25 results
Comparison of Medicare Claims-based Clostridioides difficile infection classification to chart review using a linked cohort
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s61-s62
-
- Article
-
- You have access
- Open access
- Export citation
Extended-Spectrum Beta-Lactamase Producing Enterobacterales Infections in the United States, 2012-2021
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s32-s33
-
- Article
-
- You have access
- Open access
- Export citation
Viral Kinetics of SARS-CoV-2 in Nursing Home Residents and Staff
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s73-s74
-
- Article
-
- You have access
- Open access
- Export citation
Utilization of multiplex molecular panels for urinary tract infections, Medicare claims, 2016 – 2022
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, p. s78
-
- Article
-
- You have access
- Open access
- Export citation
Organism-specific Trends in Carbapenem-resistant Enterobacterales Infections in a Cohort of Hospitalized Patients, 2012–2022
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, p. s156
-
- Article
-
- You have access
- Open access
- Export citation
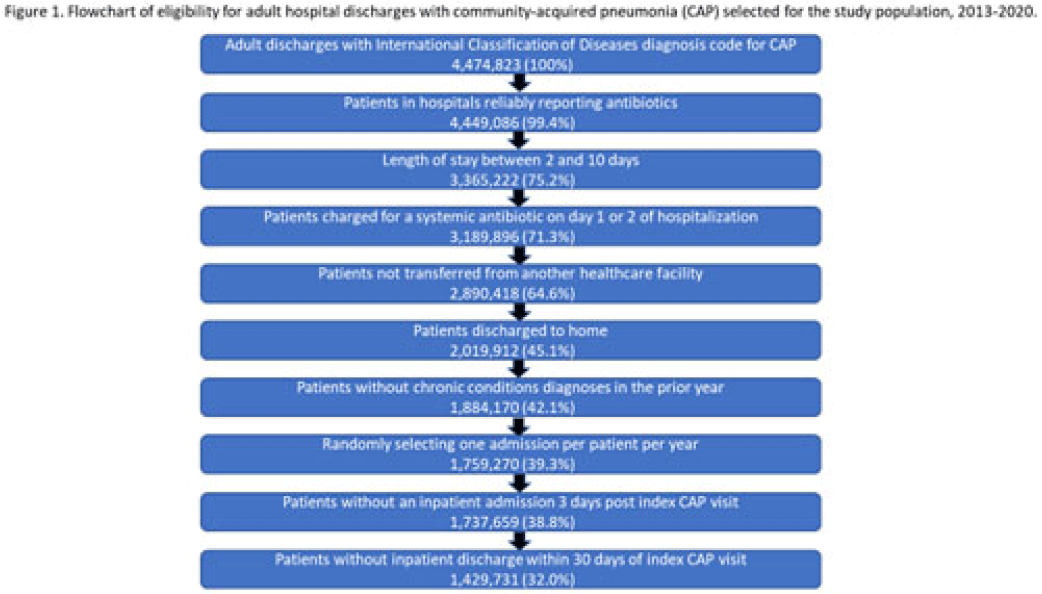
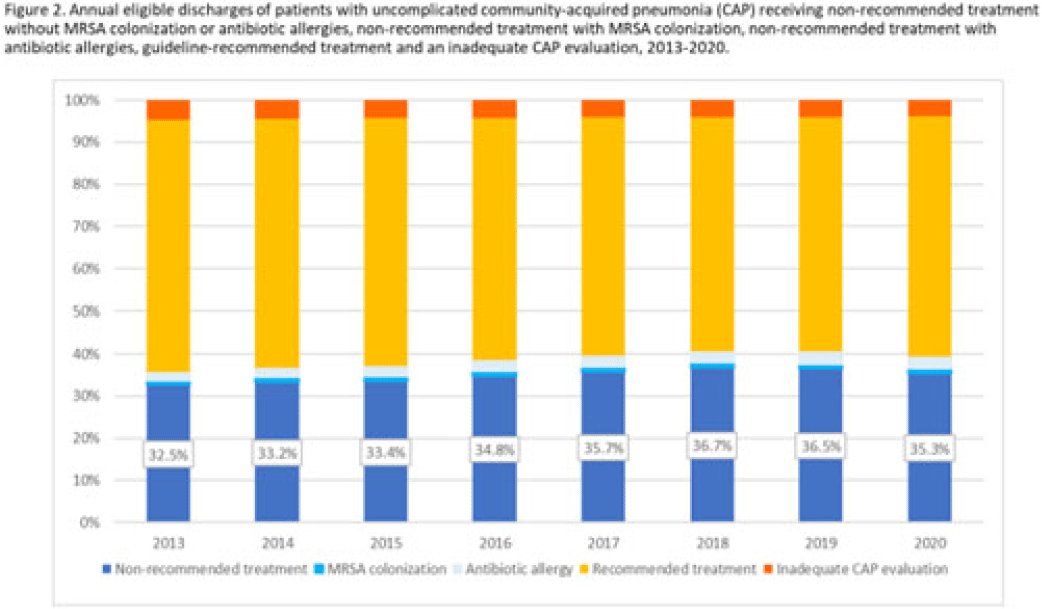
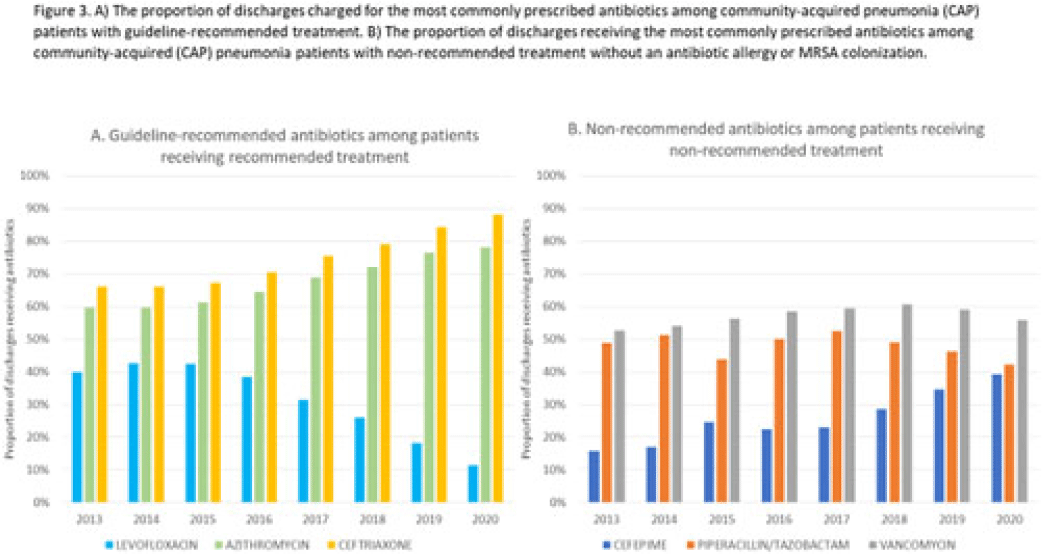
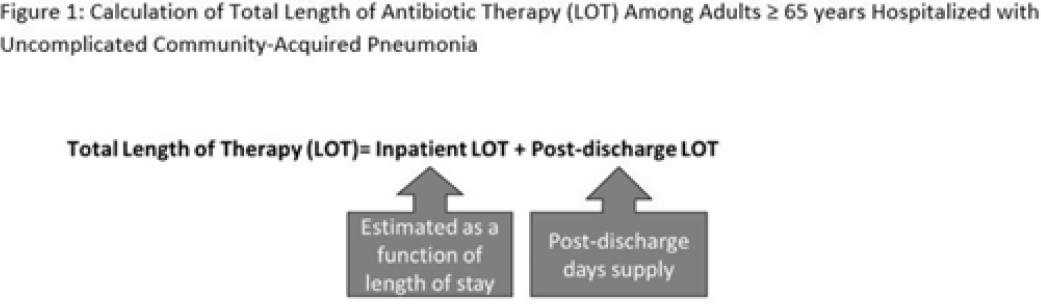
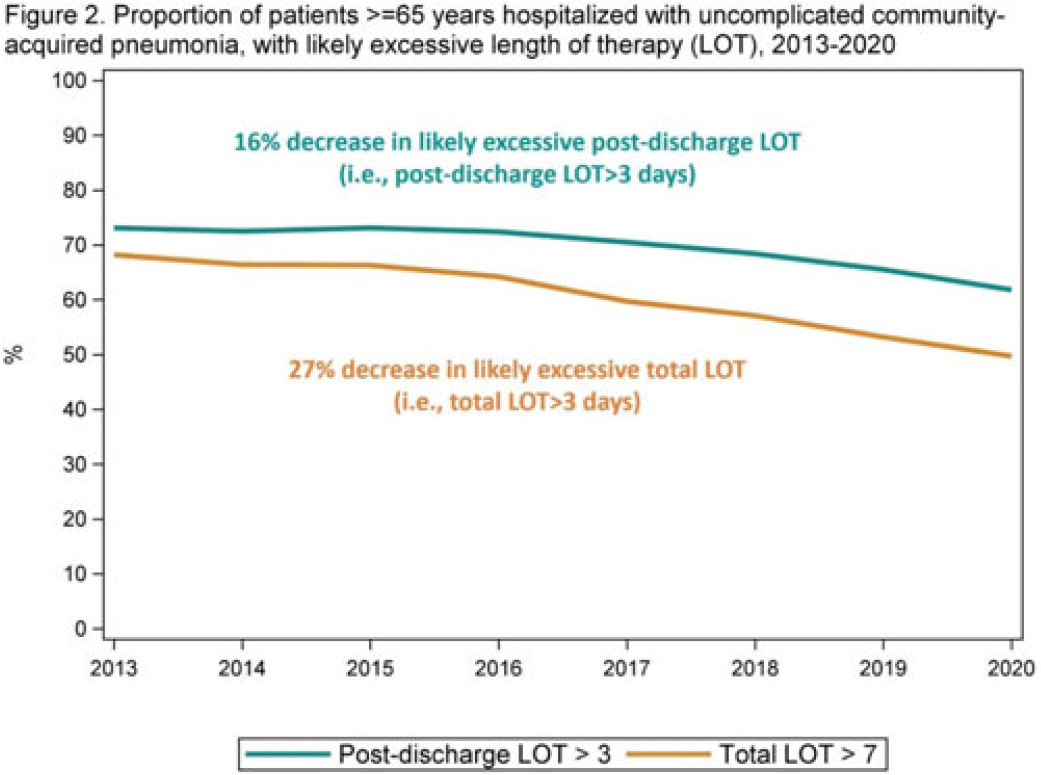
Length of antibiotic therapy among adults hospitalized with uncomplicated community-acquired pneumonia, 2013–2020
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 6 / June 2024
- Published online by Cambridge University Press:
- 14 February 2024, pp. 726-732
- Print publication:
- June 2024
-
- Article
-
- You have access
- HTML
- Export citation
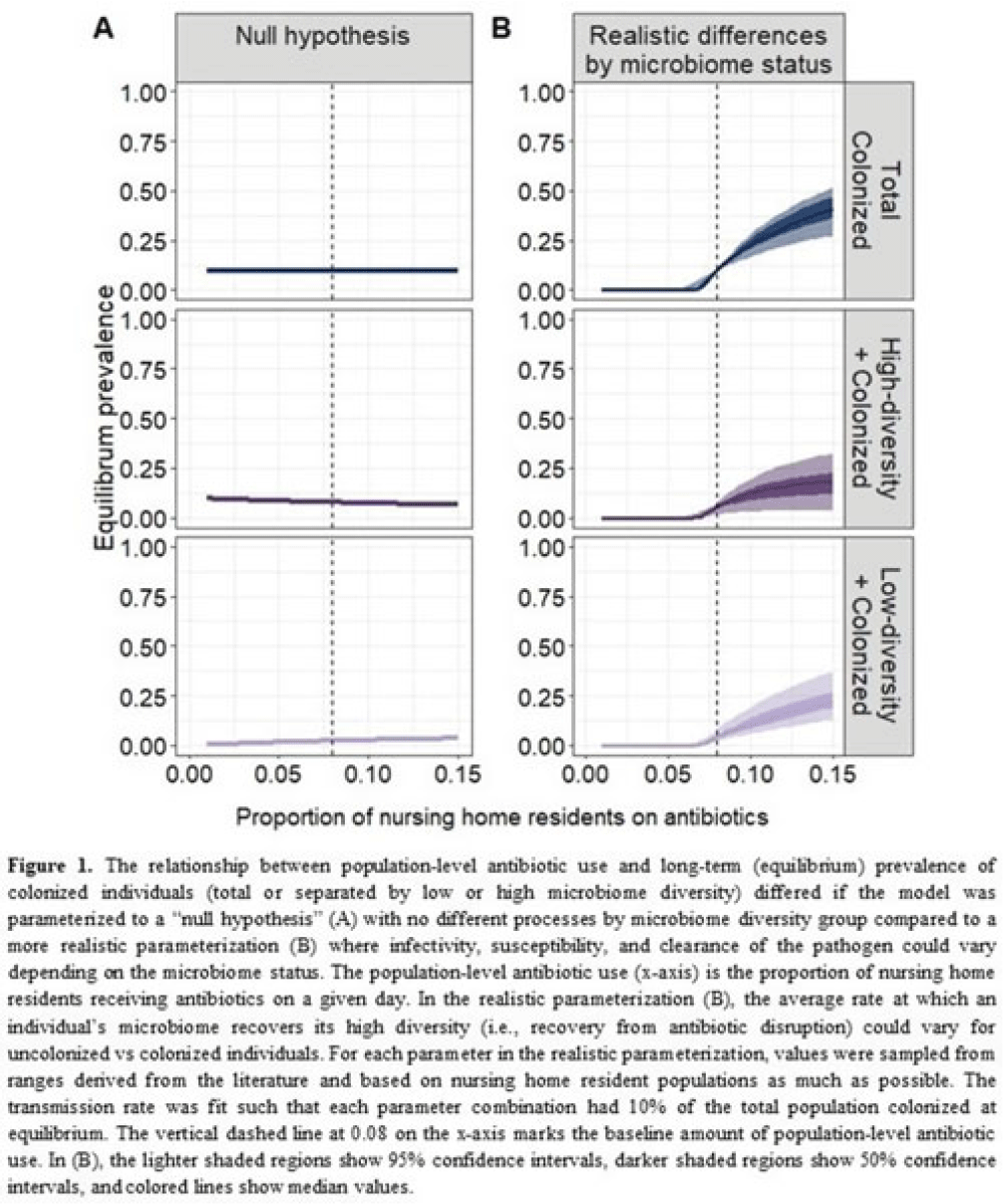
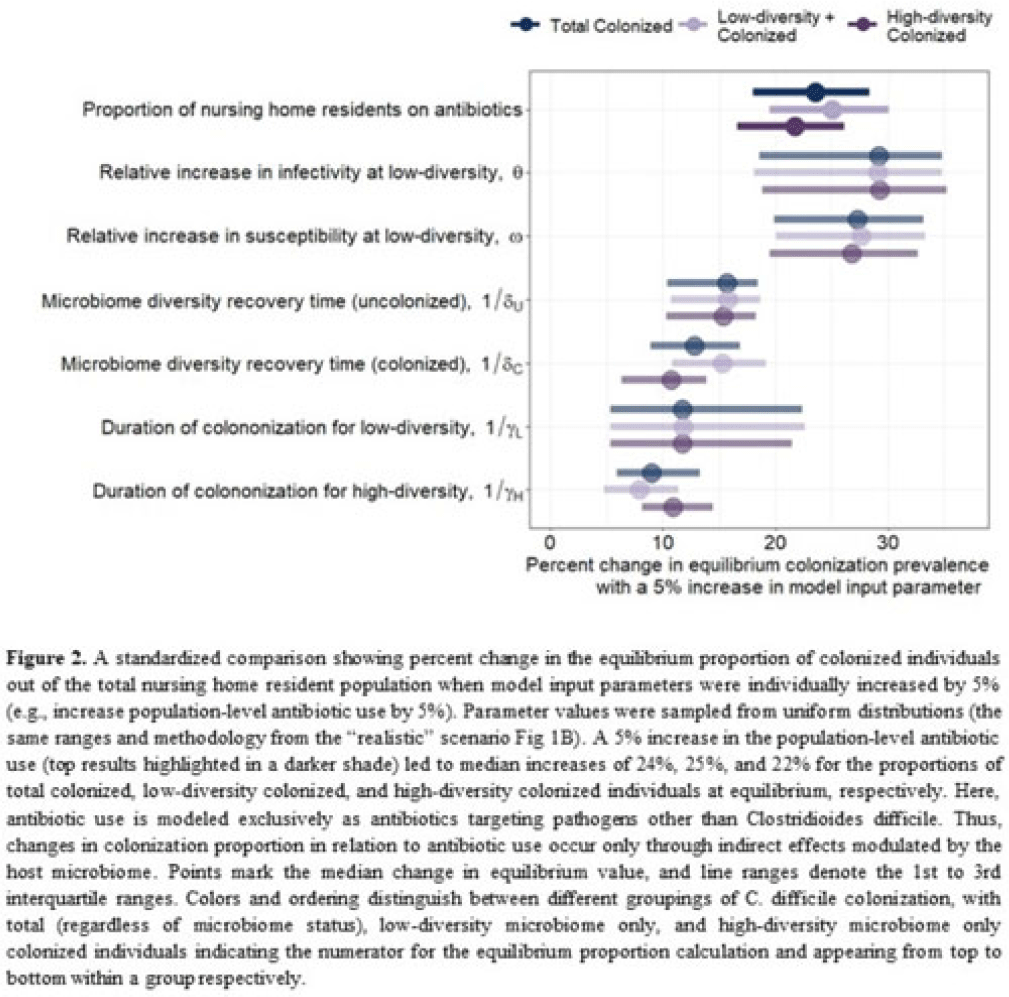
Uncovering gut microbiota-mediated indirect effects of antibiotic use on Clostridioides difficile transmission
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s104-s105
-
- Article
-
- You have access
- Open access
- Export citation
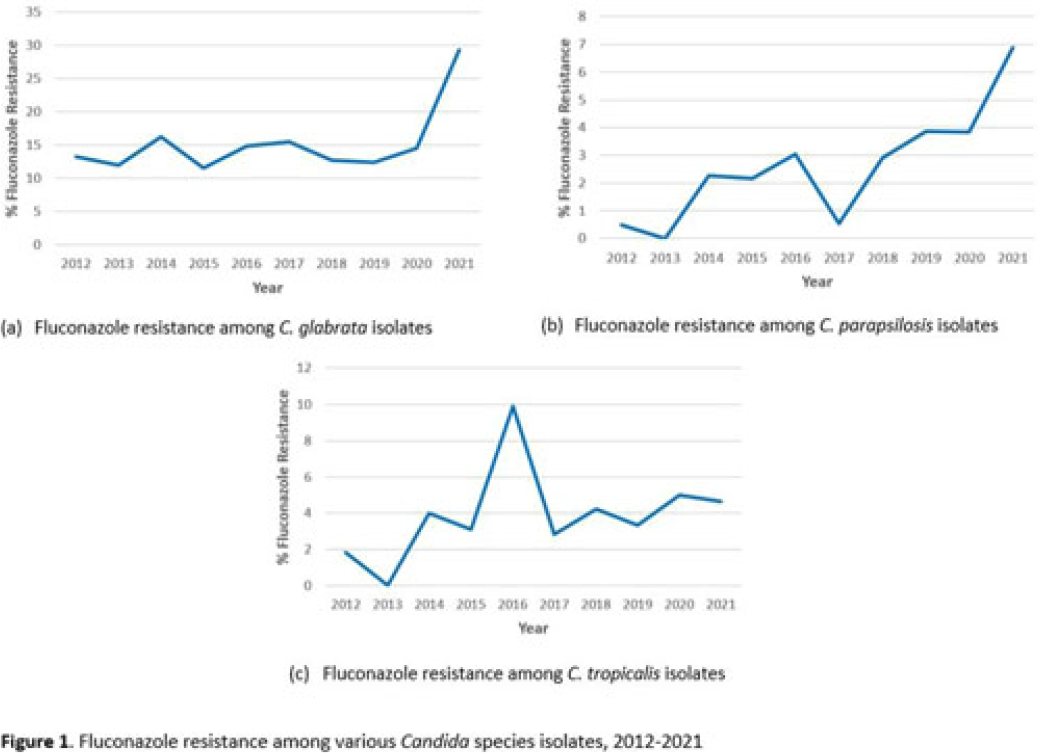
Fluconazole resistance in non-albicans Candida species in the United States, 2012-2021
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s29-s30
-
- Article
-
- You have access
- Open access
- Export citation
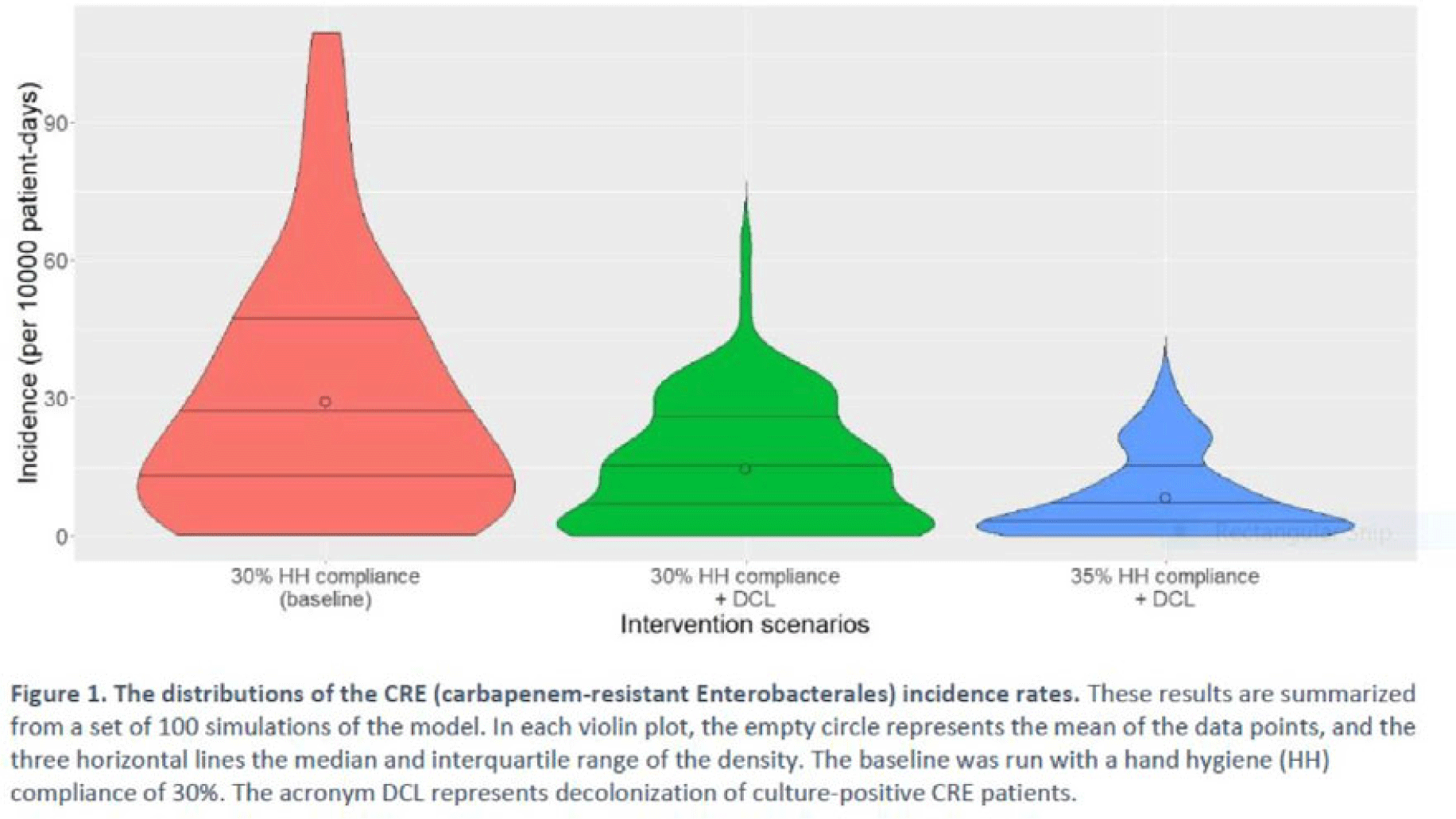
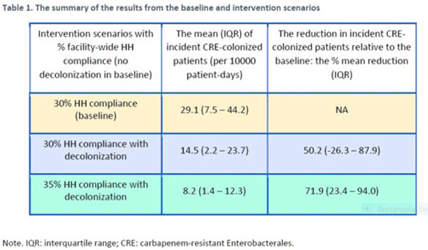
Decolonization of hospital patients may aid efforts to reduce transmission of carbapenem-resistant Enterobacterales
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s59-s60
-
- Article
-
- You have access
- Open access
- Export citation
Empiric antibiotic selection for community-acquired pneumonia in US hospitals, 2013–2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s26-s27
-
- Article
-
- You have access
- Open access
- Export citation
Length of antibiotic therapy among adults aged ≥65 years hospitalized with uncomplicated community-acquired pneumonia, 2013-2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s26
-
- Article
-
- You have access
- Open access
- Export citation
Characteristics of nursing home residents and healthcare personnel with repeated severe acute respiratory coronavirus virus 2 (SARS-CoV-2) tests positive ≥90 days after initial infection: Four US jurisdictions, July 2020–March 2021
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 5 / May 2023
- Published online by Cambridge University Press:
- 20 May 2022, pp. 809-812
- Print publication:
- May 2023
-
- Article
- Export citation
Trends in facility-level rates of Clostridioides difficile infections in US hospitals, 2019–2020
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 2 / February 2023
- Published online by Cambridge University Press:
- 19 May 2022, pp. 238-245
- Print publication:
- February 2023
-
- Article
- Export citation
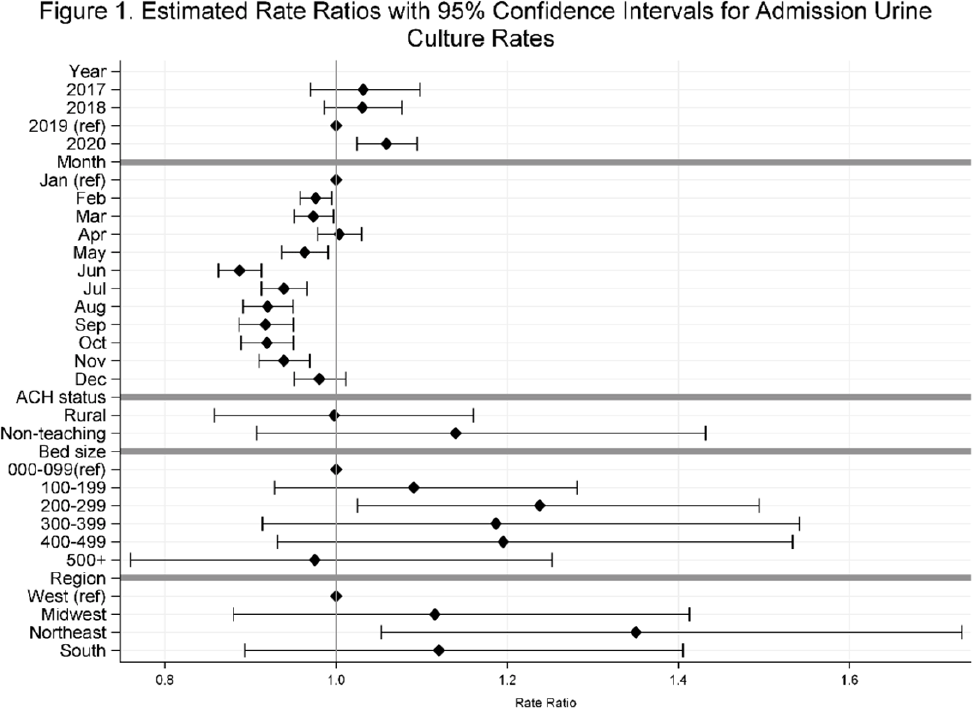
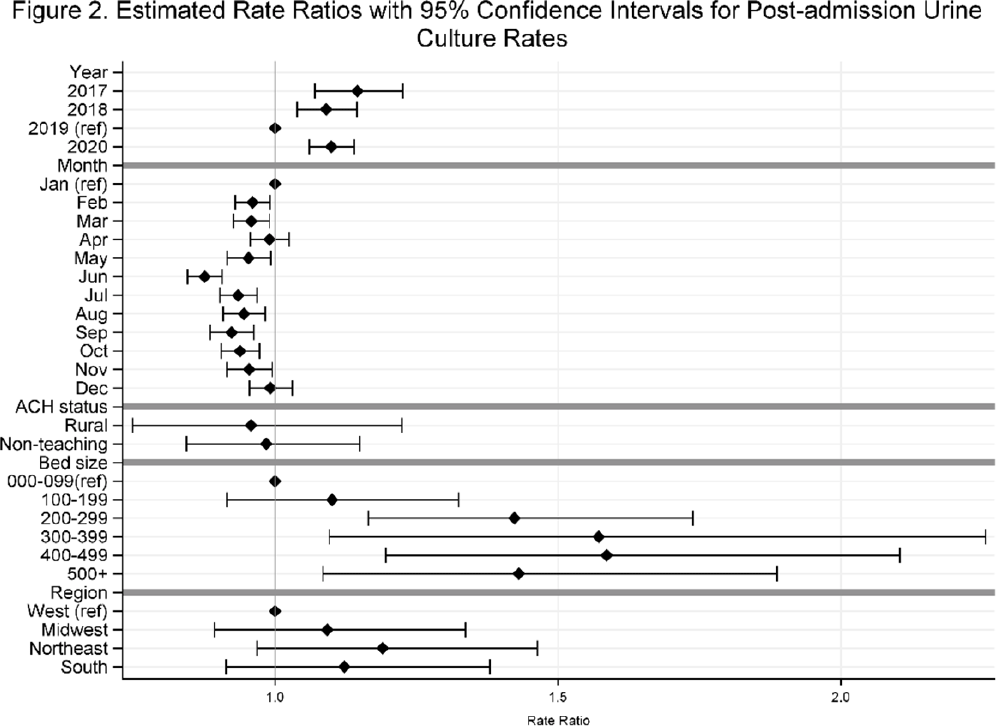
Temporal trends in urine-culture rates in the US acute-care hospitals, 2017–2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s12
-
- Article
-
- You have access
- Open access
- Export citation
Associations of facility-level antibiotic use and hospital-onset Clostridioides difficile infection in US acute-care hospitals, 2012–2018
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 8 / August 2022
- Published online by Cambridge University Press:
- 07 May 2021, pp. 1067-1069
- Print publication:
- August 2022
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Pediatric research priorities in healthcare-associated infections and antimicrobial stewardship
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 5 / May 2021
- Published online by Cambridge University Press:
- 26 November 2020, pp. 519-522
- Print publication:
- May 2021
-
- Article
- Export citation
Regional Impact of a CRE Intervention Targeting High Risk Postacute Care Facilities (Chicago PROTECT)
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s48-s49
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Burden and Trends of Hospital-Associated Community-Onset (HACO) Infections From Antibiotic Resistant and Nonresistant Bacteria
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s145
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Substance Use Diagnoses Among Persons with Community-Onset Methicillin-Resistant Staphylococcus aureus Bloodstream Infections
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s392-s393
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Evaluation of Care Interactions Between Healthcare Personnel and Residents in Nursing Homes Across the United States
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s36-s38
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation