A better therapeutic relationship in the treatment of psychosis predicts better treatment adherence, less severe symptoms, better social functioning and fewer hospital admissions. Reference Priebe, Dimic, Wildgrube, Jankovic, Cushing and McCabe1–Reference Priebe, Richardson, Cooney, Adedeji and McCabe4 A meta-analysis found the odds of a patient adhering to treatment to be 2.16 times greater if there is a good doctor–patient relationship. Reference Zolnierek, Matteo and Robin5 However, although patients rate the therapeutic relationship as the most important component of psychiatric care, Reference Johansson and Eklund6 currently there are no trial-based interventions for how psychiatrists can improve the therapeutic relationship.
The psychiatrist–patient therapeutic relationship is negotiated in psychiatrist–patient communication, and psychiatrists consider effective communication skills to be one of the most important characteristics of a good psychiatrist. Reference Priebe, Dimic, Wildgrube, Jankovic, Cushing and McCabe1,Reference Bhugra, Sivakumar, Holsgrove, Butler and Leese7 Hence, improving communication is central to improving the relationship. Using conversation analysis, a method increasingly applied to medicine which analyses what people do rather than what they say they do, Reference Drew, Chatwin and Collins8,Reference Heritage, Robinson, Elliott, Beckett and Wilkes9 previous research identified a lack of shared understanding in psychiatrist–patient communication in the treatment of psychosis. This often centred on exchanges about psychotic symptoms in the context of mental state assessment. Patients repeatedly attempted to discuss the content and emotional consequences of their hallucinations and delusions, whereas psychiatrists tended to avoid engaging with these concerns in an attempt to avoid disagreement. This led to patients asking direct questions about these experiences (e.g. ‘Why don't people believe me?’, ‘Do you believe me?’) in an attempt to establish a shared understanding. Reference McCabe, Heath, Burns and Priebe10
One specific index of good communication is ‘self-repair’, explained in detail elsewhere. Reference Schegloff, Jefferson and Sacks11,Reference Hayashi, Raymond and Sidnell12 This is a conversation analytic term, which reflects attempts to achieve shared understanding. It refers to the ‘online’ processes of editing or reworking an utterance while it is being produced. Self-repairs are ubiquitous in natural, unscripted dialogue and have proved to be a useful measure of how hard people are working to make their talk understandable and acceptable to the listener in conversation generally and in psychiatric encounters. Reference Brennan and Schober13,Reference Themistocleous, McCabe, Rees, Hassan, Healey and Priebe14 For example, in the following excerpt, the psychiatrist asks the patient about reducing or stopping his procyclidine, reformulating the utterance as he produces it.
I mean [1] what if we, ah erhhh [2] what would your thoughts be about – [3] what if I said to you well we should look about reducing them or stopping them, what would you think about that?
His first formulation ‘what if we’ is abandoned and reworked as ‘what would your thoughts be about’. This is reworked again as ‘what if I said to you well we should look about reducing or stopping them?’. The final formulation presents a hypothetical proposal for the patient to consider, indicating that the subsequent action will take the patient's position into account. In a previous observational study of psychiatrist–patient communication, more psychiatrist self-repair was associated with a better patient-rated therapeutic relationship (details available from the author on request). In non-medical interaction, self-repair has also been identified as an index of effort by conversational partners in sustaining mutual intelligibility in dialogue. Reference Colman, Healey, Carlson and Shipley15
Currently, mental health professionals receive little specific training, beyond basic communication and counselling skills, in how to communicate effectively with patients with psychosis. We developed a brief training programme for psychiatrists based on research findings that psychiatrist–patient shared understanding – assessed by means of psychiatrist ‘self-repair’ – is associated with better relationships. Reference Themistocleous, McCabe, Rees, Hassan, Healey and Priebe14 The training was novel as it was developed from micro-analysis of psychiatrist–patient communication in previously recorded routine psychiatric encounters. It focused on the challenges of communicating in this therapeutic context along with effective ways of overcoming these challenges and empowering patients. This trial tested whether the novel training programme would increase psychiatrist self-repair and improve therapeutic relationships.
Method
Study design
This was a cluster randomised control trial (trial registration: ISRCTN94846422). Psychiatrists were randomised to the training or control group. Data were collected from psychiatrists and their patients at two time points: baseline (i.e. before psychiatrists received the training) and follow-up (i.e. at the first out-patient encounter after the training for each psychiatrist–patient pair (about 5 months after baseline)). At baseline and follow-up, encounters between participating psychiatrists and their patients were video-recorded in the clinic as usual. Researchers set up the camera and left the room. Psychiatrists and patients completed questionnaires.
Sample size
The target sample size was 72 patients (and 12 psychiatrists), i.e. 36 patients in each group, providing 85% power at the 5% significance level to detect an effect size of 1 (a doubling in the rate of repair) based on an assumed correlation between pre- and post-training measures of 0.7 and an intracluster correlation coefficient (ICC) of 0.65 in a previous study. Reference McCabe, Healey, Priebe, Lavelle, Dodwell and Laugharne16 As there was more psychiatrist turnover than anticipated during recruitment, additional participants were recruited so that the final sample size was 97 patients and 21 psychiatrists.
Participants
Recruitment took place between September 2011 and October 2012. Psychiatrist inclusion criteria were: specialist psychiatric trainees working in out-patient clinics or community mental health teams. Psychiatrists working at this level have basic knowledge and experience in psychiatry gained through at least 3 years' core psychiatric training and practice without direct supervision.
Patient inclusion criteria were: adults aged 18–65; meeting ICD-10 criteria for a diagnosis of schizophrenia or schizoaffective disorder; 17 currently attending psychiatric out-patient clinics or being cared for by community mental health teams; and capable of giving informed consent. Exclusion criteria were: organic impairment or an encounter requiring an interpreter.
Intervention
The training was developed over 1 year by specialists in communication in psychiatry (R.M., D.K. and P.J.) and general medicine (A.C.) with input from patients. It was fully manualised (available in print/DVD on request or online, http://medicine.exeter.ac.uk/media/universityofexeter/medicalschool/profiles/TEMPO_full_manual.pdf). Four sessions focused on: (a) understanding the patient with psychotic experiences: reflecting on the patient's experience and the professional and emotional response to psychotic symptoms; (2) communication techniques for working with positive and negative symptoms; (3) empowering the patient: agenda-setting at the start of the meeting and explaining/normalising psychosis; and (4) involvement in decision-making about medication. Reference McCabe, Khanom, Bailey and Priebe2
The programme was based on previous research highlighting: engaging with the patient to acknowledge their distressing experience without an underlying goal of changing the patient's beliefs; Reference McCabe, Heath, Burns and Priebe10,Reference Federico, Priebe, Fusco, Strapelli, Singh and McCabe18 negative symptoms as protective and working with patients with long-standing negative symptoms to set their own, albeit small, treatment goals; Reference Kingdon and Turkington19 and involving the patient in decisions about treatment. Reference McCabe, Khanom, Bailey and Priebe2
The training was administered in four consecutive weekly group sessions lasting 3 h each and one individualised feedback session where participants reflected on their video-recorded communication with patients in the clinic. Each session was run by two facilitators (R.M., A.C., D.K. or P.B.). The weekly interval facilitated practising new skills with different patients and feeding back positive and negative experiences in the next session. The sessions were delivered to groups of up to nine participants. Each session included transcripts and video clips of each topic (e.g. delusions, agenda-setting, decision-making) in previously recorded psychiatrist–patient encounters using high levels of self-repair for each topic. Reference Priebe, Dimic, Wildgrube, Jankovic, Cushing and McCabe1,Reference McCabe, Khanom, Bailey and Priebe2,Reference McCabe, Heath, Burns and Priebe10,Reference Federico, Priebe, Fusco, Strapelli, Singh and McCabe18 Clips of excerpts, previously micro-analysed using conversation analysis, were played and then stopped to ask participants how they would respond to a particular patient utterance. This stimulated group discussions reflecting on alternative ways of communicating. This was followed by role-play, trying out new ways of communicating with each other and with simulated patients (professional actors) along with the use of real-time video feedback. The role-plays used actual scenarios from video-recorded encounters, for example an exchange where a patient wants to come off medication but the psychiatrist does not support this.
In the first session, psychiatrists participated in a simulated ‘hearing voices exercise’. Reference Deegan20 Psychiatrists performed various tasks (e.g. a cognitive assessment) while listening to simulated voices. This exercise was highly rated by the participants, with most commenting on how distressing it was and that they now understood why patients feel a need to make sense of such experiences.
Control condition
Psychiatrists in the control condition did not receive the training and delivered treatment as usual.
Framework for evaluating the training
The framework for evaluating the training was that psychiatrists would feel more confident in communicating with patients with psychosis at the end of the training and apply the new communication skills – reflected behaviourally in increased use of self-repair – leading to improved therapeutic relationships from both psychiatrist and patient perspectives. Each of these outcomes was assessed.
Outcome measures
Primary outcome
Self-repair. The pre-determined primary outcome was psychiatrist self-repair in out-patient encounters with participating patients after the training, reflecting engagement with the patient and effort in establishing shared understanding. All pre- and post-training encounters were transcribed and self-repair was automatically annotated on the transcripts using a computer program STIR (STrongly Incremental Repair) to detect self-repair. STIR detects the presence and extent of self-repairs, including repetitions, substitutions and deletions by detecting key phrases that indicate repair (‘er’, ‘sorry’, ‘I mean’, etc.), and using statistical measures of fluency and likely sentential sequence. The STIR program has been validated in clinical and non-clinical data and with people for whom English is not a first language. Reference Howes, Hough, Purver and McCabe21,Reference Hough and Purver22 The accuracy (i.e. F-score) of the STIR algorithms' classifications of self-repairs applied to psychiatric data was 0.68. Reference Howes, Hough, Purver and McCabe21 To adjust for number of words spoken by each psychiatrist, self-repair was normalised by calculating mean number of self-repairs per 1000 words.
Secondary outcomes
Psychiatrist confidence. A self-rated questionnaire to assess psychiatrist self-confidence in communicating with patients with psychosis before and after the training was developed. Ten items (rated from 0 to 10) relating to each area in the training (e.g. ‘I feel comfortable communicating with patients with negative symptoms’, ‘I feel comfortable explaining psychotic illness to patients’). A mean score was calculated, ranging from 0 to 10, a higher score indicating higher self-confidence.
Therapeutic relationship. The therapeutic relationship was assessed using the Scale To Assess Therapeutic Relationship (STAR) Reference McGuire-Snieckus, McCabe, Catty, Hansson and Priebe23 by each patient and psychiatrist at baseline and follow-up. The STAR scale was developed in a 4-year study beginning with item generation from interviews with patients and professionals and existing therapeutic relationship scales before rigorous psychometric validation. Reference McGuire-Snieckus, McCabe, Catty, Hansson and Priebe23 STAR has a patient and professional version. Each version has 12 items and 3 distinct factors: positive collaboration, positive clinician input and non-supportive clinician input and emotional difficulties. The total score range is 0–48 (a higher score equals a better relationship). Length of therapeutic relationship was documented.
Originally, a further follow-up point was planned, 6 months after the post-training encounters were recorded. However, this turned out to be impractical because many of the psychiatrists rotated posts after 1 year and so were no longer treating the patients in the trial.
Procedure
Consent was sought from individual psychiatrists prior to randomisation by the researchers on the study (P.J. and J.D.). Specialist psychiatric trainees working in out-patient clinics in East and North East London were identified. The number of eligible trainees was lower than anticipated. Hence, the inclusion criteria were widened to include fully qualified psychiatrists, i.e. staff and associate specialist grade (SASG) and consultant psychiatrists. Information letters were sent to 35 psychiatrists. Participating psychiatrists identified eligible patients in out-patient clinics. Eligible patients were approached by an independent researcher before their appointment with the psychiatrist, and were masked to whether their psychiatrist was part of the intervention or control group. Patients who provided written informed consent had their encounter video-recorded. When the training was complete, the next time each participating patient attended the clinic, this follow-up encounter was video-recorded. Ethical approval was granted by East London Research Ethics Committee 1 (10/H0703/12).
Psychiatrists' and patients' age, gender and ethnicity were recorded along with length of time psychiatrists had been qualified. Data were collected on patients' employment status and treatment history.
Researchers assessed symptoms on the 30-item Positive and Negative Syndrome Scale (PANSS) at baseline and follow-up. Reference Kay, Fiszbein and Opler24 Researchers were trained in the assessment and interrater reliability was good (ICC = 0.90).
Randomisation and masking
Consenting psychiatrists were randomly allocated using simple randomisation in a 1:1 ratio to the control or intervention group. This was generated by the statistician (S.B.) using a sequence generated in Excel with the RAND function. There was no allocation concealment. Each psychiatrist was assigned to the next allocation in the sequence. The primary outcome, self-repair, was masked. For the secondary outcome, the therapeutic relationship, patients were masked but it was not possible to mask psychiatrists.
Data analysis
Data analysis was conducted in Stata 12.0. Data were summarised as numbers and percentages or means and standard deviations. Using all available cases, the adjusted treatment differences (intervention v. control group) along with 95% confidence intervals and P-values were estimated following intention-to-treat principles. The ICCs were estimated for each outcome using an adaptation of one-way analysis of variance which does not truncate negative ICCs at zero. Reference Ukoumunne25
For the primary outcome, self-repair, and for STAR psychiatrist, linear mixed effects regression models were fitted by restricted maximum likelihood, adjusting for baseline measure of the outcome and including a random effect (random intercept) for psychiatrist. Additionally, for STAR psychiatrist, patient-reported number of months under the care of their psychiatrist was adjusted for.
For STAR patient, the estimated ICC was negative. Hence, a linear regression model ignoring clustering was fitted so as not to bias the standard error of treatment effect downwards. Baseline STAR patient score was adjusted for along with baseline PANSS total score based on previously reported negative associations between the PANSS and STAR patient. Reference McGuire-Snieckus, McCabe, Catty, Hansson and Priebe23
Results
Psychiatrists
Out of 35 psychiatrists that were approached, 25 (71%) agreed to participate and were randomised. One psychiatrist was excluded before randomisation due to changing post. Four psychiatrists (control n = 2, intervention n = 2) had to be excluded after randomisation because they had too few eligible patients/changed post, leaving ten psychiatrists in the intervention group and eleven in the control group. All ten psychiatrists in the intervention group participated in training. Participant flow can be seen in the CONSORT diagram (Fig. 1). Psychiatrist characteristics are presented in Table 1.

Fig. 1 CONSORT diagram showing participant flow in the study.
Table 1 Psychiatrists' sociodemographic characteristics
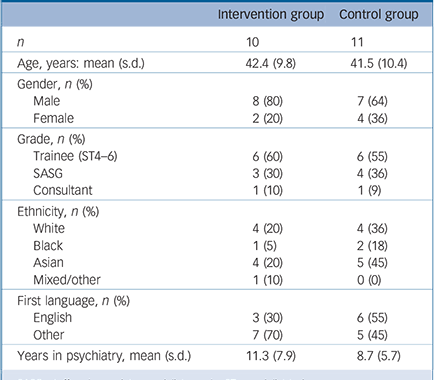
| Intervention group | Control group | |
| n | 10 | 11 |
| Age, years: mean (s.d.) | 42.4 (9.8) | 41.5 (10.4) |
| Gender, n (%) | ||
| Male | 8 (80) | 7 (64) |
| Female | 2 (20) | 4 (36) |
| Grade, n (%) | ||
| Trainee (ST4–6) | 6 (60) | 6 (55) |
| SASG | 3 (30) | 4 (36) |
| Consultant | 1 (10) | 1 (9) |
| Ethnicity, n (%) | ||
| White | 4 (20) | 4 (36) |
| Black | 1 (5) | 2 (18) |
| Asian | 4 (20) | 5 (45) |
| Mixed/other | 1 (10) | 0(0) |
| First language, n (%) | ||
| English | 3 (30) | 6 (55) |
| Other | 7(70) | 5 (45) |
| Years in psychiatry, mean (s.d.) | 11.3 (7.9) | 8.7 (5.7) |
SASG, staff and associate specialist grade; ST, specialist trainee.
Patients
A total of 407 patients were eligible; 191 did not attend their appointment. Twenty-five were not approached (considered too ill to approach for consent or appointment overlapped with another participant). Ninety-three did not consent. Of those approached, 97 patients (51%) were recruited. An average of 4.6 patients (s.d. = 1.9, range 1–7) were recruited per psychiatrist: 5.0 patients (s.d. = 1.6) per psychiatrist in the intervention group and 4.3 patients (s.d. = 2.1) per psychiatrist in the control group.
At baseline, patient data were collected from 97 patients: 96 encounters were video-recorded, 1 was missing due to equipment malfunction. Sixty-four patients were followed up and had the second encounter video-recorded. Patients could only be followed up if they were seeing the same psychiatrist again. Reasons for loss to follow-up are provided in Fig. 1.
There was an average of 152 days (approximately 5 months) between baseline and follow-up recordings (s.d. = 80.2, range 47.2–500 days). The average baseline encounter length was 18.9 min (s.d. = 7.6, range 7.3–37.1) and at follow-up was 18.4 min (s.d. = 8.7, range 4.0–43.5).
Patient characteristics are summarised in Table 2.
Table 2 Patients' sociodemographic and clinical characteristics a
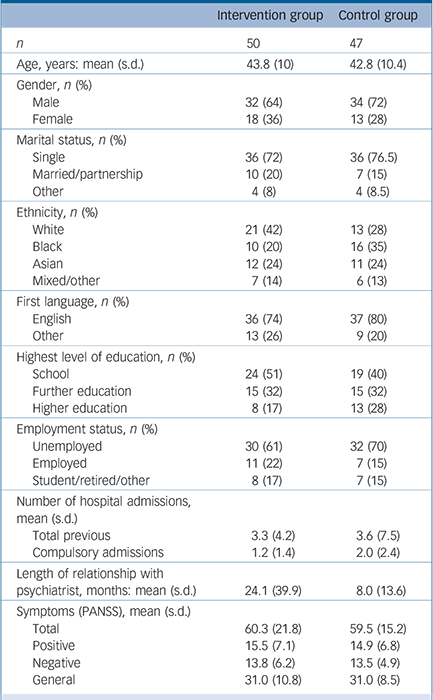
| Intervention group | Control group | |
|---|---|---|
| n | 50 | 47 |
| Age, years: mean (s.d.) | 43.8 (10) | 42.8 (10.4) |
| Gender, n (%) | ||
| Male | 32 (64) | 34 (72) |
| Female | 18 (36) | 13 (28) |
| Marital status, n (%) | ||
| Single | 36 (72) | 36 (76.5) |
| Married/partnership | 10 (20) | 7(15) |
| Other | 4(8) | 4 (8.5) |
| Ethnicity, n (%) | ||
| White | 21 (42) | 13 (28) |
| Black | 10 (20) | 16 (35) |
| Asian | 12 (24) | 11 (24) |
| Mixed/other | 7 (14) | 6 (13) |
| First language, n (%) | ||
| English | 36 (74) | 37 (80) |
| Other | 13 (26) | 9 (20) |
| Highest level of education, n (%) | ||
| School | 24 (51) | 19 (40) |
| Further education | 15 (32) | 15 (32) |
| Higher education | 8 (17) | 13 (28) |
| Employment status, n (%) | ||
| Unemployed | 30 (61) | 32 (70) |
| Employed | 11 (22) | 7(15) |
| Student/retired/other | 8 (17) | 7(15) |
| Number of hospital
admissions, mean (s.d.) |
||
| Total previous | 3.3 (4.2) | 3.6 (7.5) |
| Compulsory admissions | 1.2 (1.4) | 2.0 (2.4) |
| Length of relationship
with psychiatrist, months: mean (s.d.) |
24.1 (39.9) | 8.0 (13.6) |
| Symptoms (PANSS), mean (s.d.) | ||
| Total | 60.3 (21.8) | 59.5 (15.2) |
| Positive | 15.5 (7.1) | 14.9 (6.8) |
| Negative | 13.8 (6.2) | 13.5 (4.9) |
| General | 31.0 (10.8) | 31.0 (8.5) |
PANSS, Positive and Negative Syndrome Scale.
a. Ethnicity was missing for 1 patient, first language was missing for 2 patients, highest level of education was missing for 3 patients and employment status was missing for 2 patients.
Associations between clinical characteristics and outcomes
At baseline, the Spearman correlation between patient-rated therapeutic relationship on the STAR and the patient-reported length of relationship with the psychiatrist was r = −0.02 (P = 0.85) The Spearman correlation between psychiatrist-rated therapeutic relationship on the STAR and the length of the relationship was r = 0.27 (P = 0.02).
Primary outcome
Psychiatrist self-repair
Psychiatrist self-repair was significantly higher in the intervention than the control group (Table 3; adjusted mean difference 6.4 self-repairs per 1000 words, 95% CI 1.46–11.33, P<0.011). The model-based ICC for self-repair was 0.03: 3% of the variability in psychiatrist self-repair could be attributed to differences between psychiatrists. This corresponded to a large effect, Cohen's d = 0.91.
Table 3 Adjusted differences in means between the intervention and control groups on the primary and secondary outcomes a
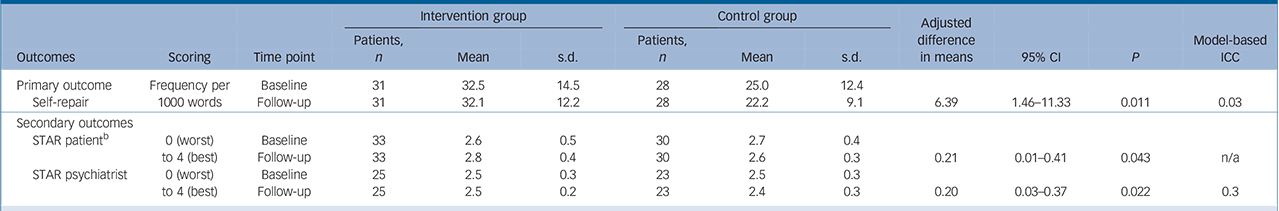
| Intervention group | Control group | Adjusted difference in means |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcomes | Scoring | Time point | Patients, n |
Mean | s.d. | Patients, n |
Mean | s.d. | 95% CI | P | Model-based ICC |
|
| Primary outcome | Frequency per 1000 words |
Baseline Follow-up |
31 | 32.5 | 14.5 | 28 | 25.0 | 12.4 | ||||
| Self-repair | 31 | 32.1 | 12.2 | 28 | 22.2 | 9.1 | 6.39 | 1.46–11.33 | 0.011 | 0.03 | ||
| Secondary outcomes | ||||||||||||
| STAR patient b | 0 (worst) to 4 (best) |
Baseline Follow-up |
33 | 2.6 | 0.5 | 30 | 2.7 | 0.4 | ||||
| 33 | 2.8 | 0.4 | 30 | 2.6 | 0.3 | 0.21 | 0.01–0.41 | 0.043 | n/a | |||
| STAR psychiatrist | 0 (worst) to 4 (best) |
Baseline Follow-up |
25 | 2.5 | 0.3 | 23 | 2.5 | 0.3 | ||||
| 25 | 2.5 | 0.2 | 23 | 2.4 | 0.3 | 0.20 | 0.03–0.37 | 0.022 | 0.3 | |||
ICC, intracluster correlation coefficient; n/a, not applicable; STAR, Scale To Assess Therapeutic Relationship.
a. Each model is adjusted for the outcome measure at baseline, treatment group and, additionally for STAR patient, Positive and Negative Syndrome Scale negative score at baseline, and for STAR psychiatrist, patient-reported number of months under the care of their psychiatrist.
b. Model fitted by linear regression without a random effect for psychiatrist.
Secondary outcomes
Psychiatrist self-confidence
Self-confidence in communicating with patients with psychosis increased. Paired t-tests showed a significant difference between scores before and after the training (t = 5.19, 95% CI 1.0–2.4, P<0.01). The mean score increased by 1.7 points (0–10 scale) from 6.9 (s.d. = 1.4) at baseline to 8.5 (s.d. = 1.2) at follow-up.
Therapeutic relationship
The quality of the therapeutic relationship improved significantly more in the intervention group than in the control group (Table 3), both on psychiatrist ratings (STAR mean difference 0.20, 95% CI 0.03–0.37, P = 0.022) and patient ratings (STAR mean difference 0.21, 95% CI 0.01–0.41, P = 0.043). The ICC for the psychiatrist-rated STAR was 0.3, i.e. 30% of the total variability in psychiatrists' ratings of the quality of the therapeutic relationship with their patients can be attributed to differences between psychiatrists. The ICC on the patient-rated STAR was negative, which, given the large sampling variation of ICCs, is most likely due to chance. The effect size for psychiatrist ratings of the relationship was d = 0.4, a medium effect. The effect size for patient ratings of the relationship was d = 0.56, a medium effect.
Feedback on the training
Attendance at the training was good (100% participated in at least 3 of 4 sessions). Psychiatrists who could not attend a specific session received the session later or watched a video of the session. Psychiatrists rated the training as highly beneficial (mean score 8.9) on a 0–10 scale (see Appendix for participant quotes).
Change in communication
Online Table DS1 shows examples of psychiatrist communication after the training for each of the four areas covered. For example, agenda-setting was one aspect of empowering the patient by asking them what they wanted to talk about at the beginning of the meeting rather than ‘Any questions?’ when wrapping up. The following question was posed 40 s into the meeting: ‘Well, the main thing would be perhaps today to understand what you would like from coming to meet with me today. What things did you want to talk about?’
Discussion
This study found that a brief intervention to enhance psychiatrist–patient communication in the treatment of psychosis was effective. Psychiatrists' effort in establishing shared understanding with their patients was significantly higher after training. Both psychiatrists' and patients' views of the therapeutic relationship improved.
Strengths and limitations
The strengths of the study were that psychiatrist encounters in the clinic were video-recorded before and after training so that the change in communication could be compared between the control and the intervention group, adjusting for baseline communication. There was a range of experience among the psychiatrists and also varying lengths of relationships with their patients. The limitations were that the follow-up sample was smaller than at baseline because some psychiatrists changed post or were on sick leave. The participating patients may not be representative of all patients as they are likely to be more engaged in services and agreeable to participate in research. Moreover, the psychiatrists who participated may not be representative as they are likely to be more motivated than those who did not participate.
Theoretical model: shared understanding
The findings are in line with the theoretical model that guided the training and the trial. Psychiatrists' confidence in communicating with patients with psychosis improved. Communication was better after the training and the therapeutic relationship improved. This is the first communication intervention in mental healthcare to show these effects. Training in communication skills may benefit from an underlying theoretical model. The focus on self-repair may appear rather technical. However, with psychiatrists in the intervention group using 44% more self-repair than the control group, it appears to be a valid index of communicative interest in and engagement with the patient. The psychiatrists were not made aware that self-repair is considered to be positive or that self-repair would be assessed as an outcome. Hence, they are unlikely to have been consciously trying to do more of it in their communication. We would predict that trying to do more self-repair would, in itself, not be helpful. Rather, repairs are symptomatic of the effort a speaker is investing in producing an utterance that is tailored for their recipient. As such, they are likely to reflect a shift in thinking about the role of communication and genuine adjustments to find the best possible expression.
Taking the listener's needs into account: preventing misunderstanding and displaying sensitivity
To illustrate the kinds of self-repairs used in practice and the different ways in which they can take a listener's needs into account, some examples are provided. In the following utterance, the psychiatrist states ‘I mean it would be good if you can keep up with the healthy life style, I mean er, not to take the medication if not really necessary’. Here ‘healthy life style’ is qualified (repaired) to ‘not to take the medication’ as a number of lifestyle factors have previously been discussed including diet and exercise. In this example, the self-repair clarifies a referent and prevents a possible misunderstanding by the patient. In the next example, the psychiatrist has proposed that it may be worth exploring how the patient could gain some more control over their overpowering voices rather than taking such a high dose of medication. The patient is somewhat resistant to the term ‘control’. In line 5, the psychiatrist amends ‘control’ in a sensitive adjustment to the patient's perspective.
-
1. Patient: But isn't controlling wrong in a sense? Strange.
-
2. Doctor: It it.
-
3. Doctor: It it it it.
-
4. Patient: You can actually control er.
-
5. Doctor: Er, when I say control, I I I I'm, I think more in living, in terms of living with them, yeah?
Finally, in the excerpt below, the psychiatrist and patient have been discussing the patient's mother's recent death and the general question format ‘How have things been in the past few months?’ is revised quickly to ‘I mean, I know that your day kind of revolved around [your mother]’, displaying sensitivity to the patient's particular circumstances. Not revising the question from its first version would be hearable as insensitive to how the patient's life has been affected by their mother's death. Revising it in this way indicates a sensitive affective stance towards the patient.
Doctor: So how have things been in the past few months, I mean, I know that your day kind of revolved around your mother?
Patient: My day revolves around seeing my brother and sister a lot now, now my Mum's no longer with us.
It is interesting that psychiatrist communicative engagement decreased in the control group but was maintained in the intervention group. This is consistent with psychiatrists' reports in the training on the challenges of communicating about psychotic experiences over time, i.e. when they first meet patients, they are more engaged in their experiences but this can be challenging to maintain when patients are keen to talk about experiences repeatedly. A focus on self-repair, as an index of engagement with patients, may be useful in training and in research. Psychiatrists identified the training as filling a gap in their training. In research, the current theoretical model could be applied in other disorders and treatment settings to advance the field of communication skills in psychiatry and medicine.
Improving the therapeutic relationship
Psychiatrists' ratings of the therapeutic relationship improved considerably (a medium effect size). Although the confidence intervals are reasonably wide, possibly due to the sample size, this is the first study to show an improvement of the therapeutic relationship through training and suggests that the proposed mechanism of effect (i.e. increased communicative engagement with the patient) does indeed improve the quality of the therapeutic relationship. This finding is encouraging given that, in psychiatric treatment, the professional's rating of the relationship appears to be a stronger predictor of outcome than the patient's rating, Reference McCabe, Bullenkamp, Hansson, Lauber, Martinez-Leal and Rössler26 the reverse of psychotherapy. Given that the odds of having adherent patients are twice as high if there is a good doctor–patient relationship, Reference Zolnierek, Matteo and Robin5 this is an important locus of intervention in improving longer-term outcomes. The current study was designed to investigate the effect of the intervention on process outcomes. Future studies would be required to investigate longer-term outcomes such as symptoms, quality of life and social functioning.
There is increasing interest in harnessing the potential of the therapeutic relationship in psychiatry. Reference Arnow and Steidtmann27 Two interventions have focused on helping patients to prioritise what they wish to discuss with their clinician. Priebe et al Reference Priebe, McCabe, Bullenkamp, Hansson, Lauber and Martinez-Leal28 found that a computer-mediated intervention to structure care coordinator–patient communication improved quality of life, reduced unmet needs for care and improved treatment satisfaction. Van Os et al Reference Van Os, Altamura, Bobes, Gerlach, Hellewell and Kasper29 used a checklist which patients completed before seeing their psychiatrist, which improved patient-reported quality of the communication and increased changes in management. Meanwhile, there is considerably more research in other fields such as primary care and oncology. Reference Dwamena, Holmes-Rovner, Gaulden, Jorgenson, Sadigh and Sikorskii30 As Fallowfield et al have noted, senior oncology doctors acknowledge that lack of communication training in complex medical contexts contributes to psychological morbidity, emotional burnout and depersonalisation. Reference Fallowfield31 Psychiatrists are also at risk of these negative outcomes. Many psychiatrists are highly skilled communicators addressing complex problems, and some of the training was based on identifiying what they are doing in everyday practice. Specifying these skills and integrating them in psychiatric training would address the need within the profession to define the skills that psychiatrists use in treating complex mental health problems. Reference Drew, Chatwin and Collins8,Reference Bhugra32,Reference Maj33
This is the first study to test an intervention for psychiatrists to enhance communication with patients with psychosis. It suggests that shared understanding, which can be challenging in the treatment of psychosis, can be targeted in training and is important for improving the quality of communication and the therapeutic relationship.
Funding
The TEMPO study was funded by the National Institute for Health Research's (NIHR) Research for Patient Benefit Programme (PB-PG-0408-16279). The work was also supported by the NIHR Collaboration for Leadership in Applied Health Research and Care South West Peninsula at the Royal Devon and Exeter NHS Foundation Trust. The funder had no role in study design, data collection, data analysis, interpretation, writing of the report, or decision to submit for publication. All study researchers were independent from the funder. The views are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health.
Acknowledgements
We are most grateful to the participating psychiatrists and patients, Dr Peter Byrne for delivering training, Husnara Khanom and Mary Lavelle for assistance with data collection, and Chris Howes and Julian Hough for assistance with self-repair annotation. We also thank the anonymous reviewers for their constructive comments in revising the manuscript.
Appendix
Participant perspectives on the training
‘The hearing voices exercise was very powerful. I now understand why patients want to talk about their voices.’
‘How to explore in depth patients’ concerns and listen actively with more attention to patients' cues.’
‘EAR (Explore, Active Listening and Respond) and GUNS (Give overview of options, Understanding check, Negotiate, Summarise decision) were excellent!’
‘I learned useful approaches and insight into my abilities (both strength and weaknesses) as a psychiatrist’
‘Understanding that I need to explore patients’ concerns before coming to a decision-making stage.’
‘The art of discussion and negotiation in sharing decisions.’
‘Paying more attention to the patient's agenda and their priorities … reduces the feeling of yet another routine.’
‘Goal-setting with patients with negative symptoms – realistic and achievable.’
‘Thinking about the patient perspective of psychosis.’
‘Advanced techniques for explaining psychotic symptoms.’
‘Even more of a focus on conflict and disagreement could be good.’
‘I thought I knew a lot about the story of his delusions, but going through the “voices checklist” makes me realise that I don't really how and when it started and so on.’
‘I've never realised how much taking notes in the consultation affects the connection with the patient.’
‘It sounds as if I'm just going through a checklist, rather than talking to the patient.’
‘I should invite the patient to ask more questions.’









eLetters
No eLetters have been published for this article.