking capabilities, baseline practice patterns, and regional microbiological profiles. While handshake stewardship is a resource-intensive intervention, the benefit of that upfront cost in effort is the inherent flexibility and adaptability of the intervention to specific contexts. Because of the personal and individual nature of the intervention, relationships between the stewardship team and multiple provider teams can be built and strengthened, which can then affect an overall shift in prescriber culture. These benefits are less tangible and difficult to track; however, the high intervention acceptance rate and enthusiastic provider survey feedback reflect these benefits. And indeed, following the initial enthusiastic response on the medicine floors, we expanded the intervention to the surgical floors and one of our ICUs. Based on the positive feedback from medical residents, we created a formal medical resident rotation, which has become popular. Handshake stewardship can also change with the institution—as patient populations, antibiograms, and institutional priorities shift, handshake stewardship teams can adapt in real time.
Limitations in this study stem in part from limitations in tracking capabilities. Only inpatient DOT were included in this analysis, and it is unknown whether the shorter durations recommended inpatient reliably translated to shorter outpatient durations. Second, our analysis was limited by waves of COVID surges that occurred through both the pre- and post-intervention periods, although we tried to account for this with some subgroup analysis excluding remdesivir. It has been shown that COVID surges are correlated with increased antibiotic use, which influenced our results. Reference Dodds Ashley, Lokhnygina and Doughman16,Reference Barlam, Al Mohajer and Al-Tawfiq17 Third, we did not assess appropriateness or adherence with guidelines as part of this analysis.
In conclusion, adding a handshake stewardship team does endow additional benefits—both tangible, with shifts in antibiotic use patterns toward narrower and oral regimens, and less tangible benefits in changing overall hospital culture toward antibiotics by being a visible and accessible resource to help guide antibiotic use.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/ash.2023.465.
Acknowledgements
None.
Financial support
None reported.
Competing interests
All authors report no conflicts of interest relevant to this article.