Article contents
Reactive epoxies with functional zeolite fillers: IR spectroscopy and PALS studies
Published online by Cambridge University Press: 10 October 2011
Abstract
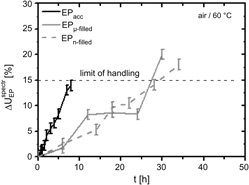
Epoxy–dicyandiamide (Dicy) formulations frequently contain a free accelerator for reducing the curing temperature and the time for network formation, which reduces the shelf life of these adhesives. This study compared the reaction kinetics during the storage at 60 °C for a precured epoxy adhesive (EP = diglycidyl ether of bisphenol A and Dicy, mass ratio 100:6.7, precured at 150 °C for 1 h) mixed either with free accelerator (=EPacc) or with the same concentration of accelerator immobilized in micro- or nanozeolite fillers (EPμ-filled and EPn-filled, respectively). During storage, the infrared (IR) study probed the chemical modifications. They lead to increasing cross-linking density and a loss of free volume as detected by positron annihilation lifetime spectroscopy (PALS). Cross-linking precedes to the chemical vitrification. Additionally, the glass transition and the free-volume parameters were investigated for the three EP’s as a function of temperature by PALS after thermal curing.
- Type
- Articles
- Information
- Copyright
- Copyright © Materials Research Society 2011
References
REFERENCES
- 3
- Cited by


