33 results
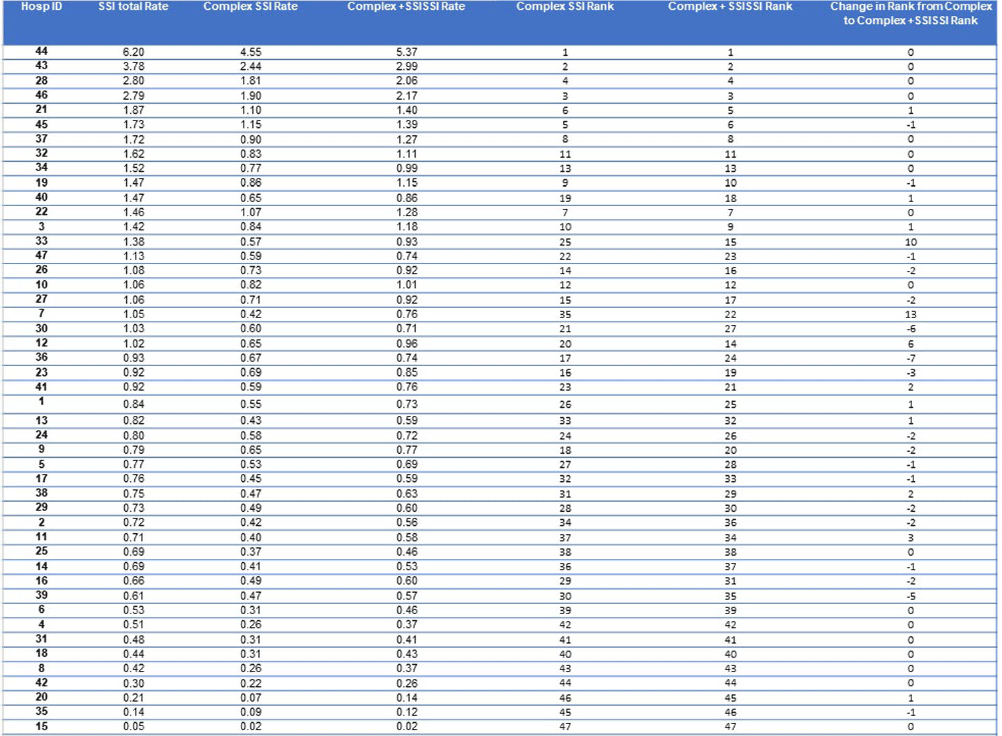
A Deeper Look at Proposed Surveillance of Superficial Incision Surgical Site Infections (SSISSIs)
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, p. s142
-
- Article
-
- You have access
- Open access
- Export citation
Sustainability of Surgical Site Infection (SSI) Prevention Bundle for Pediatric Cardiothoracic Surgery Patients
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s145-s146
-
- Article
-
- You have access
- Open access
- Export citation
Comparative epidemiology of hospital-onset bloodstream infections (HOBSIs) and central line-associated bloodstream infections (CLABSIs) across a three-hospital health system
-
- Journal:
- Infection Control & Hospital Epidemiology , First View
- Published online by Cambridge University Press:
- 20 March 2024, pp. 1-7
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
The impact of minimally invasive surgical approaches on surgical-site infections
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 5 / May 2024
- Published online by Cambridge University Press:
- 03 January 2024, pp. 557-561
- Print publication:
- May 2024
-
- Article
-
- You have access
- HTML
- Export citation
A cluster of three extrapulmonary Mycobacterium abscessus infections linked to well-maintained water-based heater-cooler devices
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 5 / May 2024
- Published online by Cambridge University Press:
- 21 December 2023, pp. 644-650
- Print publication:
- May 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
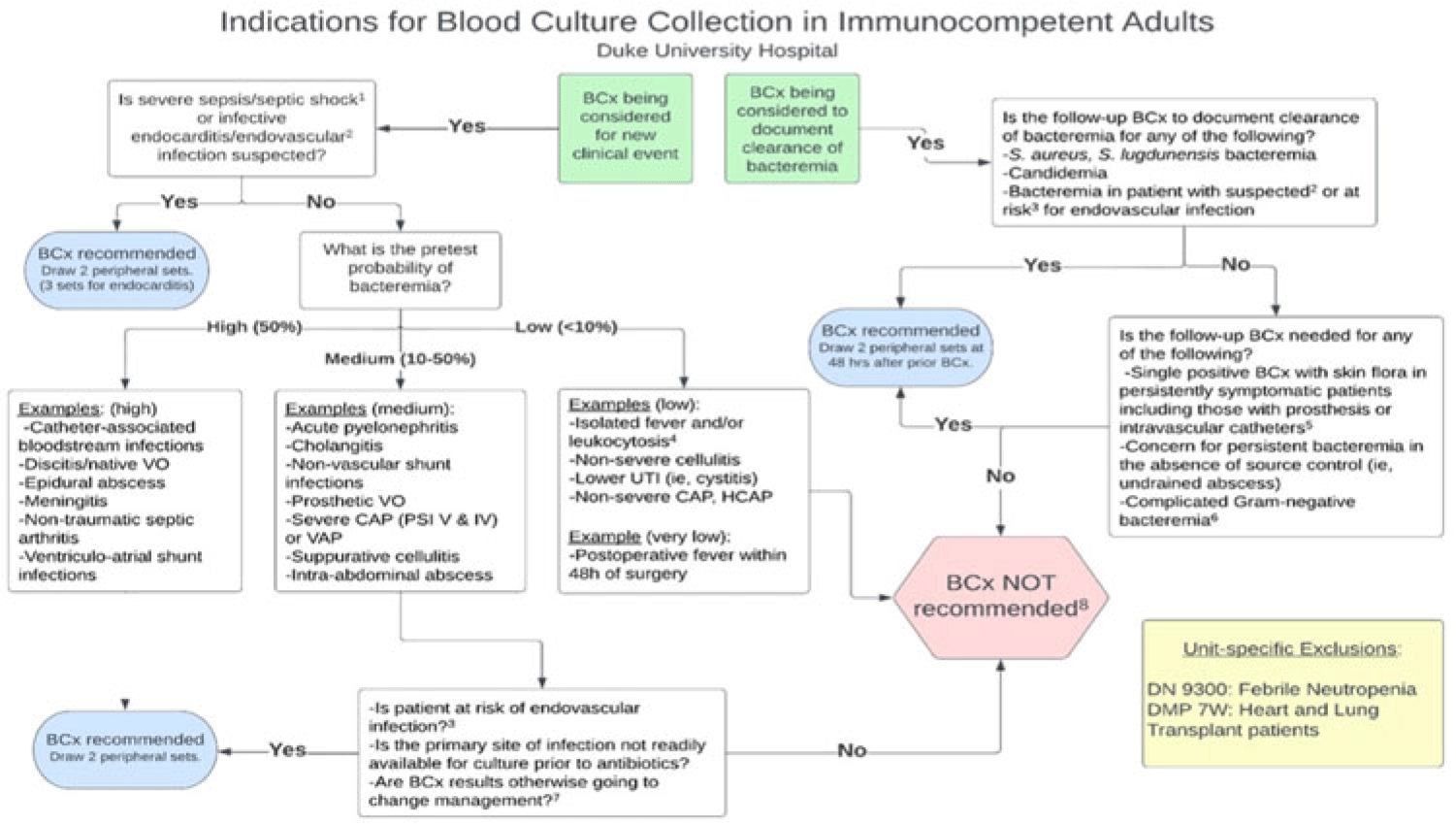
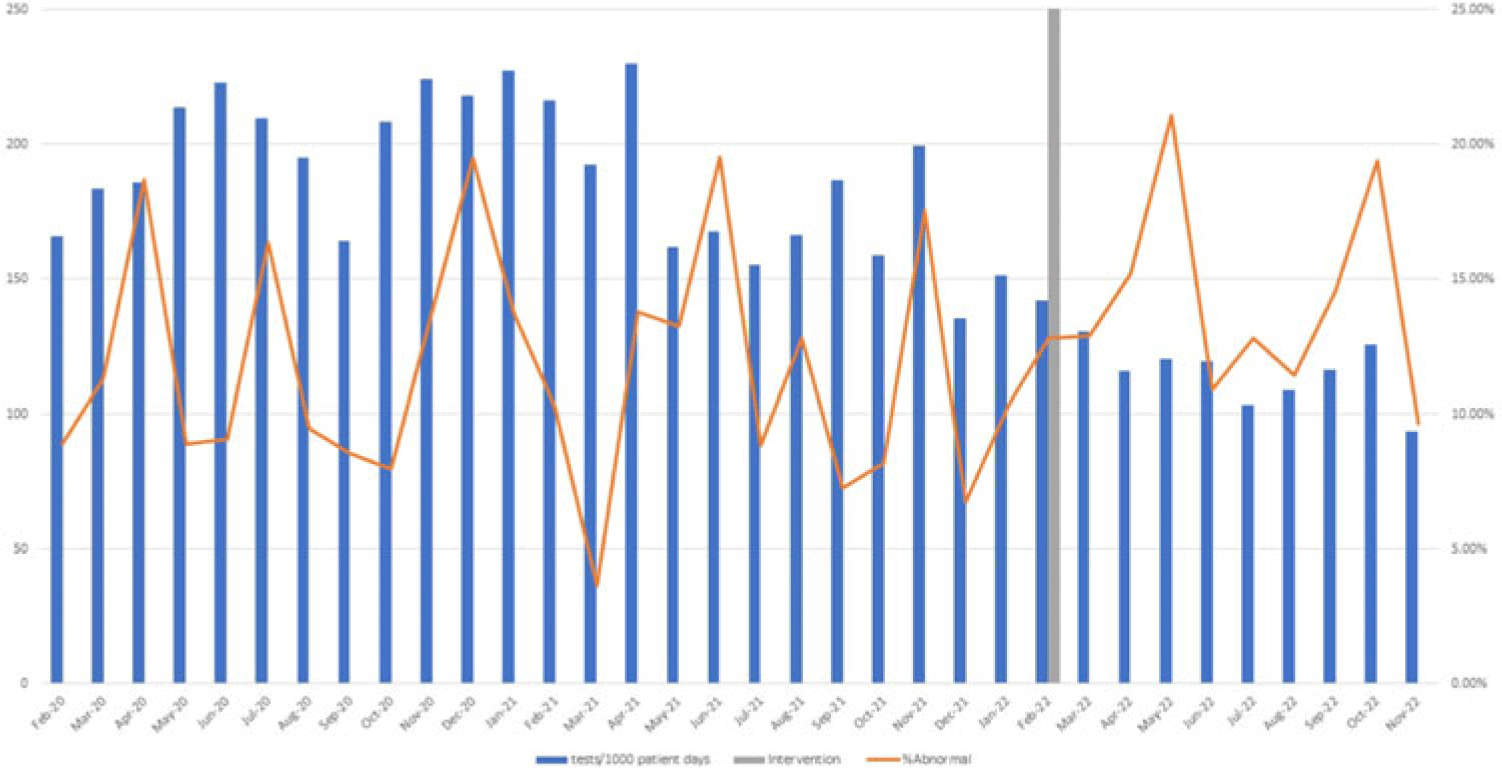
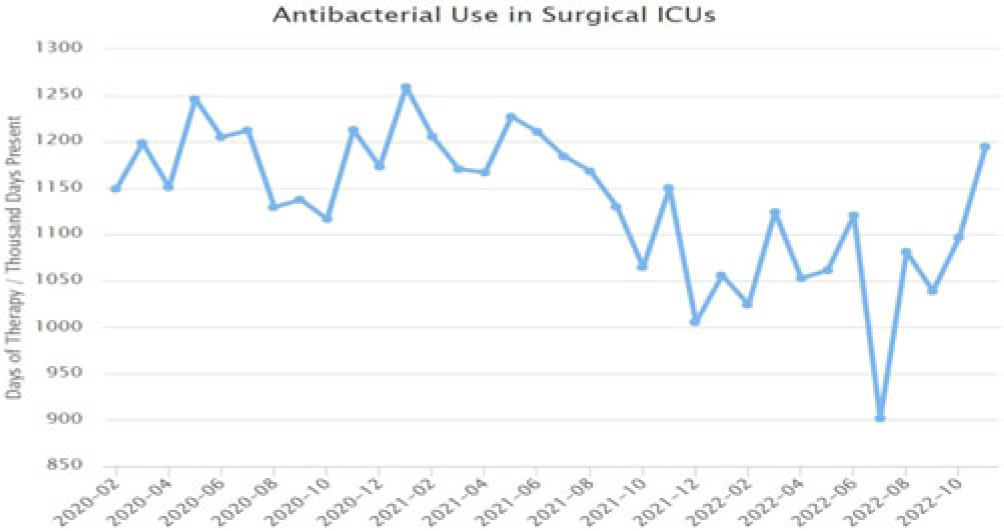
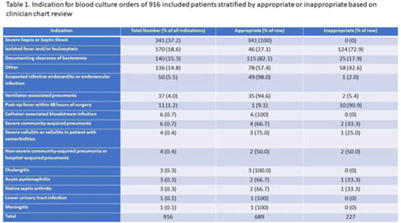
Implementation of a diagnostic stewardship intervention to improve blood-culture utilization in 2 surgical ICUs: Time for a blood-culture change
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 4 / April 2024
- Published online by Cambridge University Press:
- 11 December 2023, pp. 452-458
- Print publication:
- April 2024
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Implementation of diagnostic stewardship in two surgical ICUs: Time for a blood-culture change
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s9-s10
-
- Article
-
- You have access
- Open access
- Export citation
Using the COM-B model to identify barriers to and facilitators of evidence-based nurse urine-culture practices
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 31 March 2023, e62
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Using clinical decision support to improve urine testing and antibiotic utilization
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 10 / October 2023
- Published online by Cambridge University Press:
- 29 March 2023, pp. 1582-1586
- Print publication:
- October 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
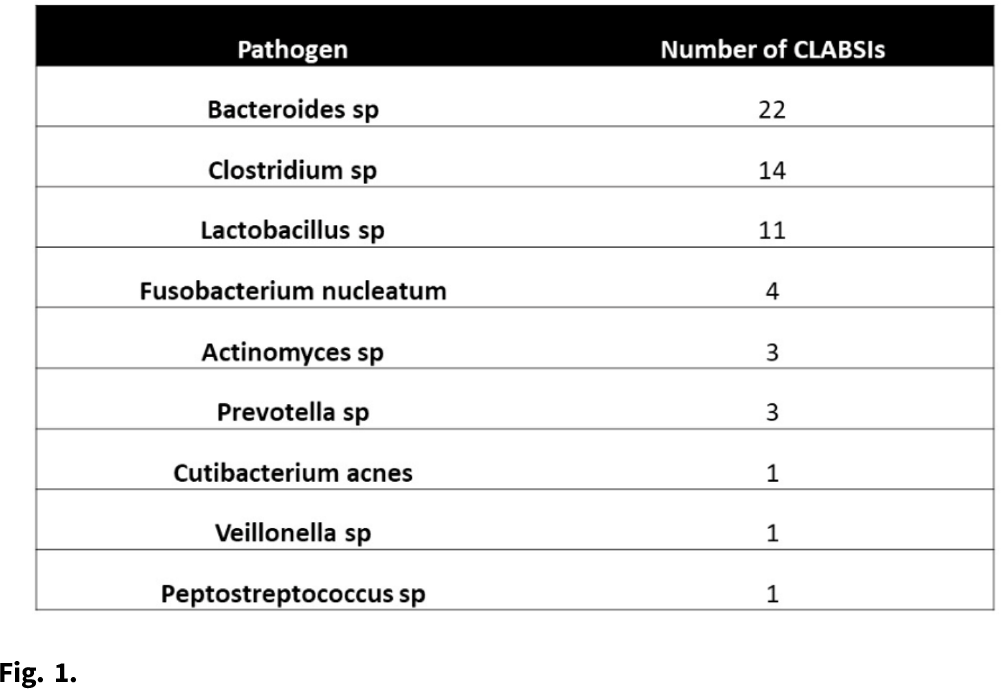
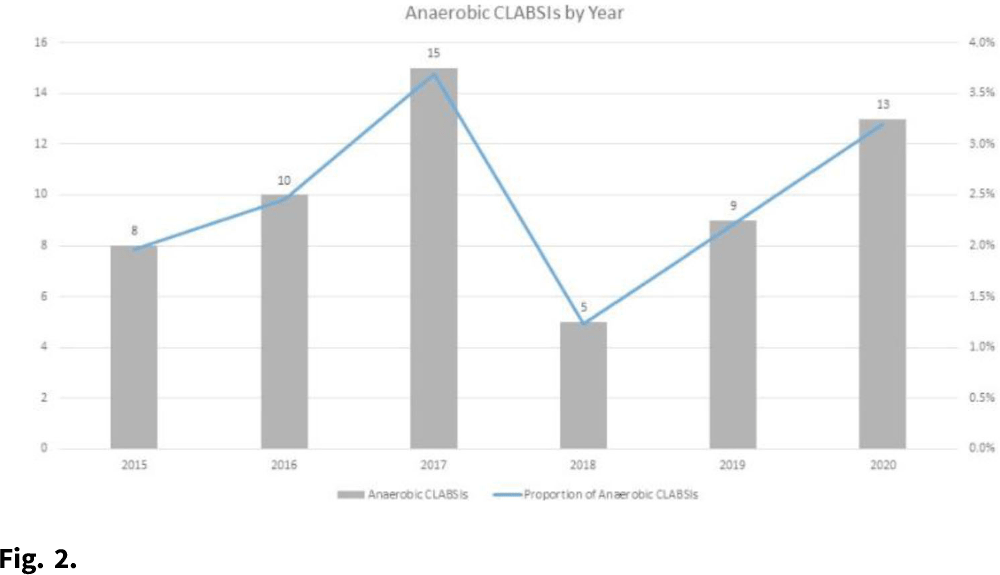
Central-line–associated bloodstream infections secondary to anaerobes: Time for a definition change?
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 10 / October 2023
- Published online by Cambridge University Press:
- 03 February 2023, pp. 1697-1698
- Print publication:
- October 2023
-
- Article
-
- You have access
- HTML
- Export citation
Colon surgical-site infections and the impact of “present at the time of surgery (PATOS)” in a large network of community hospitals
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 8 / August 2023
- Published online by Cambridge University Press:
- 22 September 2022, pp. 1255-1260
- Print publication:
- August 2023
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Surgical site infection trends in community hospitals from 2013 to 2018
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 4 / April 2023
- Published online by Cambridge University Press:
- 18 July 2022, pp. 610-615
- Print publication:
- April 2023
-
- Article
- Export citation
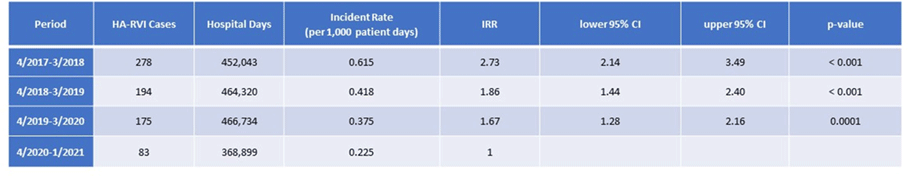
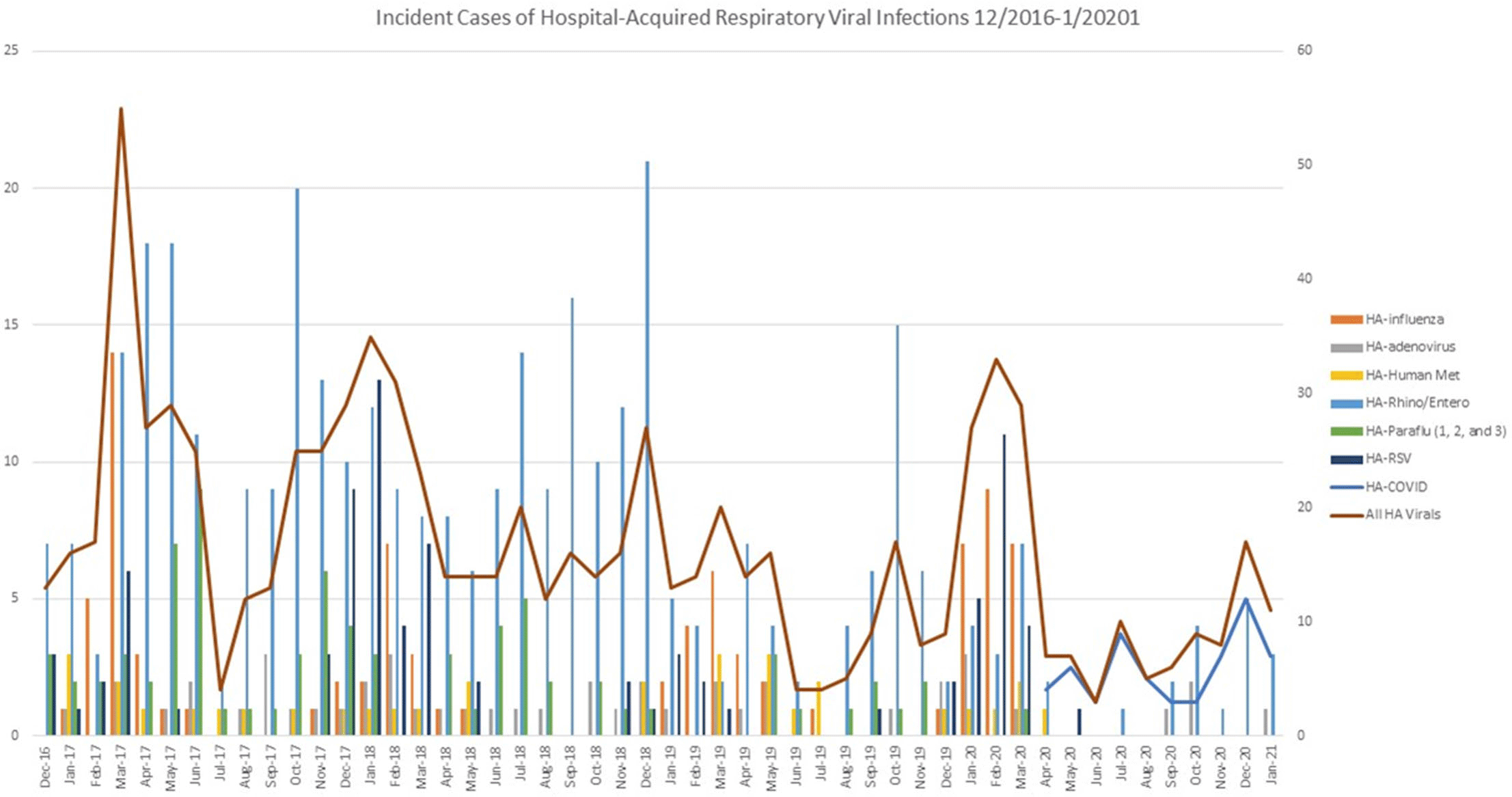
The impact of a comprehensive coronavirus disease 2019 (COVID-19) infection prevention bundle on non–COVID-19 hospital-acquired respiratory viral infection (HA-RVI) rates
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 6 / June 2023
- Published online by Cambridge University Press:
- 02 June 2022, pp. 1022-1024
- Print publication:
- June 2023
-
- Article
- Export citation
Central-line associated bloodstream infections secondary to strict anaerobes: Time for A definition change?
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s73-s74
-
- Article
-
- You have access
- Open access
- Export citation
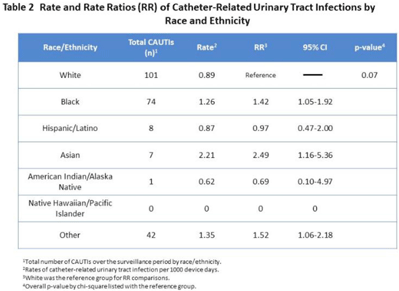
Racial disparities in rate of central-line–associated bloodstream infections and catheter-associated urinary tract infections
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s84-s85
-
- Article
-
- You have access
- Open access
- Export citation
An analysis of 90-day emergency department visits after peripherally inserted central catheter (PICC) placement for prosthetic joint infection
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 12 / December 2022
- Published online by Cambridge University Press:
- 07 March 2022, pp. 1853-1858
- Print publication:
- December 2022
-
- Article
- Export citation
Challenges in hospital-acquired coronavirus disease 2019 (COVID-19) surveillance and attribution of infection source
- Part of
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 12 / December 2022
- Published online by Cambridge University Press:
- 02 August 2021, pp. 1914-1917
- Print publication:
- December 2022
-
- Article
- Export citation
Impact of a Comprehensive SARS-CoV-2 Infection Prevention Bundle on Rates of Hospital-Acquired Respiratory Viral Infections
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s81
-
- Article
-
- You have access
- Open access
- Export citation
Comparison of metrics used to track central-line–associated bloodstream infections (CLABSIs) and catheter-associated urinary tract infections (CAUTIs) across a regional network
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 11 / November 2022
- Published online by Cambridge University Press:
- 16 July 2021, pp. 1712-1714
- Print publication:
- November 2022
-
- Article
- Export citation
The impact of patient-reported penicillin or cephalosporin allergy on surgical site infections
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 7 / July 2022
- Published online by Cambridge University Press:
- 09 June 2021, pp. 829-833
- Print publication:
- July 2022
-
- Article
- Export citation