Introduction
Opioid analgesics play a central role in cancer pain treatment but have constipation as a serious side effect. Opioid-induced constipation (OIC) has been reported to occur in 80% of patients taking opioid analgesics and can lead to a decline in patient quality of life (QOL) (Abramowitz et al. Reference Abramowitz, Béziaud and Labreze2013; Droney et al. Reference Droney, Ross and Gretton2008; Mesía et al. Reference Mesía, Virizuela Echaburu and Gómez2019). The analgesic action of opioid analgesics is expressed mainly through central μ-opioid receptors, while gastrointestinal disorders such as constipation are thought to occur mainly through peripheral μ-opioid receptors in the gastrointestinal tract (Brock et al. Reference Brock, Olesen and Olesen2012). Naldemedine has a large molecular weight and extremely low blood–brain barrier permeability, and has the effect of improving constipation without affecting the analgesic effect of opioid analgesics. Therefore, no central nervous system symptoms related to opioid withdrawal are observed (Coluzzi et al. Reference Coluzzi, Scerpa and Pergolizzi2020; Katakami et al. Reference Katakami, Harada and Murata2017; Webster et al. Reference Webster, Nalamachu and Morlion2018), although the same is true in patients with blood–brain barrier disruption (Osaka et al. Reference Osaka, Ishiki and Yokota2019).
Patients receiving palliative care are known to exhibit various neuropsychiatric symptoms (Minagawa et al. Reference Minagawa, Uchitomi and Yamawaki1996). However, to our knowledge, there are no reports of opioid withdrawal marked by psychiatric symptoms in patients receiving palliative care. Here, we present a case of a patient undergoing palliative care who received naldemedine for OIC and developed symptoms centered around restlessness and sweating from about 40 days after administration. Opioid withdrawal syndrome (OWS) was diagnosed by close examination and the symptoms improved with appropriate treatment.
Case report
The patient was a 78-year-old woman with stage IVB uterine sarcoma (liver and lung metastases) who had a history of type 2 diabetes. She had abnormal vaginal discharge at around 2 years prior to her visit to our clinic , and was diagnosed with stage IVB uterine sarcoma as a result of a detailed examination after her initial consultation with a doctor in October, 1 year prior to her visit . A simple total hysterectomy and bilateral salpingectomy were performed in December of the same year. Despite undergoing 3 courses of chemotherapy (doxorubicin) as an outpatient from January to March of the same year as her visit to our clinic, she was later diagnosed with progressive disease due to the increase in liver and lung metastases. Despite receiving medical treatment, she was admitted to hospital after a fall at home. As a result of a detailed examination, metastasis to the fourth thoracic vertebra (Th4) and paraplegia were diagnosed. On the same day, spinal fusion surgery and bone grafting were performed postoperative day 1 (POD 1), and she was hospitalized and limited to movement by wheelchair.
On POD 9, loxoprofen 180 mg/day and acetaminophen 1,800 mg/day were started for Th4 pain; however, as the pain did not improve, oxycodone hydrochloride hydrate 10 mg/day were additionally administered on POD 11. Magnesium oxide 990 mg/day was started for OIC on POD 22, and naldemedine 0.2 mg/day was added on POD 24 (we hereafter refer to this as day 1 of naldemedine treatment or Day 1).
Thereafter, the patient’s prognosis was expected to be about 3 months based on her general condition, and she was transferred from the general ward to the palliative care ward.
Her Eastern Cooperative Oncology Group Performance Status (Oken et al. Reference Oken, Creech and Tormey1982) on admission to the palliative care unit was 3, and the numerical rating scale for bilateral chest pain was 8 (Hjermstad et al. Reference Hjermstad, Fayers and Haugen2011).
There were no noticeable changes for a few weeks after hospitalization; however, on Day 43, she complained that “I want you to put me in a wheelchair as soon as possible. I feel bad if I can’t move,” and the frequency of requests to staff to transfer from her bed to a wheelchair suddenly increased. In addition, she was sitting in her wheelchair and repeatedly going in and out of the hallway and her room with a distinctly different degree of restlessness than before.
The patient became more fidgety and restless on Day 44. She was unable to concentrate during conversations with staff, sometimes interrupting conversations, and repeatedly asking to be transferred from wheelchair to bed and from bed to wheelchair. Thereafter, she continued to go back and forth in the hallway of the ward with an irritated look, and began to request transfer to her wheelchair both day and night.
The patient’s vital signs were heart rate 83 beats/min, blood pressure 93/56 mmHg, body temperature 36.4°C, respiratory rate 12 beats/min, and there was no disturbance of consciousness observed. Her blood glucose level was 146 mg/dL, and her electrolytes were normal. However, sweating was observed around the forehead region despite the fact that the patient was examined during bed rest. Computerized tomography scans of the head showed no abnormal brain findings.
Forehead sweating and other symptoms of restless led us to suspect opioid withdrawal due to naldemedine administration, and the naldemedine was stopped. The Clinical Opiate Withdrawal Scale (COWS) (Wesson and Ling Reference Wesson and Ling2003) score at the time of diagnosis was 10. On the 4th day after the discontinuation of naldemedine, her COWS score was 8 and no sweating was observed, while on the 7th day, her COWS score was 3 and both anxiety and irritability had decreased. No further changes in symptoms were observed thereafter (Table 1).
Table 1. The Clinical Opiate Withdrawal Scale (COWS) before and after discontinuation of naldemedine
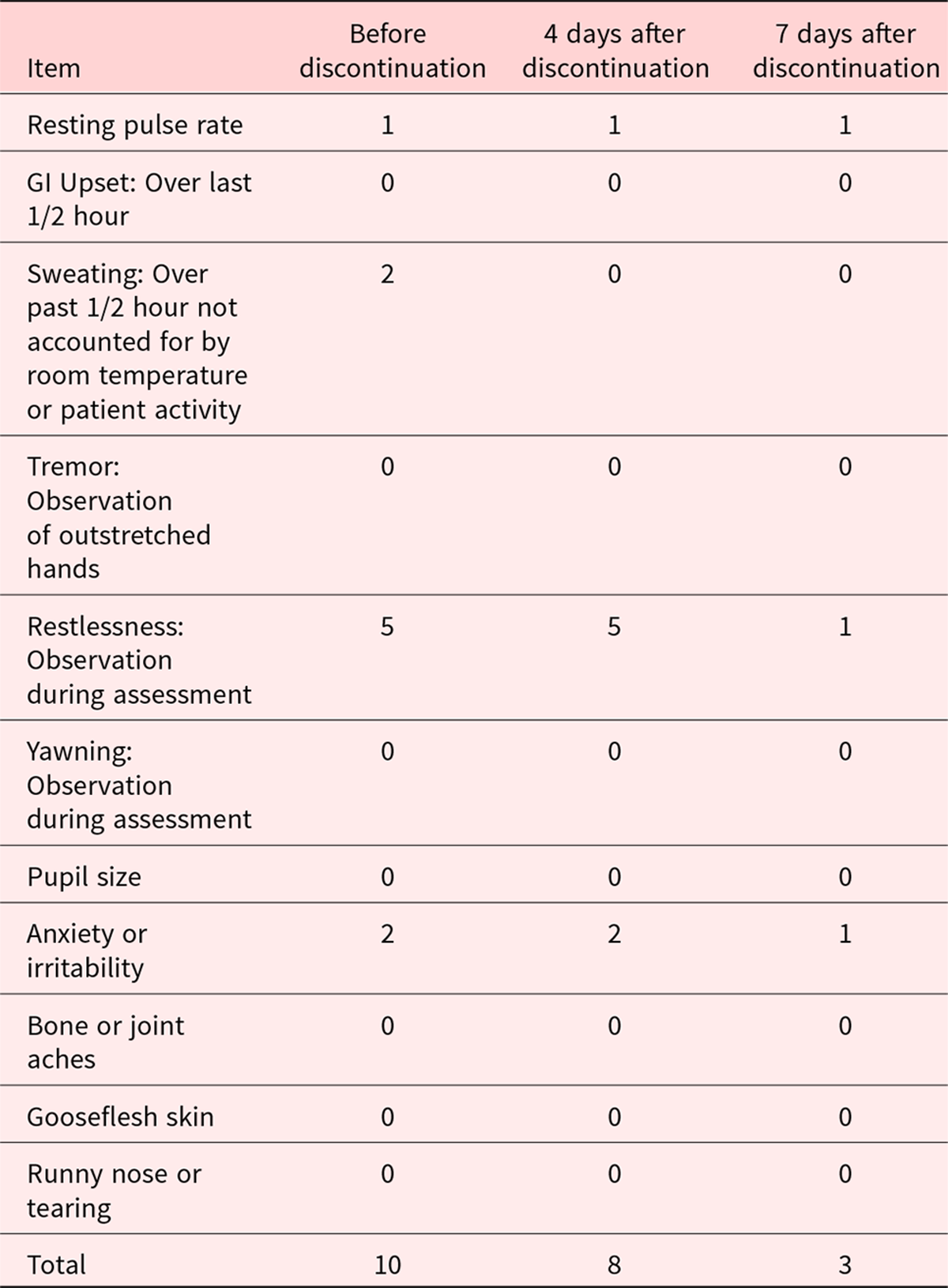
Score: 5–12 = mild; 13–24 = moderate; 25–36 = moderately severe; >36 = severe withdrawal.
Discussion
Herein we presented a case of OWS in a patient receiving palliative care that developed 43 days after the start of naldemedine administration (Day 43). This case demonstrated psychiatric symptoms frequent in palliative care, symptoms that closely resemble those of anxiety, depression, delirium, and restlessness (Minagawa et al. Reference Minagawa, Uchitomi and Yamawaki1996), and are very useful for differential diagnosis.
In this case, it was the restlessness and sweating that led to the suspicion of OWS. Akathisia was the differential diagnosis in palliative care patients demonstrating restlessness (Kawanishi et al. Reference Kawanishi, Onishi and Kato2007), but no causative agents with the potential to induce akathisia were administered in this case. Next, shock, malignant syndrome (Kawanishi et al. Reference Kawanishi, Onishi and Kato2005; Sato et al. Reference Sato, Onishi and Kawanishi2020), and hypoglycemia are mentioned as differentials when sweating appears; however, the vital signs were stable for this patient. She was not taking any medications likely to induce malignant syndrome, and no hypoglycemia was observed. Further, the patient was negative for physiological sweating based on the situation at the time of examination.
This patient developed withdrawal symptoms 43 days after the start of naldemedine administration. There have been 2 previous reports of onset within 1 hour (Ishida et al. Reference Ishida, Hiraoka and Yaguchi2021; Ishii et al. Reference Ishii, Yamashita and Yamaguchi2020) and 1 after 4 months (Ishida et al. Reference Ishida, Uchida and Yabuno2022). As this drug improves OIC and is beneficial in terms of QOL, taken together with the previous reports, the current case suggests that OWS should always be monitored in patients receiving naldemedine, even in patients receiving it long-term.
In conclusion, we experienced a patient using opioid analgesics who received naldemedine for OIC and in whom OWS was suspected on the basis symptoms. The OWS disappeared after the discontinuation of naldemedine administration.
Patients receiving palliative care experience a range of psychiatric symptoms, and physical causes of such symptoms, such as OWS, must be considered.
Competing interests
None.




