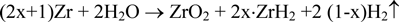Corrosion behavior is a key issue for the waste disposal of irradiated metals, such as hulls and endpieces, and is considered to be a leaching source of radionuclides including C-14. However, little information about Zircaloy corrosion in anticorrosive conditions has been provided.
In the present study, long-term corrosion tests of Zircaloy-4 and Zircaloy-2 were performed in assumed disposal conditions (dilute NaOH solution, pH 12.5, 303 K) by using the gas flow system for 1500 days. The corrosion rate, which was determined by measuring gaseous hydrogen and the hydrogen absorbed in Zircaloy, decreased with immersion time and was lower than the value of 2×10−2 μm/y used in performance assessment (1500-day values: 5.84×10−3 and 5.66×10−3 μm/y for Zircaloy-4, 1000-day values: 8.81×10−3 μm/y for Zircaloy-2). The difference in corrosion behavior between Zircaloy 4 and Zircaloy-2 was negligible. The average values of the hydrogen absorption ratios for Zircaloy-4 and Zircaloy-2 during corrosion were 91% and 94%, respectively.
The hydrogen generation kinetics of both gas evolution and absorption into metal can be shown by a parabolic curve. This result indicates that the diffusion process controls the Zircaloy corrosion in the early corrosion stage of the present study, and that the thickness of the oxide film in this stage is limited to approximately 25 nm and may therefore be in the form of dense tetragonal zirconia.