Contact precautions are commonly used as an infection prevention strategy. With advances in the field of infectious prevention, the use of personal protective equipment (PPE) evolved from leather gloves and coats during medieval ages to disposable gloves and isolation gowns in the 21st century.Reference Morgan, Wenzel and Bearman 1 The use of isolation precautions (ie, gloves and gowns) for patients infected with or colonized by methicillin-resistant Staphylococcus aureus (MRSA) or vancomycin-resistant Enterococcus (VRE) is recommended by the Centers for Disease Control and Prevention (CDC) in the most recent guidelines to prevent healthcare-associated infections (HAIs) caused by these organisms.Reference Siegel, Rhinehart, Jackson and Chiarello 2 While contact precautions have been considered an important strategy to prevent HAIs caused by MRSA and VRE, a growing body of evidence suggests that this may be unnecessary.Reference Morgan, Wenzel and Bearman 1 , Reference Morgan, Murthy and Munoz-Price 3 , Reference Harris, Pineles and Belton 4 Currently, more than 40 hospitals in the United States limit their use of contact precautions to situations such as patients with draining wounds, infectious diarrhea, and the care of high-risk patients.Reference Morgan, Wenzel and Bearman 1 While no reports exist of harm related to the discontinuation of contact precautions, this strategy remains controversial. We employed an interrupted time series (ITS) analysis to assess the impact of discontinuing contact precautions for MRSA- or VRE-infected or -colonized patients in an academic medical center.
METHODS
Data Collection
The Virginia Commonwealth University Medical Center is an 865-bed hospital with 146 intesive care unit (ICU) beds. The infection prevention program is based on a horizontal infection prevention platform that emphasizes and tracks process-of-care measures such as hand hygiene, contact precautions, daily chlorhexidine gluconate (CHG) bathing, implementation of central-line checklists, daily urinary catheter review, a bare below the elbows (BBE) approach to inpatient care, and use of disinfecting port protector caps (Table 1). The direct-observation hand hygiene program was initially implemented in 2004 with a baseline compliance of 45%, which increased to 91% by 2008. Observations reflecting compliance with contact precautions (ie, donning gown and gloves prior to room entry) hospital-wide were completed monthly by the institution’s hand hygiene monitoring team for the months of January through August in 2016 and 2017. Concurrent HAI surveillance was conducted by infection preventionists from 2010 through 2016 using the CDC’s National Healthcare Safety Network System (NHSN) definitions for central-line–associated bloodstream infection (CLABSI), catheter-associated urinary tract infection (CAUTI), and ventilator-associated event (VAE). These data were analyzed to calculate monthly rates of MRSA device-associated HAI, VRE device-associated HAI, and total device-associated HAI from 2010 to 2017. Patient days were collected monthly from a report generated by the information technology (IT) department. Overall, 7 different infection prevention interventions were implemented beginning in January 2011 (Table 3). The first of these was a urinary catheter bundle that included compliance with hand hygiene prior to catheter insertion, using aseptic technique for insertion, maintaining a closed system, using silver alloy catheters, keeping the urine drainage bag below the level of the hip, and avoiding catheter use if possible. Hospital-wide CHG perineal cleaning and CHG patient bathing were introduced next in June 2011 and March 2012, respectively. Notably, CHG bathing for ICU patients was implemented in 2007. Prior to 2013, all patients infected or colonized with MRSA or VRE were placed in contact isolation. In April 2013, we discontinued contact precautions for patients infected or colonized with MRSA or VRE. Patients infected with other pathogens of epidemiologic significance or those with actively draining infected wounds unable to be contained with a bandage remained in contact isolation. Furthermore, a BBE strategy for inpatient care was encouraged institution-wide by the infectious control committee beginning in 2009. Direct monitoring by dedicated staff members was instituted to monitor compliance with this recommendation, as well as adherence to contact precautions in August 2014. In October 2014, daily assessment by nursing staff of urinary catheter need was formalized by documentation in the electronic medical record (EMR). This intervention was coupled with an EMR-generated 72-hour automatic urinary catheter discontinuation. Deployment of ultraviolet-C (UV-C) disinfection technology was implemented in March 2015 for terminal disinfection of rooms occupied by Clostridium difficile–infected patients.
TABLE 1 Infection Prevention Process of Care Measures

NOTE. ICU, intensive care unit; CHG, chlorhexidine gluconate; N/A, not available.
a Direct observation by 2 trained hand hygiene monitors, recorded using iScrub app on iPod.
b Data obtained from electronic medical records.
c Point prevalence assessment conducted by infection preventionist and bedside nurses.
Statistical Analysis
We used a 2-proportion Z test to compare the rates of MRSA, VRE, and device-associated HAI before and after discontinuation of contact precautions. We used segmented regression analysis of ITS design to estimate the change in the monthly incidence rates of MRSA, VRE, and device-associated HAI following 7 different interventions while controlling for secular changes that may have occurred in the absence of the interventions. The impact of each intervention was expressed as the absolute difference between the outcome at the end of the intervention and its counterfactual value extrapolated by levels and trends of respective HAI rates prior to the intervention. We adjusted for serial autocorrelation using the Durbin-Watson statistic. We also tested for seasonality and stationarity using the Dicky-Fuller Unit root test. All analyses were 2-tailed and were conducted using SAS Proc ARIMA and Proc Autoreg version 9.4 software (SAS Institute, Cary, NC).
RESULTS
The process-of-care measures, including contact precautions, daily CHG bathing, central-line checklist completion, BBE, hand hygiene, daily urinary catheter review, and the use of IV port disinfecting caps hospital-wide are summarized in Table 1.
Table 2 summarizes the 2-proportion Z-test conducted to compare MRSA, VRE, and all pathogen device-associated infection rates between January 2010 and March 2017. The impacts of 7 different infection-prevention interventions introduced between 2011 and 2016 are summarized in Table 3. The rates of MRSA, VRE, and device-associated HAI using an ITS analysis are summarized in Figures 1 through 6.
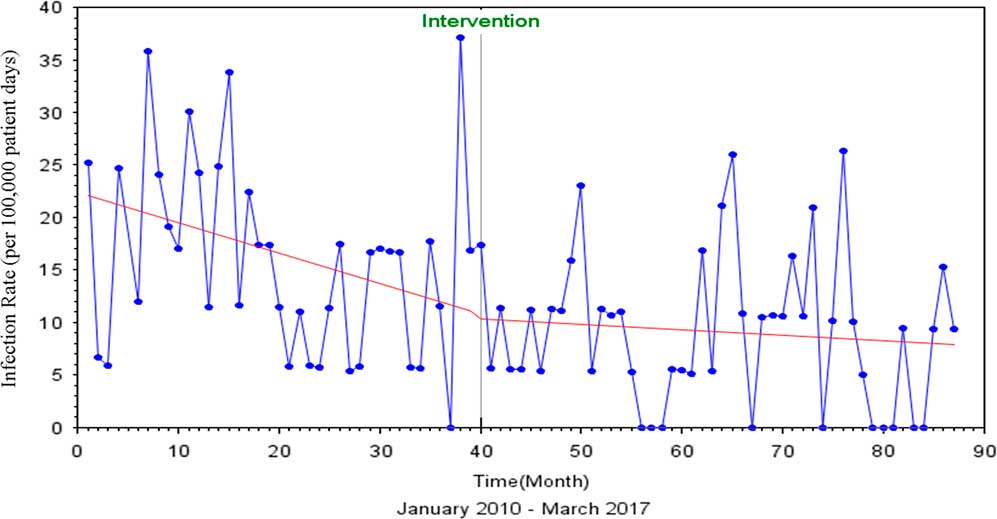
FIGURE 1 Interrupted Time Series Analysis of MRSA and VRE HAIs, Pre and Post Discontinuation of Contact Precautions
TABLE 2 The 2-Sample Z Test Comparing MRSA, VRE, and All Device-Associated Infection Rates Before and After Discontinuation of Contact Precautions

NOTE. CP, contact precautions; MRSA, methicillin-resistant Staphylococcus aureus; VRE, vancomycin-resistant Enterococcus.
TABLE 3 Summary of Horizontal Infection Prevention Strategies with Impact on Infection Prevention Outcomes

NOTE. MRSA, methicillin-resistant Staphylococcus aureus; VRE, vancomycin-resistant Enterococcus; ↓, decrease; ↑, increase; CHG, chlorhexidine gluconate; BBE, bare below the elbows; EMR, electronic medical record.
The Durbin-Watson statistic of 1.761 indicated no autocorrelation among the regressors, and there was a 3% decrease in MRSA and VRE rates after discontinuation of contact precautions (P=.85) (Figure 1). The Dickey-Fuller unit root test P value for single mean was 0.001, indicating that the series was stationary.
Similarly, for total device-associated HAI, the Durbin-Watson statistic of 1.723 indicated no autocorrelation among the regressors. After discontinuation of contact precautions, a 7% (P=.85) decrease in the rate of total device-associated HAI was observed (Figure 2). The Dickey-Fuller unit root test P value for single mean was 0.001, indicating that the series was also stationary.
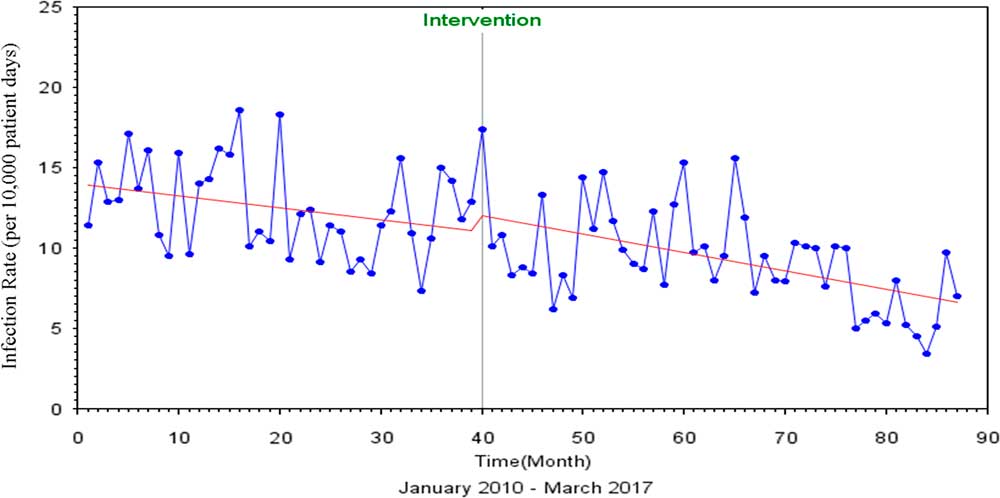
FIGURE 2 Interrupted Time Series Analysis of All Device Associated HAIs, Pre and Post Discontinuation of Contact Precautions
Following the introduction of urinary catheter bundles, the rate of MRSA infection decreased by 5.21 per 100,000 patient days (P=.44), the rate of VRE infection decreased by 3.01 per 100,000 patient days (P=.70), and the rate of infections caused by these organisms collectively decreased by 8.22 per 100,000 patient days (P=.39). The rate of device-associated HAI increased by 4.21 per 10,000 patient days (P=.19) (Figures 3 and 4).
With the next intervention (hospital-wide CHG perineal care), the rate of MRSA HAI increased by 1.20 per 100,000 patient days (P=.84), the rate of VRE infection decreased by 9.84 per 100,000 patient days (P=.16), and the rate for both organisms combined decreased by 8.64 per 100,000 patient days (P=.31) (Table 1). A similar trend was observed for the rate of device-associated HAI, which dropped by 1.11 per 10,000 patient days following this intervention (P=.70).
The implementation of hospital-wide CHG bathing also resulted in similar infection trends: the rate of MRSA infection increased by 1.37 per 100,000 patient days (P=.78), the rate of VRE infection decreased by 2.74 per 100,000 patient days (P=.63), and the rate of HAI for both organisms combined decreased by 1.36 per 100,000 patient days (P=.84). The rate of device-related HAI decreased by 1.80 per 10,000 patient days following this intervention.
Following discontinuation of contact precautions for MRSA and VRE in 2013, the rate of MRSA HAI decreased by 1.31 per 100,000 patient (P=.76), the rate of VRE HAI decreased by 6.25 per 100,000 patients (P=.21), and the rate of combined MRSA and VRE HAI decreased by 7.56 per 100,000 patients (P=.21). The rate of device-associated infections decreased by 2.44 per 10,000 patient days following this intervention (P=.23).
Direct monitoring by dedicated staff members to assess compliance with hand hygiene and BBE, with feedback to unit leadership, yielded a similar impact in infection rates. The rate of MRSA infection decreased by 4.33 per 100,000 patient days (P=.44), the rate of VRE decreased by 11.09 per 100,000 patient days (P=.09), the rate of infection by both organisms combined decreased by 15.42 per 100,000 patient days (P=.05), and the rate of device-associated HAI caused by organisms dropped by 0.26 per 10,000 patient days following this intervention (P=.92).
The use of UV-C disinfection robots was implemented in March 2015 for terminal disinfection of C. difficile rooms. Following this intervention, the rate of HAI increased by 1.58 per 100,000 for MRSA (P=.76), by 1.02 per 100,000 for VRE (P=.87), and by 2.60 per 100,000 (0.99) patient days for both organisms.
The urinary catheter review and EMR-generated 72-hour automatic discontinuation order was associated with a reduction of MRSA HAI of 2.34 per 100,000 patient days (P=.61). The rate of VRE HAI exhibited an upward trend following this intervention, with a rate that increased by 2.44 per 100,000 (P=.65). However, the rate of both organisms combined decreased by 0.10 per 100,000 (P=.99). The rate of device-associated infections caused by all organisms decreased by 1.13 per 10,000 patient days following this intervention. While the rates of HAI continued to trend downward between 2010 and 2016, no single intervention was statistically significant.
DISCUSSION
We employed an ITS analysis to assess the impact of discontinuing contact precautions for MRSA and VRE colonized or infected patients in an academic medical center. To date, a minority of academic medical centers in the United States have adopted policies for the discontinuation of contact precautions for these patients.
The use of contact precautions is controversial and may result in fewer patient visits by healthcare workersReference Harris, Pineles and Belton 4 , Reference Siegel, Rhinehart, Jackson and Chiarello 5 and may decrease the likelihood of examination by physicians.Reference Siegel, Rhinehart, Jackson and Chiarello 5 Moreover, this strategy may lead to depression, anxiety, and dissatisfaction among patientsReference Siegel, Rhinehart, Jackson and Chiarello 5 through delays in diagnostic procedures,Reference Croft, Liquori and Ladd 6 charting of vital signs, transport and even hospital discharge.Reference Morgan, Wenzel and Bearman 1 The controversy was further heightened when Croft et alReference Croft, Liquori and Ladd 6 recently reported no differences in preventable adverse events in isolated patients during a matched cohort trial. The most recent CDC guidelines for isolation precautions date back to 2007 and recommend the use of PPE, including gloves and isolation gowns, upon contact with all patients infected or colonized with multidrug-resistant organisms (MDROs), including MRSA and VRE.Reference Siegel, Rhinehart, Jackson and Chiarello 2 The data supporting this strategy largely rest on observational studies conducted during outbreaks rather than clinical trials.Reference Morgan, Wenzel and Bearman 1 , Reference Morgan, Murthy and Munoz-Price 3 , Reference Siegel, Rhinehart, Jackson and Chiarello 5 Notably, these studies employed a bundle approach, combining multiple interventions to achieve successful MDRO control.Reference Siegel, Rhinehart, Jackson and Chiarello 5
The discontinuation of contact precautions in acute-care settings remains controversial, with widespread variation. Recent studies suggest that colonization with MRSA and VRE may be short-lived, even in the event of a recent infection,Reference Shenoy, Lee and Taige 8 and several have questioned the potential for nosocomial transmission.Reference Harris, Pineles and Belton 4 , Reference Edmond, Masroor, Stevens, Ober and Bearman 7 , Reference Long, Beres, Olsen and Musser 9 In a randomized multicenter study, a universal glove and gown strategy for ICU patients did not impact the primary outcome of combined MRSA and VRE acquisition rates.Reference Harris, Pineles and Belton 4 When employing a universal gloving and gowning strategy, more than 300 patient encounters may be required to prevent a single instance of MRSA acquisition.Reference Harris, Pineles and Belton 4 In another study, whole-genome sequencing of 398 S. aureus isolates from 4 different hospitals in Texas did not reveal similarity among isolates to reflect nosocomial transmission. This finding leads to further questions regarding the use the contact precautions for these infections.Reference Long, Beres, Olsen and Musser 9 A recent meta-analysis suggests that contact precautions for MRSA and VRE may be safely discontinued with optimization of hand hygiene and horizontal infection prevention strategies.Reference Marra, Edmond, Schweizer, Ryan and Diekema 10
Gown and glove use is associated with significant cost for PPE.Reference Morgan, Wenzel and Bearman 1 , Reference Edmond, Masroor, Stevens, Ober and Bearman 7 Several prior studies have reported substantial cost savings after the discontinuation of contact precautions for MRSA- and VRE-infected or -colonized patients.Reference Edmond, Masroor, Stevens, Ober and Bearman 7 , Reference Huang, Septimus and Avery 11 One quasi-experimental before-and-after study demonstrated that discontinuation of contact precautions for MRSA and VRE may decrease isolation days by 45% and healthcare costs by up to $622,405 annually.Reference Long, Beres, Olsen and Musser 9
Contact precautions for MRSA and VRE are still widely implemented, with no consensus regarding the best practice.Reference Morgan, Murthy and Munoz-Price 3 , Reference Shenoy, Lee and Taige 8 Policies vary from active surveillance and isolation for MRSA and VRE to universal decolonization with or without contact precautions, universal gloving and gowning in ICUs, and discontinuation of contact precautions except in select circumstances (eg, open or draining wounds, infectious diarrhea, and outbreak control).Reference Harris, Pineles and Belton 4 , Reference Edmond, Masroor, Stevens, Ober and Bearman 7 , Reference Long, Beres, Olsen and Musser 9 , Reference Huang, Septimus and Avery 11 – Reference Roghmann, Johnson and Sorkin 13 Contact precautions were previously used at our institution for all patients infected or colonized with MRSA and/or VRE. This policy was revised in 2013, and afterward patients were isolated only if they had draining wounds not containable by a bandage that were positive for these organisms. This change was based on the assumption that a robust and reliable horizontal infection prevention program was sufficient to limit the cross transmission of all pathogens transmitted by patient contact with healthcare workers and the inanimate environment.Reference Edmond, Masroor, Stevens, Ober and Bearman 7 We observed no increase in the rates of MRSA and VRE HAI following the change in contact precautions policy. On the contrary, we continue to observe a sustained decrease in the overall rates of HAI, consistent with a national decrease in device-associated infection rates. 14 During the study period, no outbreaks were observed in our institution. Although no single intervention was responsible for the decreases in hospital-acquired infection at our institution, these findings suggest that discontinuation of contact precautions, when implemented in conjunction with horizontal infection prevention strategies, such as the use of central-line checklists, urinary catheter care bundles, CHG patient bathing, hand hygiene adherence monitoring, BBE, formalized EMR-based urinary catheter review and discontinuation protocols, and selective deployment of UV-C disinfection technologies, may not adversely impact clinical outcomes.
This study has several strengths. Healthcare-associated infections were identified using CDC National Healthcare Safety Network (NHSN) definitions to eliminate bias by utilizing a standard determined on a national level for HAI surveillance. All data were collected by certified infection prevention nurses. Trained observers used standardized hand hygiene, BBE, and contact precautions assessment methodology to minimize the potential for bias. Furthermore, the use of an ITS analysis allowed for a month-to-month assessment of infection trends over a course of years in relation to 7 different infection control measures implemented over time. This statistical method facilitates the assessment of the impact of infection rates following a particular intervention, as well as trends over a given period when multiple interventions are implemented in a sequential fashion.Reference Huang, Yokoe and Hinrichsen 12 It also eliminates the need for randomization, minimizes user bias and confounding associated with traditional study designs, and is effective in informing infection prevention programs while retrospectively reviewing interventions.Reference Bernal and Cummins 15
The study has several limitations. This study was a nonrandomized, quasi-experimental study conducted at a single academic center. We acknowledge that our compliance with contact precautions and other process of care measures was high (Table 1), which may reflect the random compliance audits and may not be an accurate marker of daily hospital-wide adherence. Additionally, data reflecting compliance with contact precautions were only available for January through August of 2016 and 2017. As multiple interventions were simultaneously implemented, this study may not predict the result of a particular intervention but rather the benefit of combining horizontal infection prevention strategies with discontinuation of contact precautions for MRSA and VRE patients. Notably, no active surveillance cultures for MRSA and VRE were performed during the study; therefore, the colonization burdens of MRSA and VRE are unknown. As a result, patients acquiring VRE or MRSA at our facility but admitted elsewhere with infection were not captured with this study design. Because our institution is the safety-net hospital for central Virginia, patient readmission to other neighboring facilities is not likely.
We add to the body of literature on the discontinuation of contact precautions for MRSA- and VRE-infected or -colonized patients. Our study is an extension of previously published work, with an extended time frame for analysis and a different methodology, an ITS analysis.Reference Gandra, Barysaukas, Mack, Barton, Finberg and Elison 16 Few studies have assessed the impact of discontinuing contact precautions for MRSA and VRE with an ITS analysis. The findings of this ITS analysis support the discontinuation of contact precautions for VRE- and MRSA-infected or -colonized patients when combined with comprehensive horizontal infection prevention strategies. Our study suggests that discontinuation of contact precautions for the control of endemic MRSA and VRE may be safe. Further studies, either with multicenter ITS analysis methodology or cluster randomized trials are needed to further explore this potential strategy change.
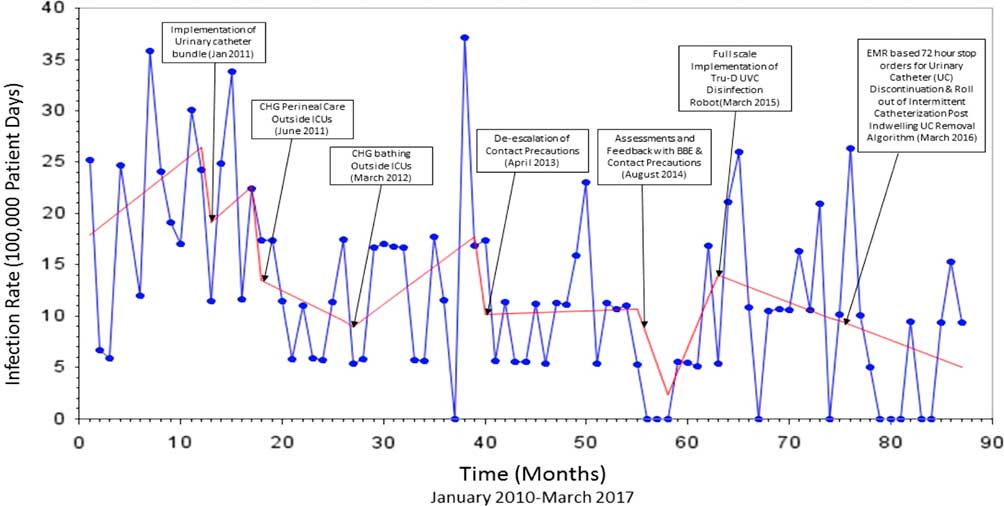
FIGURE 3 Interrupted Time Series Analysis of Device-associated MRSA and VRE HAIs
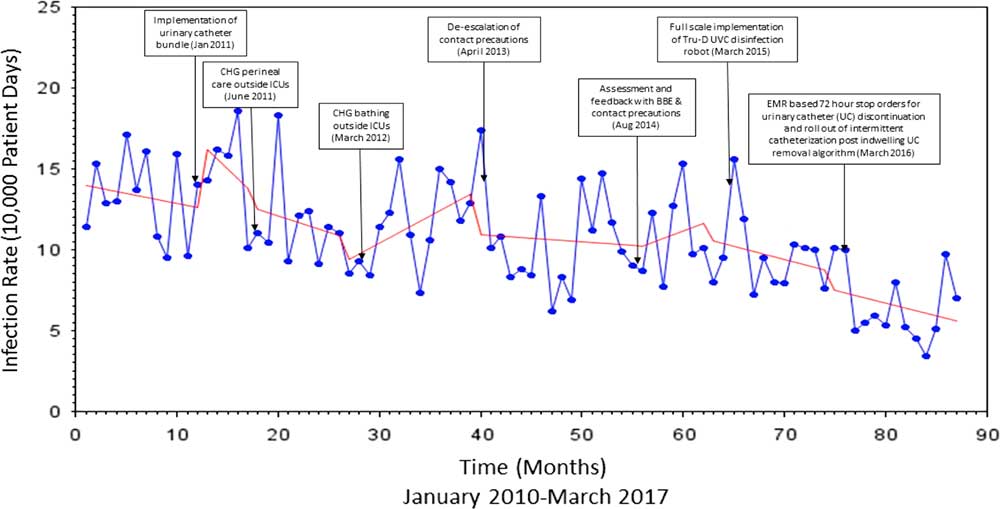
FIGURE 4 Interrupted Time Series Analysis of Device-associated HAIs
ACKNOWLEDGMENTS
Financial support: No financial support was provided relevant to this article.
Potential conflicts of interest: All authors report no conflicts of interest relevant to this article.









