18 results
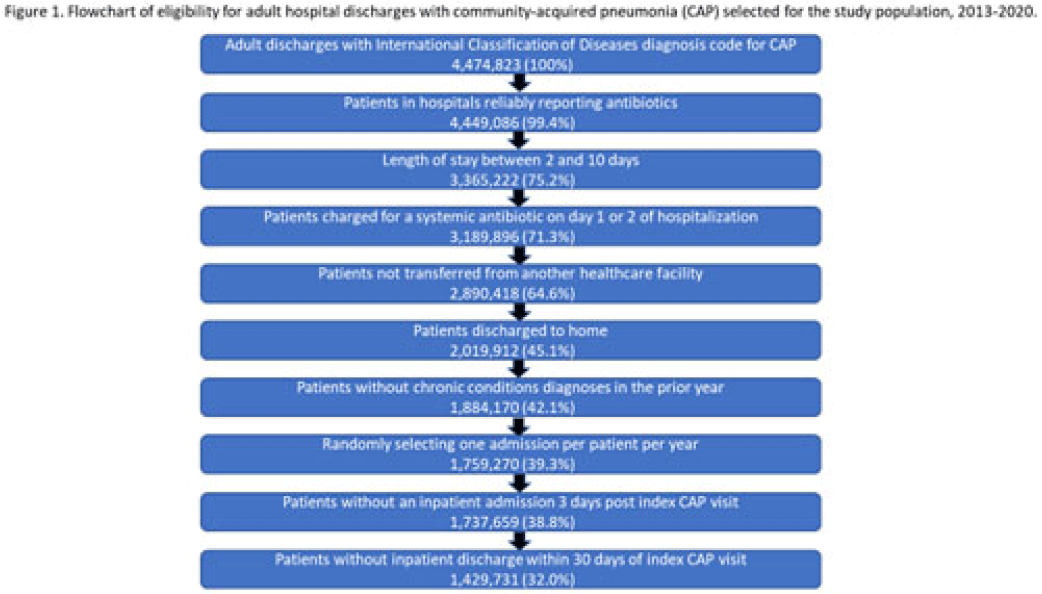
Length of antibiotic therapy among adults hospitalized with uncomplicated community-acquired pneumonia, 2013–2020
-
- Journal:
- Infection Control & Hospital Epidemiology , First View
- Published online by Cambridge University Press:
- 14 February 2024, pp. 1-7
-
- Article
- Export citation
Changes in US long-term care facility antibiotic prescribing, 2013–2021
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s108-s109
-
- Article
-
- You have access
- Open access
- Export citation
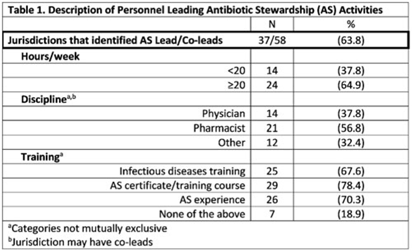
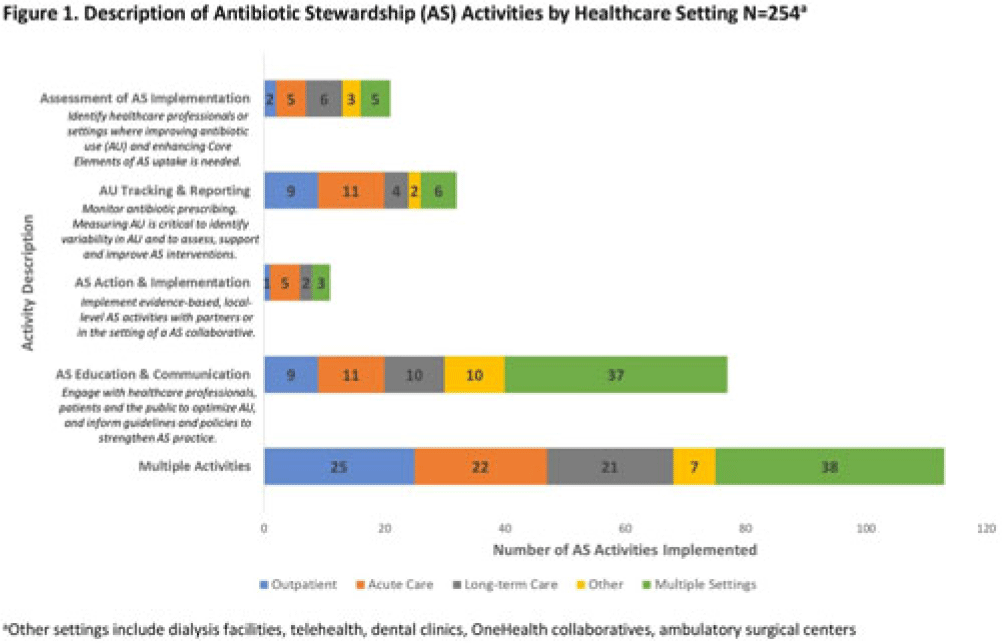
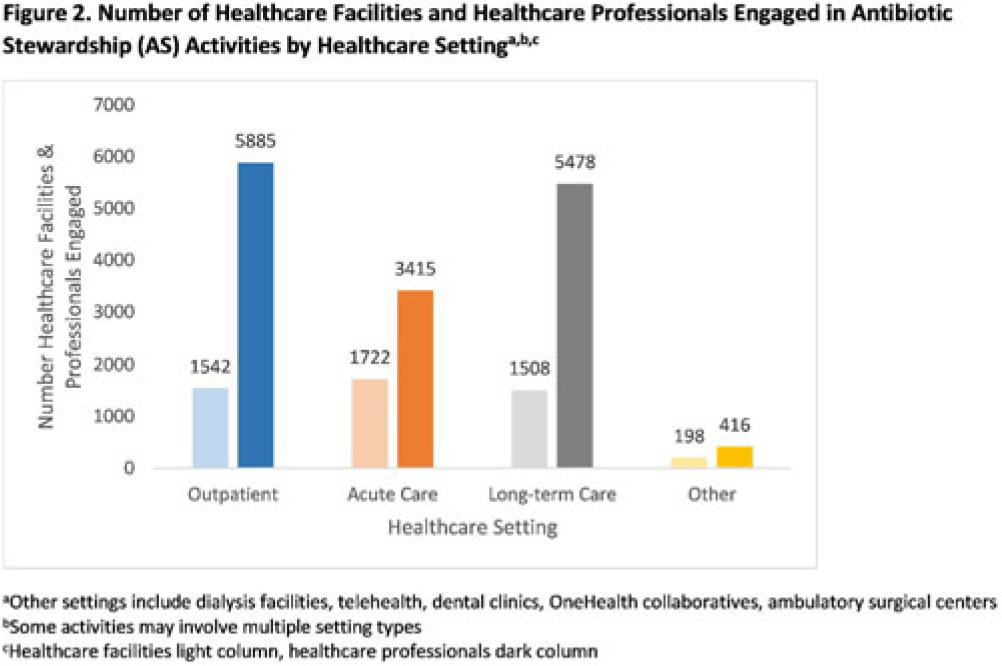
Description of antibiotic stewardship expertise and activities among US public health departments, 2022
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s3
-
- Article
-
- You have access
- Open access
- Export citation
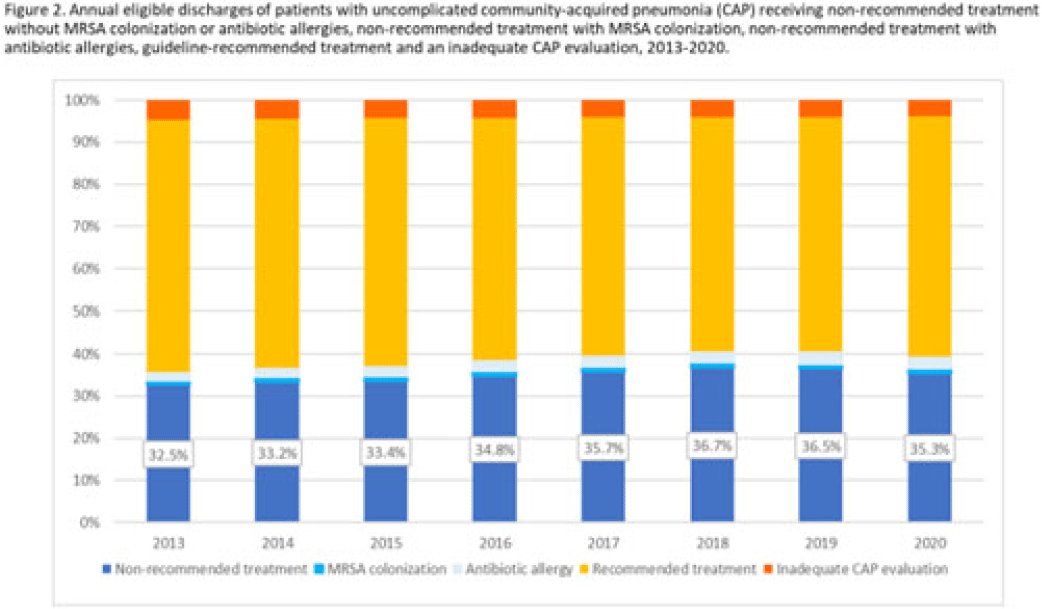
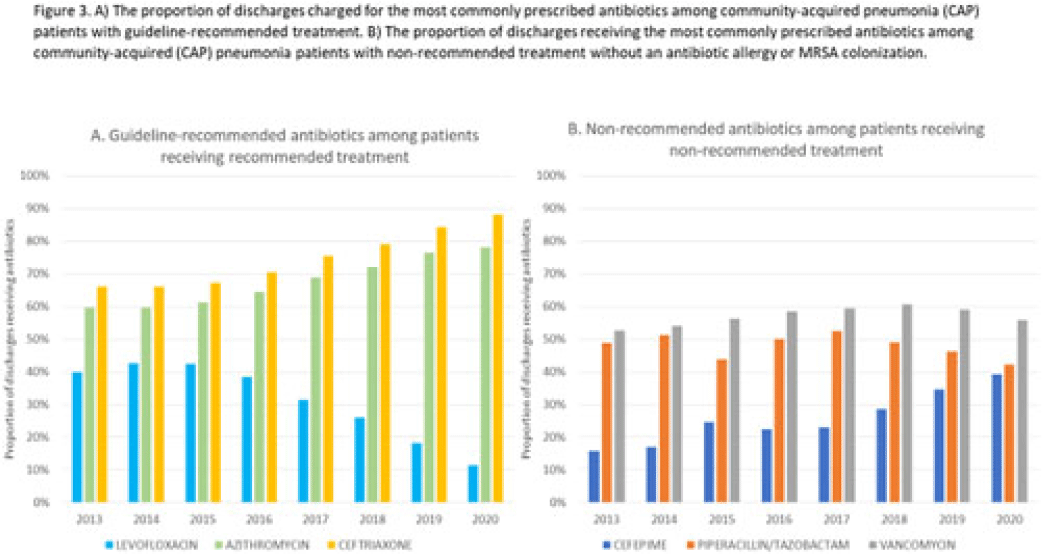
Empiric antibiotic selection for community-acquired pneumonia in US hospitals, 2013–2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s26-s27
-
- Article
-
- You have access
- Open access
- Export citation
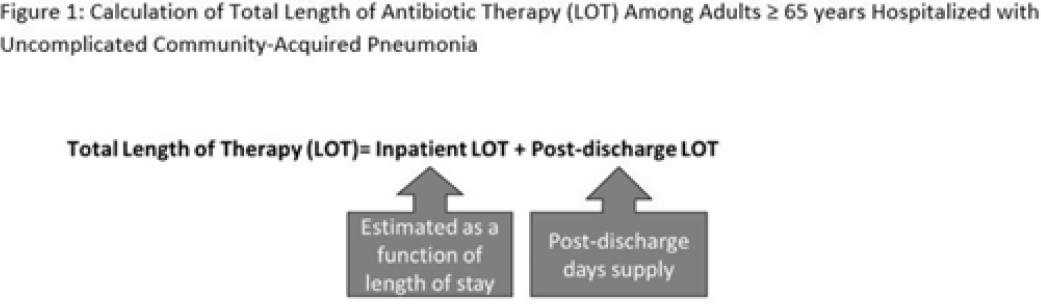
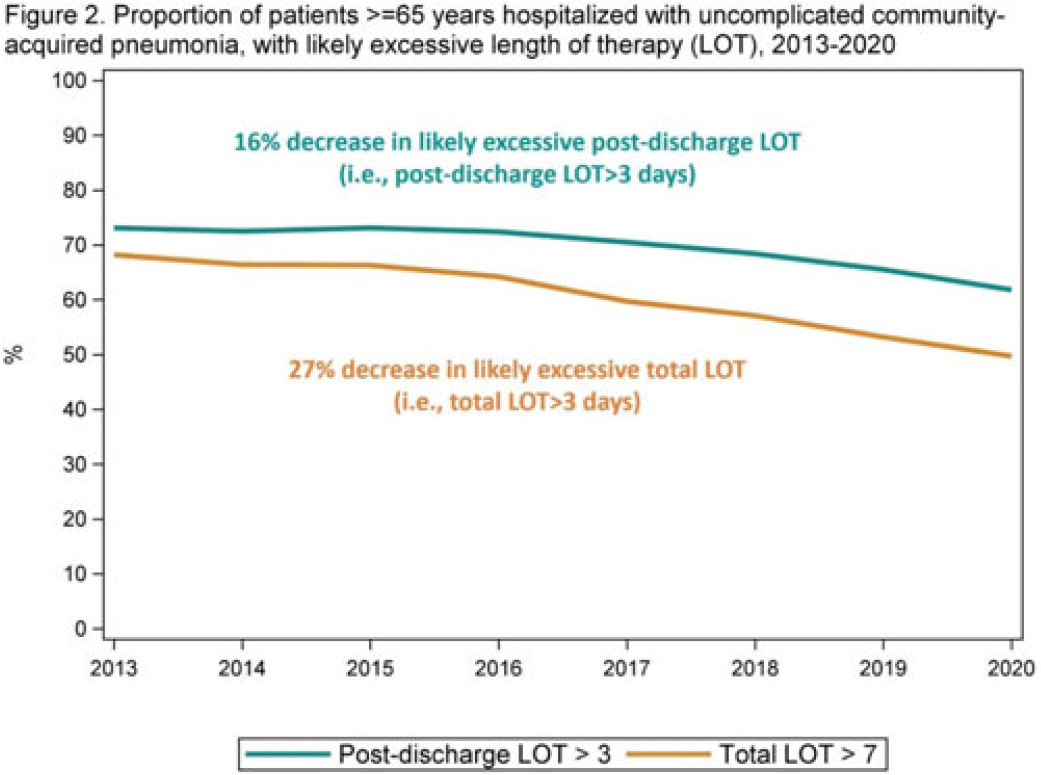
Length of antibiotic therapy among adults aged ≥65 years hospitalized with uncomplicated community-acquired pneumonia, 2013-2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s26
-
- Article
-
- You have access
- Open access
- Export citation
Characteristics of patients associated with any outpatient antibiotic prescribing among Medicare Part D enrollees, 2007–2018
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 29 June 2023, e113
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
The perfect storm: respiratory viral surges and anti-infective shortages
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 04 May 2023, e89
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Comparison of outpatient antibiotic prescriptions among older adults in IQVIA Xponent and publicly available Medicare Part D data, 2018
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 15 February 2023, e32
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Urgent-care antibiotic prescribing: An exploratory analysis to evaluate health inequities
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 14 November 2022, e184
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
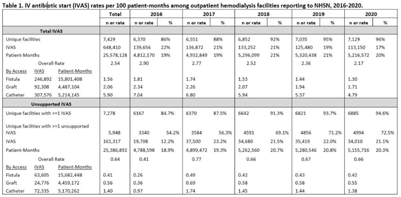
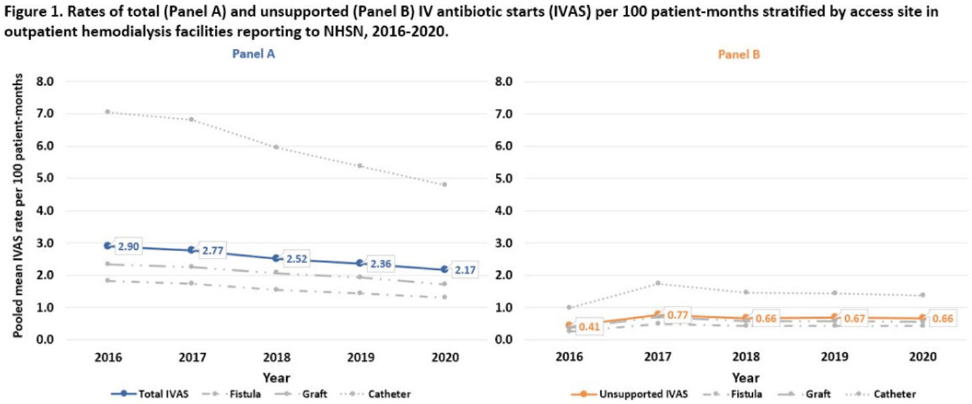
Rates of intravenous antibiotic starts among outpatient hemodialysis patients using NHSN dialysis event reporting, 2016–2020
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s71-s72
-
- Article
-
- You have access
- Open access
- Export citation
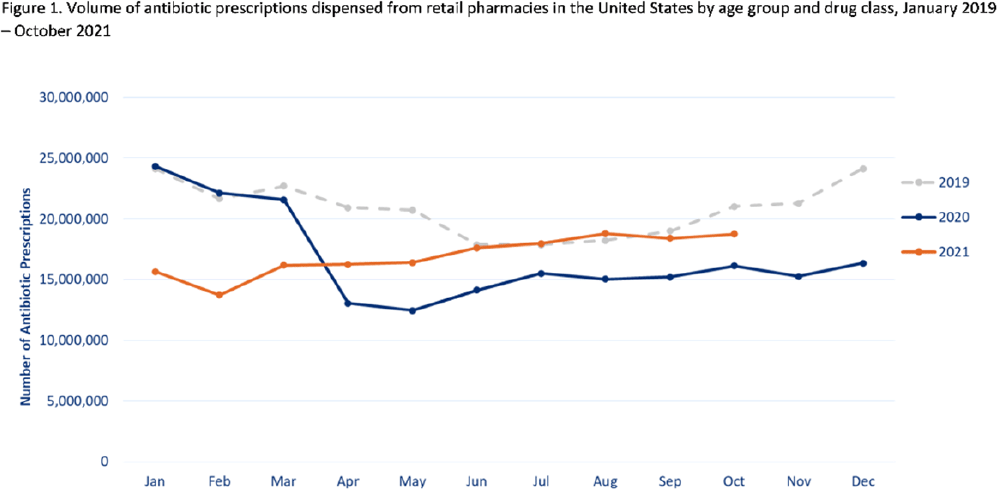
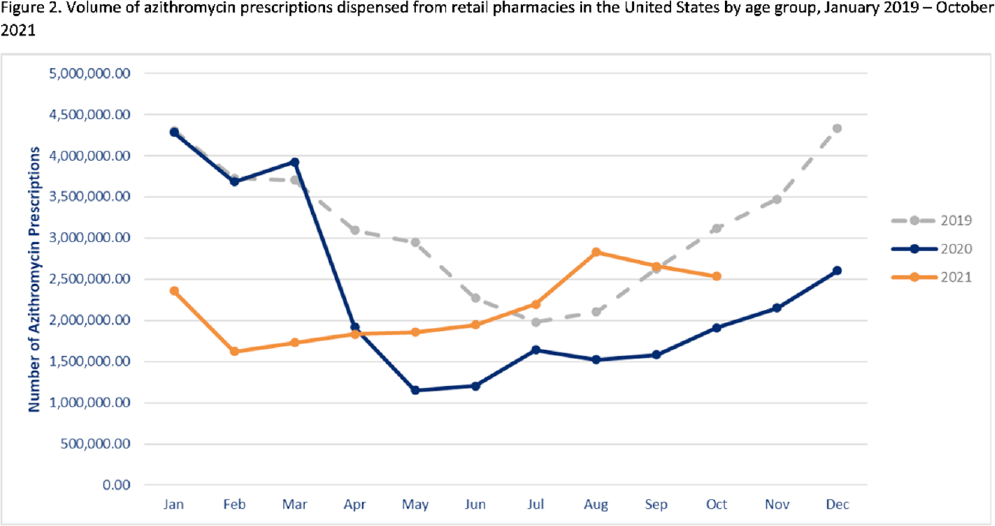
Outpatient antibiotic prescribing during the COVID-19 pandemic–United States, January 2019–October 2021
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s2-s3
-
- Article
-
- You have access
- Open access
- Export citation
Using machine learning to examine drivers of inappropriate outpatient antibiotic prescribing in acute respiratory illnesses
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 5 / May 2023
- Published online by Cambridge University Press:
- 10 January 2022, pp. 786-790
- Print publication:
- May 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Description of antibiotic use variability among US nursing homes using electronic health record data
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue 1 / 2021
- Published online by Cambridge University Press:
- 07 December 2021, e58
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
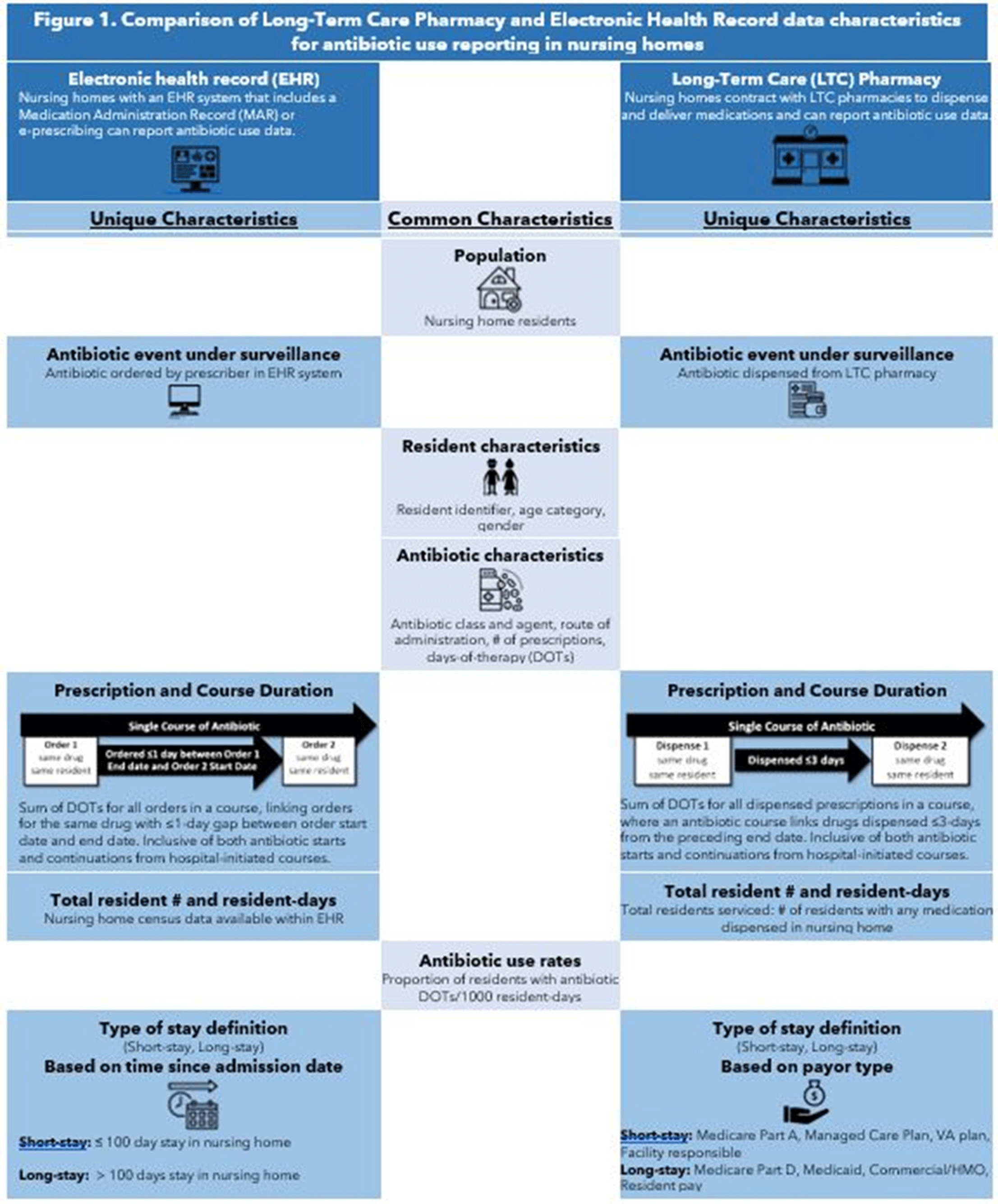
Evaluation of Electronic Health Record and Long-Term Care Pharmacy Data for Tracking and Reporting Antibiotic Use in the United States
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s13-s14
-
- Article
-
- You have access
- Open access
- Export citation
Implementation of core elements of antibiotic stewardship in nursing homes—National Healthcare Safety Network, 2016–2018
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 6 / June 2022
- Published online by Cambridge University Press:
- 26 May 2021, pp. 752-756
- Print publication:
- June 2022
-
- Article
- Export citation
Urinary tract infection treatment practices in nursing homes reporting to the National Healthcare Safety Network, 2017
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 2 / February 2022
- Published online by Cambridge University Press:
- 12 March 2021, pp. 238-240
- Print publication:
- February 2022
-
- Article
- Export citation
Potential utility of pharmacy data to measure antibiotic use in nursing homes
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 40 / Issue 7 / July 2019
- Published online by Cambridge University Press:
- 24 April 2019, pp. 819-820
- Print publication:
- July 2019
-
- Article
- Export citation
Potential Impact of Antibiotic Stewardship Programs on Overall Antibiotic Use in Adult Acute-Care Hospitals in the United States
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 39 / Issue 3 / March 2018
- Published online by Cambridge University Press:
- 15 February 2018, pp. 373-376
- Print publication:
- March 2018
-
- Article
-
- You have access
- HTML
- Export citation