8 results
Characteristics of healthcare personnel with SARS-CoV-2 infection: 10 emerging infections program sites in the United States, April 2020–December 2021
-
- Journal:
- Infection Control & Hospital Epidemiology , First View
- Published online by Cambridge University Press:
- 21 May 2024, pp. 1-9
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Residential social vulnerability among healthcare personnel with and without severe acute respiratory coronavirus virus 2 (SARS-CoV-2) infection in Five US states, May–December 2020
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 1 / January 2024
- Published online by Cambridge University Press:
- 18 July 2023, pp. 82-88
- Print publication:
- January 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
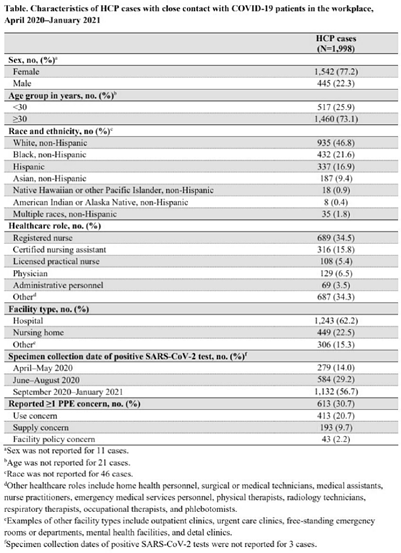
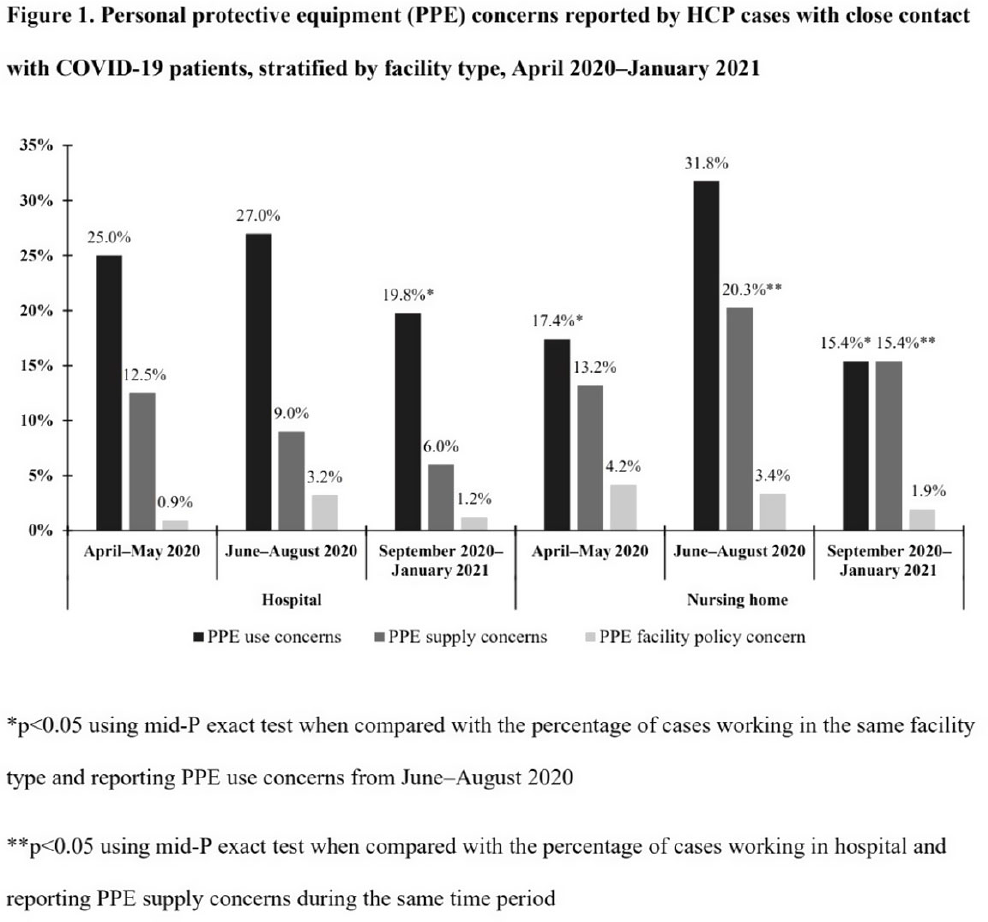
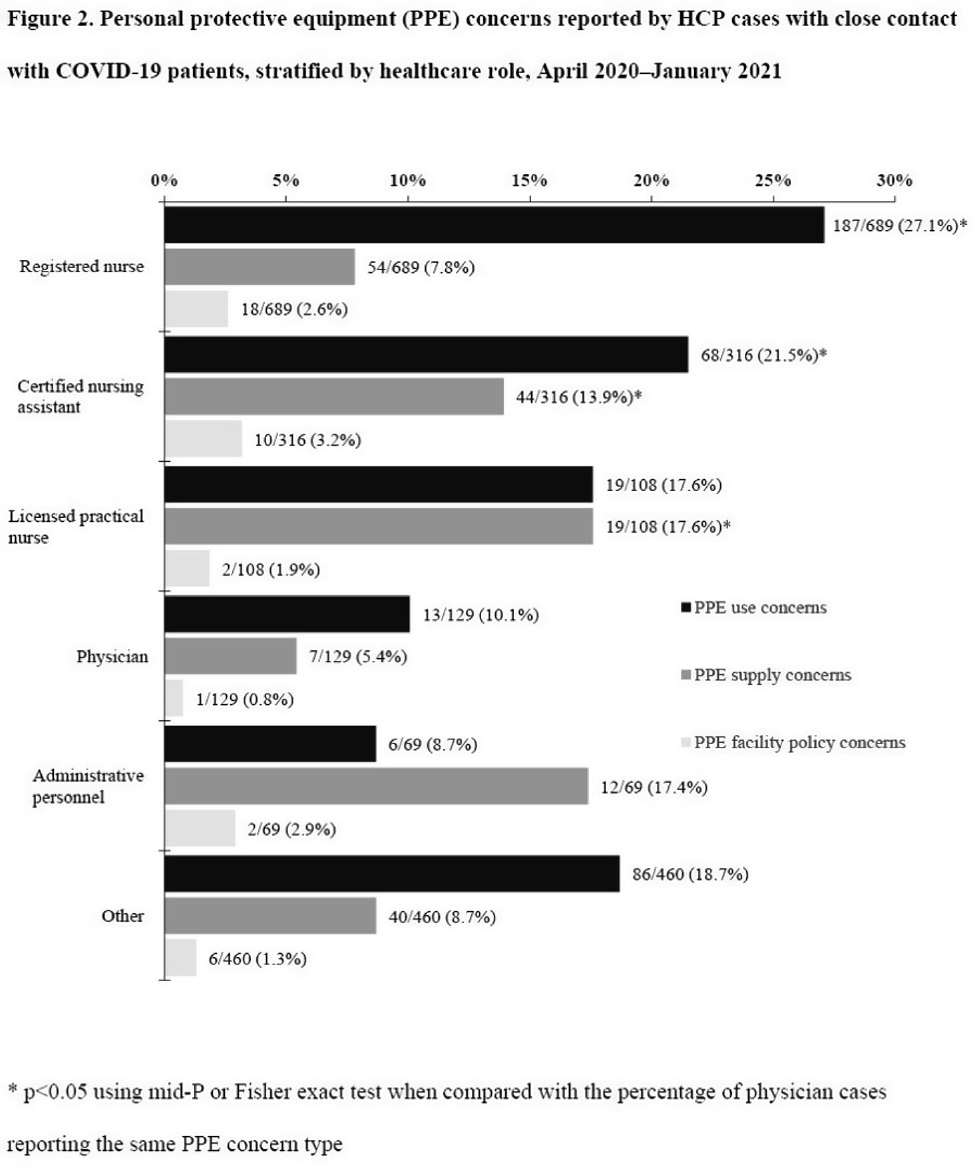
Characteristics of healthcare personnel who reported concerns related to PPE use during care of COVID-19 patients
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s8-s9
-
- Article
-
- You have access
- Open access
- Export citation
Occupational risk factors for severe acute respiratory coronavirus virus 2 (SARS-CoV-2) infection among healthcare personnel: A 6-month prospective analysis of the COVID-19 Prevention in Emory Healthcare Personnel (COPE) Study
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 11 / November 2022
- Published online by Cambridge University Press:
- 14 February 2022, pp. 1664-1671
- Print publication:
- November 2022
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Practices and activities among healthcare personnel with severe acute respiratory coronavirus virus 2 (SARS-CoV-2) infection working in different healthcare settings—ten Emerging Infections Program sites, April–November 2020
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 8 / August 2022
- Published online by Cambridge University Press:
- 02 June 2021, pp. 1058-1062
- Print publication:
- August 2022
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Occupational risk factors for severe acute respiratory coronavirus virus 2 (SARS-CoV-2) infection among healthcare personnel: A cross-sectional analysis of subjects enrolled in the COVID-19 Prevention in Emory Healthcare Personnel (COPE) study
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 3 / March 2022
- Published online by Cambridge University Press:
- 09 February 2021, pp. 381-386
- Print publication:
- March 2022
-
- Article
-
- You have access
- HTML
- Export citation
Incidence of Mucosal Barrier Injury Bloodstream Infections Reported to the National Healthcare Safety Network
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s293-s294
- Print publication:
- October 2020
-
- Article
-
- You have access
- Export citation
Pseudomonas aeruginosa Outbreak in a Neonatal Intensive Care Unit Attributed to Hospital Tap Water
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 38 / Issue 7 / July 2017
- Published online by Cambridge University Press:
- 18 May 2017, pp. 801-808
- Print publication:
- July 2017
-
- Article
- Export citation