Effective community surveillance to rapidly identify outbreaks of serious illness is important, particularly given the likelihood of emerging infectious disease and the threat of biological or chemical terrorism. In Europe surveillance has traditionally been performed by general practitioners recording the number of patients presenting with specific symptoms and collecting samples for laboratory testing [Reference Fleming, Schellevis and Paget1]. In addition, in the United Kingdom calls to a national telephone health helpline (NHS Direct) [2] are used for syndromic surveillance, aiming to provide early warning of non-specific rises in community morbidity [Reference Baker3]. A key limitation of this syndromic approach is the lack of medical or laboratory confirmation of diagnosis.
During the winter of 2003/2004, a pilot study demonstrated the feasibility of community-based virological sampling carried out by NHS Direct callers, without the intervention of a health-care worker [Reference Cooper4]. However, the work was conducted using a small sample (67 callers) and when it was known that influenza was already circulating in the community. We report a further study which investigated whether self-sampling by NHS Direct callers could provide viable samples for influenza surveillance. Moreover, given the availability of near patient tests for influenza [Reference Harnden5], this study also examines the acceptability of self-sampling by NHS Direct callers themselves.
Between 1 November 2004 and 11 February 2005 callers over the age of 15 years to three NHS Direct sites (Hampshire and Isle of Wight; West Midlands; South Yorkshire and South Humberside), who sought medical advice about reported ‘cold/flu’ symptoms and gave their consent to being contacted, were sent a specimen kit in the post. The kit included an information sheet, two nasal swabs, viral transport medium, instructions, appropriate packaging and a short questionnaire. Callers were asked to take a swab from each nostril and return the swabs by post to the national influenza reference laboratory of the Health Protection Agency. The swabs were tested by multiplex PCR for influenza viruses. Material found to be positive by PCR was cultured for viable virus isolation. We used univariate analysis to test the association between both response rate and positivity rate, and a range of call and sampling details.
During the study period the three NHS Direct sites received 244 664 calls for all syndromes, 1817 calls classified by nurses as ‘colds and flu’, from which 294 callers were recruited for the study. Sixty-one per cent of those recruited were female and 50% aged between 25–44 years, reflecting the demographic profile of demand for NHS Direct. Of the 294 sampling kits sent to NHS Direct callers, 142 samples (48%) were returned. The mean time between the NHS Direct call and swabbing was 4·1 days, between the NHS Direct call and laboratory analysis 7·4 days with the range 3–27 days. There were no significant differences in these mean times between positive and negative influenza samples. Callers aged ⩾45 years were significantly more likely to return swabs to the laboratory (P<0·001). Seven out of 141 callers who returned a questionnaire reported minor problems taking the swabs (e.g. ‘spilt the transport diluent’, ‘dropped swabs on floor’).
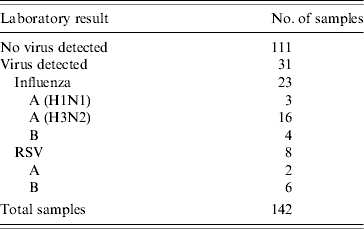
Twenty-three of the NHS Direct specimens (16·2%) were positive on PCR for influenza and eight for RSV (5·6%) (Table). Three Wellington/1/2004-like influenza A (H3N2) (the predominant strain in the United Kingdom during the winter [6]) and one Shanghai/361/2002-like influenza B virus were recovered following the culture of four of the positive specimens. The NHS Direct samples included the second community sample of influenza A (H1N1), the fourth of influenza A (H3) and the first influenza B sample tested by the national reference laboratory during the 2004/2005 influenza season. The overall positivity rate of the NHS Direct samples (16%) was lower than that of the established HPA virological surveillance scheme (26%), although the peak positivity for both schemes was during the first week of February 2005.
Table. Laboratory results of the 142 NHS Direct samples returned to the national reference laboratory (1 November 2004 to 11 February 2005)
This study has demonstrated that people can self-sample in a reasonable time-frame and that these samples provide good viability for antigenic characterization and molecular detection. This reduces the requirement for the medicalisation of sample taking. This study tested a mechanism whereby self-testing by callers could augment syndromic surveillance. Self-sampling by NHS Direct callers provided amongst the earliest reports of influenza circulating in the community and detected multiple strains of the virus.
The majority of the callers who returned samples reported no problems in taking the test. This supports evidence regarding the acceptability of self-sampling in other areas (e.g. sexually transmitted infections [Reference Wiesenfeld7]). We suggest that this methodology will allow novel approaches to be developed for surveillance of infectious disease using near-patient tests, and antimicrobial resistance. The detection of RSV highlights the scheme's potential to detect other viruses, and it would be worth exploring possibilities for surveillance of gastrointestinal infections with known surveillance bias (e.g. norovirus [Reference Wheeler8]), or where vaccines are being considered (e.g. rotavirus [9]).
ACKNOWLEDGEMENTS
We thank the call handlers, health information advisers and nurses at the Hampshire and Isle of Wight, West Midlands, and South Yorkshire and South Humberside NHS Direct sites for their help with this work. Thank you to Carol Sadler, Ruth Reith and Adriana Alvarez for assistance with technical work.
DECLARATION OF INTEREST
None.



