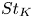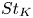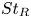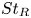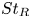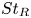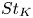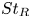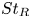95 results
Particle subgrid stress models for large Stokes numbers in particle-laden turbulence
-
- Journal:
- Journal of Fluid Mechanics / Volume 946 / 10 September 2022
- Published online by Cambridge University Press:
- 02 August 2022, A13
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Evaluation of pharyngeal swallowing pressure using high-resolution manometry during transoral surgery for oropharyngeal cancer
-
- Journal:
- The Journal of Laryngology & Otology / Volume 135 / Issue 2 / February 2021
- Published online by Cambridge University Press:
- 08 February 2021, pp. 153-158
- Print publication:
- February 2021
-
- Article
- Export citation
The Effects of Vanadium Substitution on One-dimensional Tunnel Structures of Cryptomelane: Combined TEM and DFT Study
-
- Journal:
- Microscopy and Microanalysis / Volume 26 / Issue S2 / August 2020
- Published online by Cambridge University Press:
- 30 July 2020, pp. 3162-3164
- Print publication:
- August 2020
-
- Article
-
- You have access
- Export citation
Epidemiology of hospitalised paediatric community-acquired pneumonia and bacterial pneumonia following the introduction of 13-valent pneumococcal conjugate vaccine in the national immunisation programme in Japan
-
- Journal:
- Epidemiology & Infection / Volume 148 / 2020
- Published online by Cambridge University Press:
- 17 April 2020, e91
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Elucidating the evolution of silicon anodes in lithium based batteries
-
- Journal:
- MRS Advances / Volume 5 / Issue 48-49 / 2020
- Published online by Cambridge University Press:
- 17 July 2020, pp. 2525-2534
- Print publication:
- 2020
-
- Article
- Export citation
Dust evolution in galaxies at z > 7
-
- Journal:
- Proceedings of the International Astronomical Union / Volume 15 / Issue S341 / November 2019
- Published online by Cambridge University Press:
- 10 June 2020, pp. 312-313
- Print publication:
- November 2019
-
- Article
-
- You have access
- Export citation
A new galaxy Spectral Energy Distribution model with the evolution of dust consistent with chemical evolution
-
- Journal:
- Proceedings of the International Astronomical Union / Volume 15 / Issue S341 / November 2019
- Published online by Cambridge University Press:
- 10 June 2020, pp. 152-156
- Print publication:
- November 2019
-
- Article
-
- You have access
- Export citation
Detections of far-infrared [OIII] and dust emission in a galaxy at z = 8.312: Early metal enrichment in the heart of the reionization era
-
- Journal:
- Proceedings of the International Astronomical Union / Volume 15 / Issue S341 / November 2019
- Published online by Cambridge University Press:
- 10 June 2020, pp. 211-215
- Print publication:
- November 2019
-
- Article
-
- You have access
- Export citation
Interface effects on self-forming rechargeable Li/I2-based solid state batteries
-
- Journal:
- MRS Communications / Volume 9 / Issue 2 / June 2019
- Published online by Cambridge University Press:
- 26 April 2019, pp. 657-662
- Print publication:
- June 2019
-
- Article
- Export citation
PROGRESS AND OUTLOOK ON FEW COMPONENT COMPOSITE SOLID STATE ELECTROLYTES
-
- Journal:
- MRS Advances / Volume 4 / Issue 49 / 2019
- Published online by Cambridge University Press:
- 11 June 2019, pp. 2635-2640
- Print publication:
- 2019
-
- Article
- Export citation
Efficacy of fluoroscopy-guided endoscopic cricopharyngeal myotomy
-
- Journal:
- The Journal of Laryngology & Otology / Volume 132 / Issue 12 / December 2018
- Published online by Cambridge University Press:
- 24 January 2019, pp. 1128-1133
- Print publication:
- December 2018
-
- Article
- Export citation
Detection of modified measles and super-spreader using a real-time reverse transcription PCR in the largest measles outbreak, Yamagata, Japan, 2017 in its elimination era
-
- Journal:
- Epidemiology & Infection / Volume 146 / Issue 13 / October 2018
- Published online by Cambridge University Press:
- 07 August 2018, pp. 1707-1713
-
- Article
-
- You have access
- HTML
- Export citation
Voltage Contrast Imaging with Energy-Controlled Signal in an FE-SEM.
-
- Journal:
- Microscopy and Microanalysis / Volume 24 / Issue S1 / August 2018
- Published online by Cambridge University Press:
- 01 August 2018, pp. 662-663
- Print publication:
- August 2018
-
- Article
-
- You have access
- Export citation
In-situ Probe of Lithium-ion Transport and Phase Evolution Within and Between Silver Hollandite Nanorods
-
- Journal:
- Microscopy and Microanalysis / Volume 24 / Issue S1 / August 2018
- Published online by Cambridge University Press:
- 01 August 2018, pp. 1516-1517
- Print publication:
- August 2018
-
- Article
-
- You have access
- Export citation
The Importance of Combined Spatio-Temporal Characterization: From in situ to operando Diffraction Measurements of Li/Li1.1V3O8 Batteries
-
- Journal:
- Microscopy and Microanalysis / Volume 24 / Issue S1 / August 2018
- Published online by Cambridge University Press:
- 01 August 2018, pp. 1478-1479
- Print publication:
- August 2018
-
- Article
-
- You have access
- Export citation
Insights into Structural Evolution of a Solid Electrolyte Interphase Using Thin Window Si Membrane Negative Electrodes
-
- Journal:
- Microscopy and Microanalysis / Volume 24 / Issue S1 / August 2018
- Published online by Cambridge University Press:
- 01 August 2018, pp. 1480-1481
- Print publication:
- August 2018
-
- Article
-
- You have access
- Export citation
Atomic Scale Analyses of Planar Defects in Cross-section Nanorods of K+ Stabilized a-MnO2
-
- Journal:
- Microscopy and Microanalysis / Volume 24 / Issue S1 / August 2018
- Published online by Cambridge University Press:
- 01 August 2018, pp. 130-131
- Print publication:
- August 2018
-
- Article
-
- You have access
- Export citation
Revealing the Surface Effect at Atomic Scale in Silver Hollandite
-
- Journal:
- Microscopy and Microanalysis / Volume 24 / Issue S1 / August 2018
- Published online by Cambridge University Press:
- 01 August 2018, pp. 56-57
- Print publication:
- August 2018
-
- Article
-
- You have access
- Export citation
Material Design Strategies to Achieve Simultaneous High Power and High Energy Density
-
- Journal:
- MRS Advances / Volume 3 / Issue 22 / 2018
- Published online by Cambridge University Press:
- 02 April 2018, pp. 1269-1275
- Print publication:
- 2018
-
- Article
- Export citation
The Effect of Silver Ion Occupancy on Hollandite Lattice Structure
-
- Journal:
- MRS Advances / Volume 3 / Issue 10 / 2018
- Published online by Cambridge University Press:
- 26 February 2018, pp. 547-552
- Print publication:
- 2018
-
- Article
- Export citation