40 results
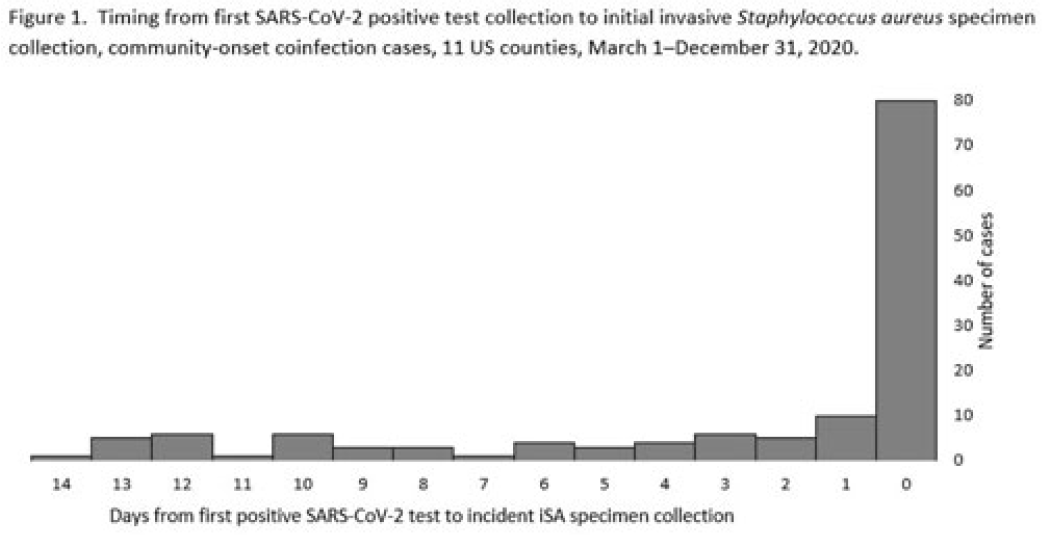
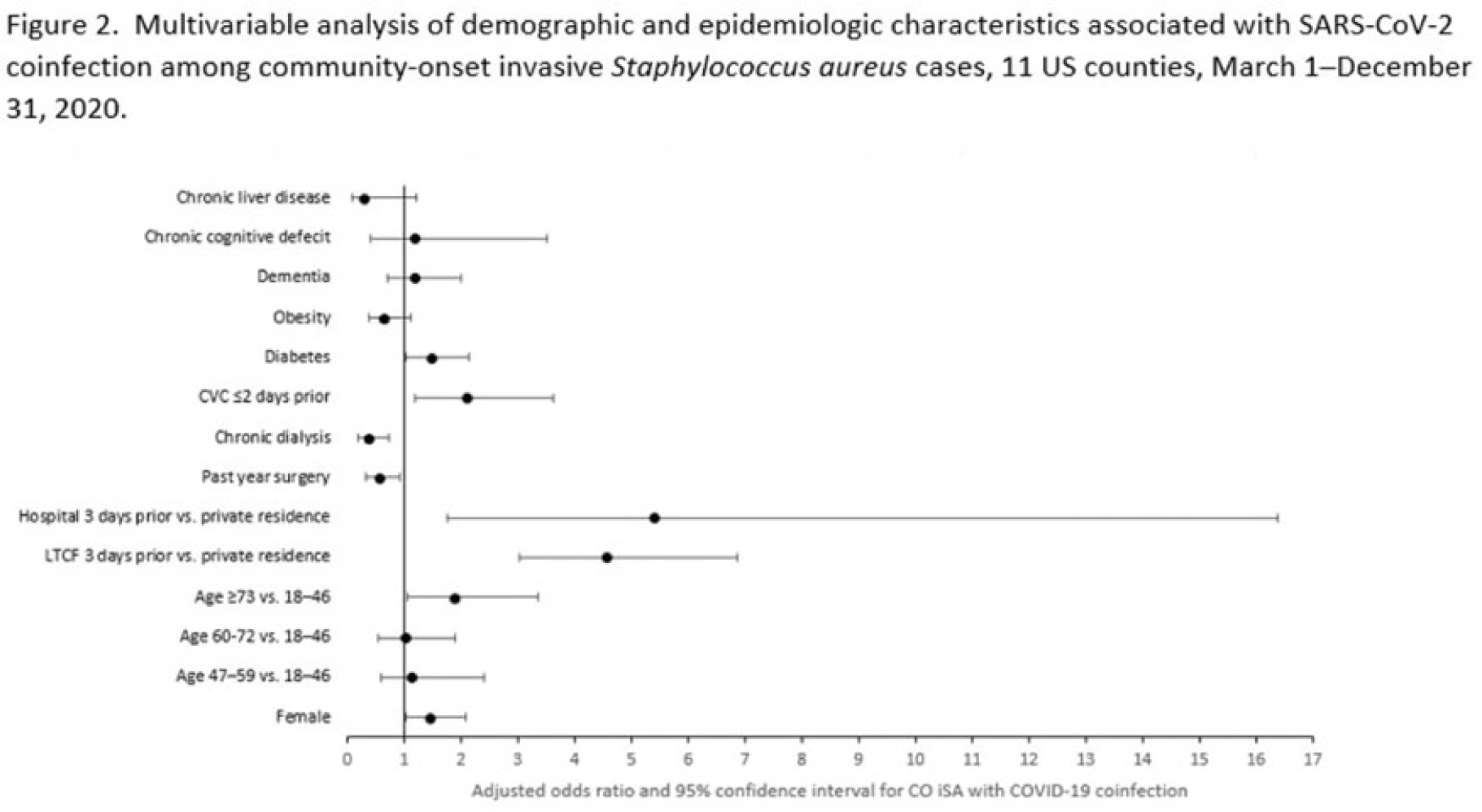
Factors associated with SARS-CoV-2 and community-onset invasive Staphylococcus aureus coinfection, 2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s84-s85
-
- Article
-
- You have access
- Open access
- Export citation
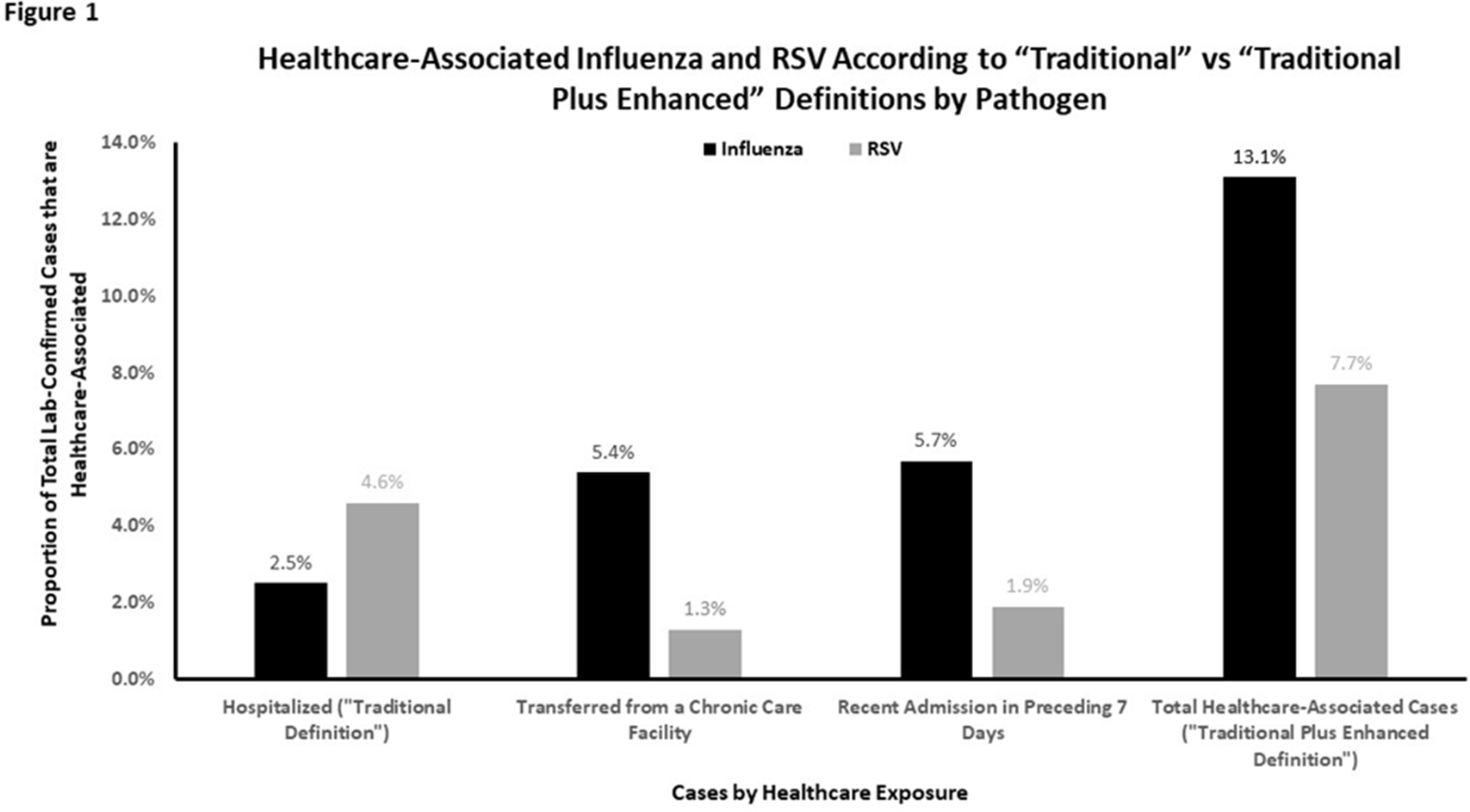
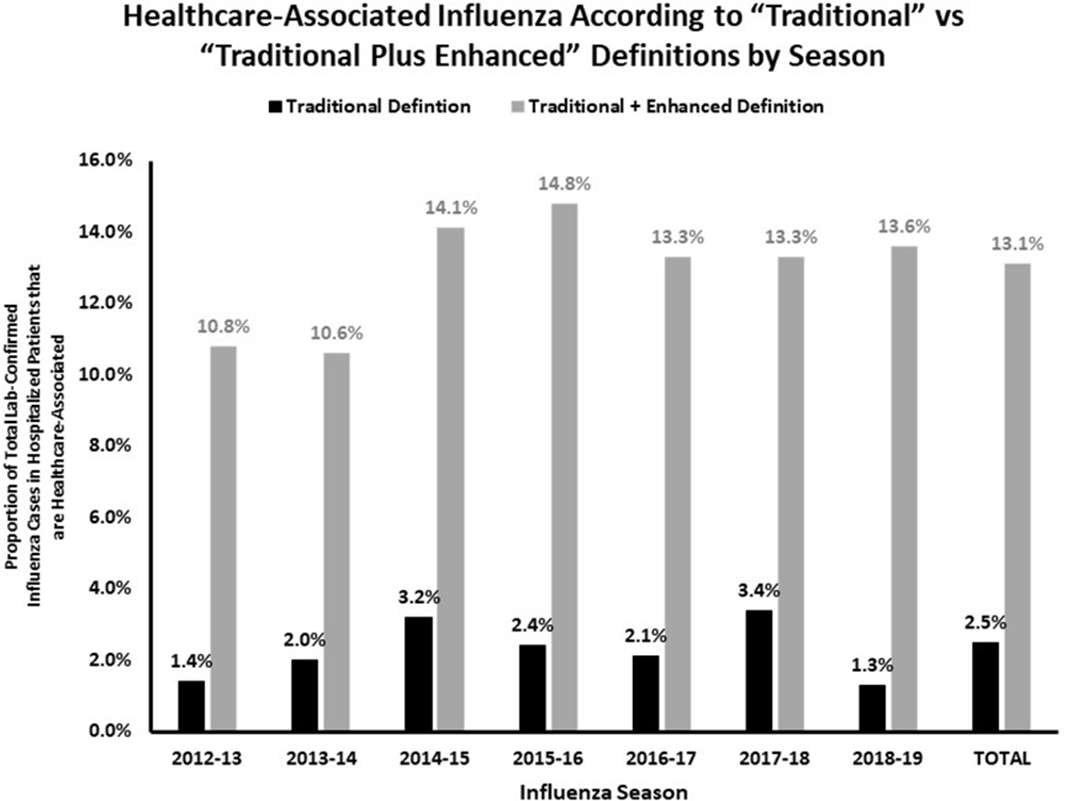
Traditional definition of healthcare-associated influenza underestimates cases associated with other healthcare exposures in a population-based surveillance system
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 11 / November 2023
- Published online by Cambridge University Press:
- 19 April 2023, pp. 1816-1822
- Print publication:
- November 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
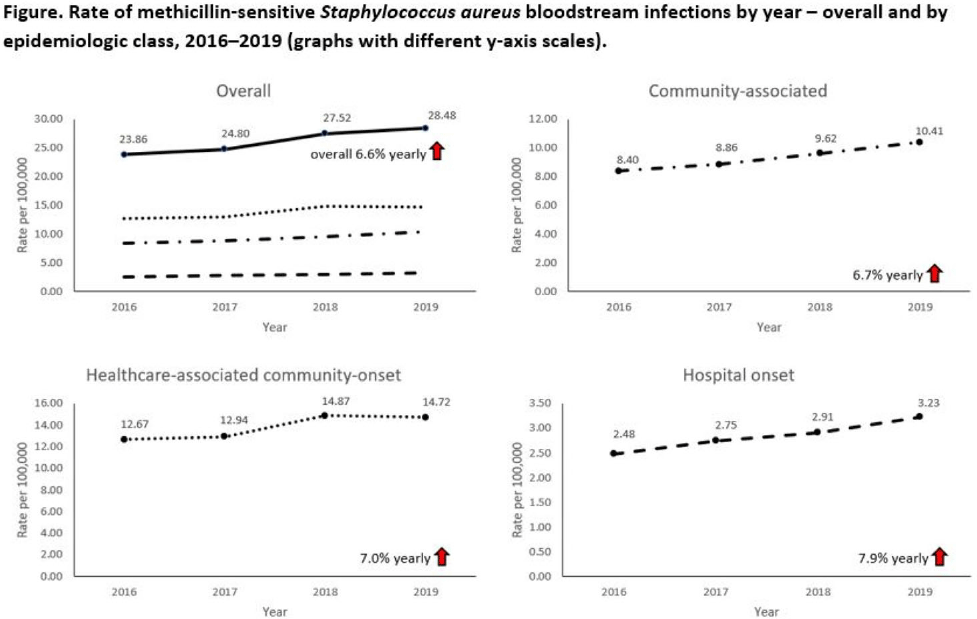
Increases in methicillin-sensitive Staphylococcus aureus bloodstream infection incidence, 2016–2019
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s63-s64
-
- Article
-
- You have access
- Open access
- Export citation
Hospital-acquired influenza in the United States, FluSurv-NET, 2011–2012 through 2018–2019
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 10 / October 2022
- Published online by Cambridge University Press:
- 05 October 2021, pp. 1447-1453
- Print publication:
- October 2022
-
- Article
- Export citation
Structural and Functional Analysis of the D614G SARS-CoV-2 Spike Protein Variant
-
- Journal:
- Microscopy and Microanalysis / Volume 27 / Issue S1 / August 2021
- Published online by Cambridge University Press:
- 30 July 2021, pp. 3260-3262
- Print publication:
- August 2021
-
- Article
-
- You have access
- Export citation
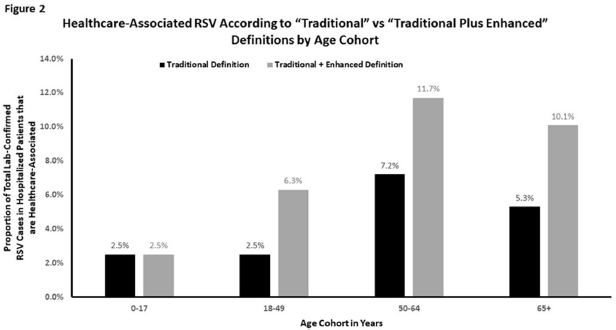
Respiratory Syncytial Virus: An Underrecognized Healthcare-Associated Infection
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s76-s77
-
- Article
-
- You have access
- Open access
- Export citation
Traditional Definition of Healthcare-Associated Influenza Underestimates Cases Associated with Other Healthcare Exposures
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s12-s13
-
- Article
-
- You have access
- Open access
- Export citation
Molecular Typing of Invasive Staphylococcus aureus from the Emerging Infections Program (EIP) Using Whole-Genome Sequencing
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s71-s72
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Characteristics Associated With Invasive Staphylococcus aureus Infection Rates in Nursing Homes, Emerging Infections Program
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s60-s61
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Characteristics of Cases With Polymicrobial Bloodstream Infections Involving Candida in Multisite Surveillance, 2017
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s163
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Antimicrobial Nonsusceptibility Among Invasive MRSA USA300 Strains by Healthcare Exposure, Three Sites, 2005–2016
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s120-s121
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Policy statement from the Society for Healthcare Epidemiology of America (SHEA): Only medical contraindications should be accepted as a reason for not receiving all routine immunizations as recommended by the Centers for Disease Control and Prevention
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 1 / January 2021
- Published online by Cambridge University Press:
- 17 September 2020, pp. 1-5
- Print publication:
- January 2021
-
- Article
-
- You have access
- HTML
- Export citation
Nosocomial hepatitis A outbreak among healthcare workers and patients in a community hospital during an ongoing statewide outbreak
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 2 / February 2021
- Published online by Cambridge University Press:
- 03 September 2020, pp. 139-141
- Print publication:
- February 2021
-
- Article
- Export citation
Russian-olive (Elaeagnus angustifolia) genetic diversity in the western United States and implications for biological control
-
- Journal:
- Invasive Plant Science and Management / Volume 12 / Issue 2 / June 2019
- Published online by Cambridge University Press:
- 17 April 2019, pp. 89-96
-
- Article
- Export citation
Completeness of Methicillin-Resistant Staphylococcus aureus Bloodstream Infection Reporting From Outpatient Hemodialysis Facilities to the National Healthcare Safety Network, 2013
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 37 / Issue 2 / February 2016
- Published online by Cambridge University Press:
- 11 November 2015, pp. 205-207
- Print publication:
- February 2016
-
- Article
- Export citation
Nursery Epidemic Due to Multiply-Resistant Klebsiella pneumoniae: Epidemiologic Setting and Impact on Perinatal Health Care Delivery
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 3 / Issue 2 / March/April 1982
- Published online by Cambridge University Press:
- 02 January 2015, pp. 150-158
- Print publication:
- March/April 1982
-
- Article
- Export citation
Sustained Improvement in Hand Hygiene Adherence: Utilizing Shared Accountability and Financial Incentives
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 34 / Issue 11 / November 2013
- Published online by Cambridge University Press:
- 02 January 2015, pp. 1129-1136
- Print publication:
- November 2013
-
- Article
-
- You have access
- Export citation
Immunization for Vaccine-Preventable Diseases: Why Aren’t We Protecting Our Students?
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 32 / Issue 9 / September 2011
- Published online by Cambridge University Press:
- 02 January 2015, pp. 912-914
- Print publication:
- September 2011
-
- Article
- Export citation
On Being the First: Virginia Mason Medical Center and Mandatory Influenza Vaccination of Healthcare Workers
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 31 / Issue 9 / September 2010
- Published online by Cambridge University Press:
- 02 January 2015, pp. 889-892
- Print publication:
- September 2010
-
- Article
- Export citation
Mortality Rates Associated With Multidrug-Resistant Acinetobacter baumannii Infection in Surgical Intensive Care Units
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 29 / Issue 11 / November 2008
- Published online by Cambridge University Press:
- 02 January 2015, pp. 1080-1083
- Print publication:
- November 2008
-
- Article
- Export citation