Introduction
Outpatient parenteral antimicrobial therapy (OPAT) is a commonly used management strategy that allows patients to receive intravenous therapy in the outpatient setting [Reference Tice1]. Approximately 250 000 patients receive OPAT in the USA each year, and OPAT usage is likely to increase given the focus on efficiency in healthcare management [Reference Williams2]. OPAT offers advantages to both the patient and healthcare provider, including accelerated physical recovery, improved patient satisfaction and a decreased risk of nosocomial infection [Reference Seaton and Barr3, Reference Chapman4]. OPAT also reduces institutional costs and is cost-effective when compared with inpatient care [Reference Chapman4, Reference Wai5].
Despite OPAT's numerous benefits, 20–30% of patients discharged on OPAT are readmitted [Reference Yan6–Reference Allison8]. Efficient and effective healthcare aims to reduce readmission rates and improve outcomes. OPAT would be of even greater value if overall healthcare utilisation (HCU) could be reduced following hospital discharge including both hospital readmissions and emergency department (ED) visits. Few studies have evaluated the incidence of and risk factors associated with HCU in patients receiving OPAT. Therefore, the primary objectives of this study were to elucidate the incidence of HCU and identify factors associated with HCU among patients receiving OPAT. The secondary objective was to further understand the causes of HCU among OPAT patients based on the hypothesis that an array of patient, healthcare and society factors are associated with HCU.
Methods
Study design
This was a retrospective, matched case–control study of patients discharged on OPAT from a tertiary care medical centre in Buffalo, NY. At the time of study, no formal OPAT clinical programme was in place at the medical centre and OPAT characteristics were based on the practitioner and patient preferences. Further, an infectious disease or antimicrobial stewardship consultation was not necessary prior to be included in OPAT. The Institutional Review Board at the University at Buffalo approved the study and waived informed consent. All patients ≥18 years of age with a current procedural terminology (CPT) code for a peripherally inserted central catheter (PICC; CPT: 36569/36571) from 1 January 2011 to 31 October 2015 were identified through the administrative records. Each participant's medical record was reviewed to ensure that they met the following inclusion and exclusion criteria. Patients were included if they had an infectious disease diagnosis during the index hospitalisation and continuation of intravenous antibiotic therapy was planned at the time of discharge. Patients were excluded if the PICC was discontinued prior to discharge, the patient died during the hospital stay, the patient self-discharged against medical advice, or if the patient had received OPAT therapy within the previous 12 months (Fig. 1). Patients were also excluded if the PICC was being utilised for reasons other than the treatment of an infectious disease, if the patients had a planned readmission within 30 days of hospital discharge, or if they were transferred to a different healthcare facility.
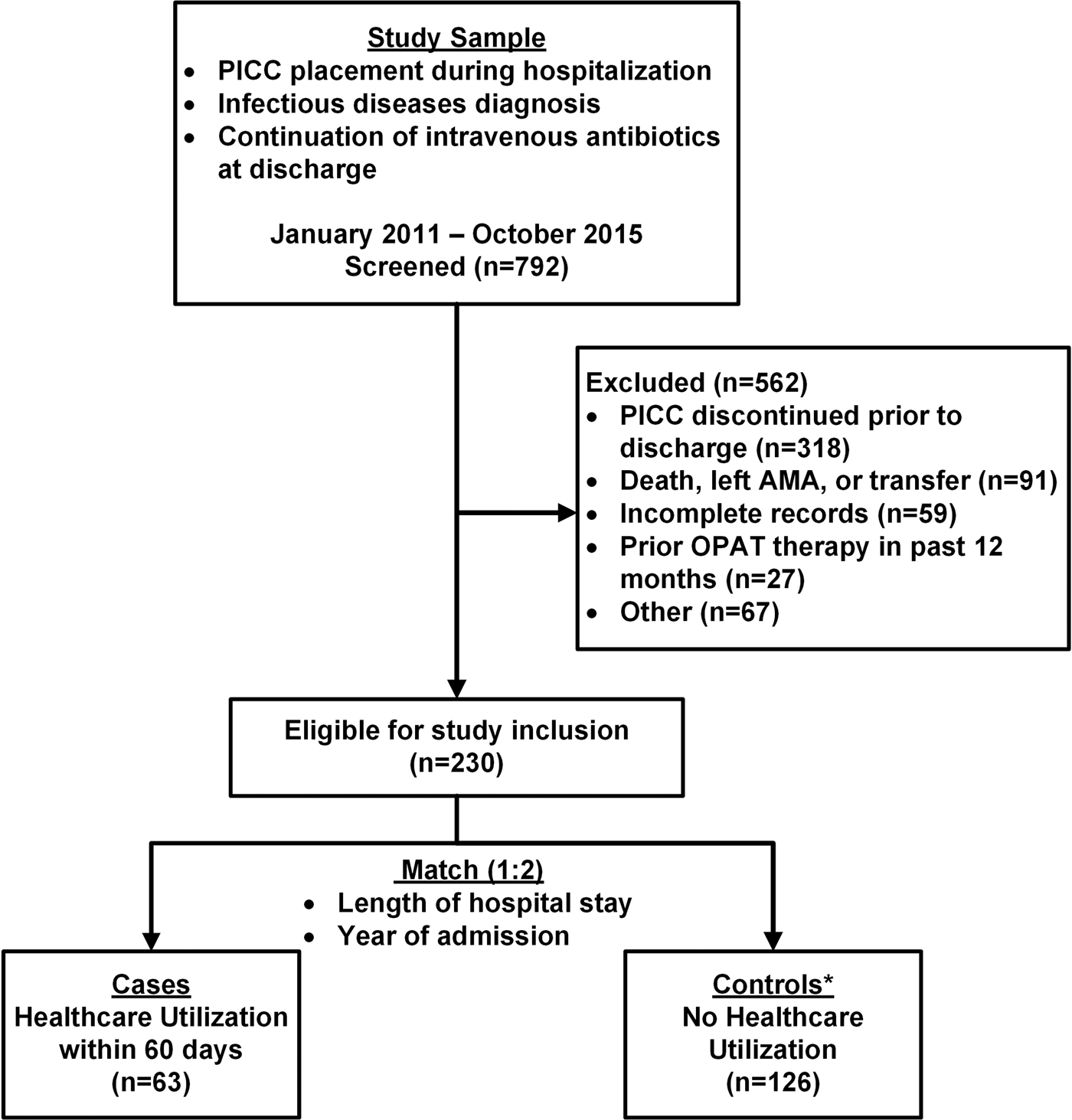
Fig. 1. Flowchart of the inclusion and exclusion process. PICC, peripherally inserted central catheter; AMA, against medical advice; OPAT, outpatient parenteral antimicrobial therapy. Other includes: PICC for reasons other than antimicrobial agent (n = 15), patient located in prison (n = 14), planned readmission within 30 days (n = 8). (*41 controls did not match cases).
Definitions
HCU patients were defined as those that utilised the healthcare system, for any reason, within 60 days of the index admission's hospital discharge date. The index admission was defined as the first hospitalisation resulting in discharge with OPAT occurring during the study period. For each case, two controls were chosen from OPAT patients that did not utilise the healthcare system within 60 days of the index admission's hospital discharge date. Controls were matched to each case based on the year of admission and length of hospital stay. HCU was defined as either an inpatient admission or presentation to the ED and was identified through the hospital's electronic medical record (EMR).
Data collection
Data were abstracted from the EMR utilising a standardised case report form and entered into a secure electronic relational database using REDCap (Research Electronic Data Capture) [Reference Harris9]. Two data abstractors reviewed each EMR independently. Any discrepancies in data collection were resolved by discussion with the data abstractors and a study investigator. Data collected included sociodemographic factors (age, social status and insurance status), Charlson comorbidity index, hospitalisation characteristics (primary service and length of stay), history of drug-resistant organisms, specific antibiotics, infectious disease diagnosis and reasons for HCU [Reference Charlson10]. History of drug-resistant organisms included a positive culture within 12 months of index admission for the following organisms: (1) carbapenem-resistant Enterobacteriaceae; (2) extended-spectrum β-lactamase producers; (3) vancomycin-resistant enterococci; and (4) methicillin-resistant Staphylococcus aureus. Antibiotics were evaluated based on antibiotic class, and infectious diagnosis categories were based on previously published work [Reference Allison8]. The following medications at discharge were recorded: (1) antiplatelet agents (aspirin or clopidogrel), (2) anti-coagulants (warfarin, low molecular weight heparin, new oral anticoagulant) and (3) HMG-CoA reductase inhibitors (statins). Medications were selected a priori to represent factors that may be associated with PICC-related adverse events. [Reference Chopra11]
Statistical analysis
Patient demographics and medication use were characterised during the index hospital stay. Continuous variables were presented as mean ± standard deviation (s.d.) and compared using the Student's t-test for normally distributed variables or median (interquartile range (IQR)) and compared using the Mann–Whitney U test for non-normally distributed variables. Normality was assessed using the Shapiro–Wilk test. Categorical variables were described as number (percentage) and compared using the chi-squared test. Crude incidence rates of 30- and 60-day HCU were estimated for hospital readmissions and ED visits. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated using logistic regression. To identify independent risk factors for 60-day HCU, variables associated in the bivariate analysis with P < 0.2 were entered into a conditional multivariable logistic regression model. Backward elimination was used for the final multivariable model, and variables were retained if they were independently (P < 0.05) associated with 60-day HCU. All analyses were performed in SAS version 9.4 (SAS Institute, Cary, NC).
Results
Study inclusion and exclusion details are presented in Figure 1. Overall, 230 patients were eligible for the study. The incidence rate of 60-day HCU following hospital discharge was 27%. Within 30-days of hospital discharge, 11% (26/230) of patients visited the ED and 9.6% (22/230) were readmitted to the hospital. Between days 31 and 60 following discharge, 2.8% (5/182) and 5.6% (10/182) visited the ED or were readmitted to the hospital, respectively.
Sixty-three patients were identified as cases and matched to 126 controls. Patient demographic and clinical characteristics are listed in Table 1. The mean age of the sample was 55 ± 16 years, and a majority of the patients were male (65%). Approximately 39% of the sample was insured by Medicare and the median Charlson comorbidity index was 3. Patients discharged home were more like to utilise the healthcare system than patients sent to a facility, though this did not reach statistical significance (i.e., home vs. skilled nursing facility or rehabilitation, 63% vs. 37%, P = 0.40). There were no significant differences between groups in terms of the type of insurance (Medicare 38% vs. 40%, P = 0.45) home support status (lives with adult: 59% vs. 48%, P = 0.46), and Charlson comorbidity index (P = 0.41). Having a history of a drug-resistant organism was slightly higher in the case group (16% vs. 8%, P = 0.09). The most frequently prescribed antimicrobials were cephalosporins (36%) and vancomycin (33%) (Table 2). The most common infectious disease diagnoses were wound infections (22%), cellulitis (14%) and bacteraemia (13%). At discharge, cases were less likely to be prescribed a statin alone (19% vs. 37%, P = 0.01) or both a statin and antiplatelet agent (13% vs. 22%, P = 0.12).
Table 1. Demographics and clinical characteristics of cases and controls receiving outpatient parenteral antimicrobial therapy
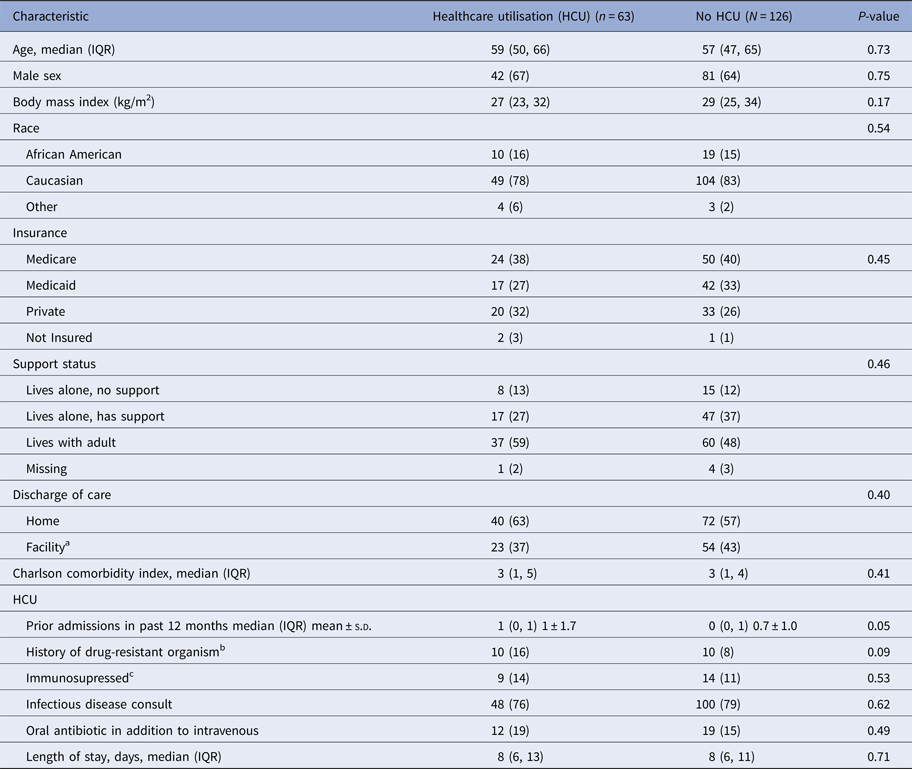
IQR, interquartile range, MRSA, methicillin-resistant S. aureus; VRE, vancomycin-resistant Enterococcus; ESBL, extended spectrum beta-lactamase; KPC, Klebsiella pneumoniae carbapenemase.
Data are presented as N (%) and median (IQR) unless otherwise specified.
a Includes rehabilitation, skilled nursing facility and other.
b Within 12 months of index discharge and includes MRSA, VRE, ESBL-producing organism, KPC-producing organism.
c Includes a history of bone marrow transplant, solid organ transplant, malignancy or HIV/AIDS.
Table 2. Bivariate analysis of cases and controls by antimicrobials, infectious disease diagnosis and concurrent medications at discharge
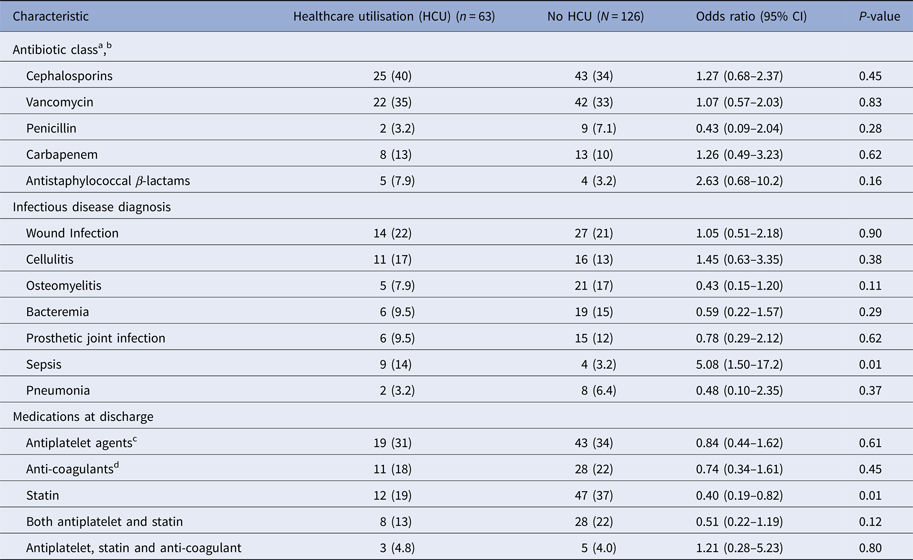
a Antibiotics used that were not included in the table (No.): daptomycin (9), piperacillin/tazobactam (9), and aminoglycosides (9), tigecycline (6), other (4), ampicillin (4), metronidazole (2), fluoroquinolones (1).
b Includes cefazolin, ceftriaxone, cefepime and ceftaroline.
c Includes clopidogrel and aspirin.
d Includes warfarin, enoxaparin, heparin, dabigatran, apixaban, endoxaban and dalteparin.
The following variables met a priori inclusion criteria of a significance level of P < 0.2 and therefore were included in the initial multivariable model: body mass index (continuous), number of admissions in previous 12 months, history of drug-resistant organisms, one antibiotic class (anti-staphylococcal β-lactams), two diagnosis categories (osteomyelitis and sepsis) and two medication groups (statins alone and combination antiplatelet agent and statin). The final regression model consisted of being prescribed a statin at discharge (aOR 0.23; 95% CIs 0.09–0.57, P < 0.01), number of admissions in previous 12 months (aOR 1.48; 95% CIs 1.05–2.10, P = 0.03), and a diagnosis of sepsis (aOR 4.62; 95% CIs 1.23–17.3, P = 0.02) (Table 3). In a sensitivity analysis, we examined predictors associated with 30-day HCU. Being prescribed a statin at discharge (aOR 0.39; 95% CIs 0.16–0.98, P = 0.046) and a diagnosis of sepsis (aOR 6.65; 95% CIs 1.69–26.2, P = 0.01) was associated with 30-day HCU. The number of admissions in the previous 12 months trended towards statistical significance (aOR 1.28; 95% CIs 0.88–1.87, P = 0.19).
Table 3. Independent risk factors for healthcare utilisation following OPAT discharge
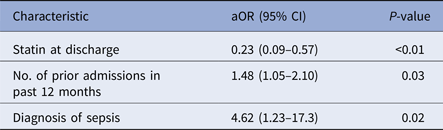
aOR, adjusted odds ratio; CI, confidence interval.
The major reasons for HCU included non-infection related issues (44%) and worsening of the primary infection (31%) (Table 4). Non-infection-related problems included uncontrolled comorbidities (16%), uncontrolled pain (11%), and other factors (17%). The uncontrolled comorbidities included those related to cardiovascular (n = 3) and respiratory (n = 3) diseases. The other non-infection-related factors were dehydration (n = 2), anaemia (n = 2), and gastrointestinal complications (n = 2).
Table 4. Reasons for healthcare utilisation (HCU)a (n = 63)
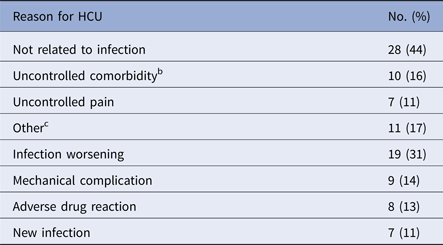
a Patients may have more than one reason to utilise the healthcare system.
b Uncontrolled comorbidities include: cardiovascular disease (n = 3), respiratory disease (n = 3), diabetes (n = 2), acute renal failure (n = 1) and pneumocephalus (n = 1).
c Other include: dehydration (n = 2), anaemia (n = 2), gastrointestinal complication (n = 2), fracture (n = 1), haematuria (n = 1), facial oedema (n = 1), chronic flare (n = 1) and caregiving problems (n = 1).
Discussion
In this study of 230 patients receiving OPAT via a PICC, the overall 60-day HCU incidence rate was 27%, in the range found in previous OPAT studies [Reference Yan6, Reference Means7, Reference Duncan12, Reference Huck13]. Yan et al. recently conducted a single-centre retrospective cohort study of 104 patients discharged on OPAT and reported an all-cause 60-day HCU rate of 43%, 26% of whom were readmitted to the hospital [Reference Yan6]. Their higher HCU rate may be due to the fact that the authors could collect hospital readmissions and ED visits from five neighbouring institutions, our study perhaps underestimating the true HCU incidence rate due to data collection being limited to HCU within our own facility. Allison et al. performed a retrospective cohort study to examine the patient and healthcare system factors associated with readmission for patients discharged on OPAT [Reference Allison8]. Their 30-day hospital readmission incidence rate was 26%, but they did not take ED visits into account and their analysis was limited to 30 days post-discharge. Many patients are discharged on OPAT for the treatment of osteomyelitis and endocarditis, and since the recommended treatment for these diagnoses may be greater than 4 weeks, it is important to evaluate beyond 30 days. Means et al. performed a retrospective cohort study and found that 20% of OPAT patients experienced a hospital readmission, although this study did not specify the duration of follow-up [Reference Means7]. In our study, both inpatient hospital readmissions and ED visits were included in the endpoint as both indicate an exacerbation in a patient's health status and may represent a failure of OPAT as a step-down approach.
Two factors were independently associated with a higher HCU rate: the number of admissions in the previous 12 months and a diagnosis of sepsis. Other studies have reported similar findings [Reference Yan6–Reference Allison8]. A history of hospital admissions in the preceding 12 months is not entirely unexpected as an HCU predictor since these patients could be expected to be sicker and therefore hospitalised for comorbidities related to a declining health condition. HCU is also more common in sepsis survivors due to the severity of the illness, physiological stress of hospitalisation and incurred organ damage [Reference Goodwin14].
As well as these risk factors, we also had a novel finding of decreased HCU risk in patients discharged on a statin. Chopra et al. conducted a nested case–control study to investigate the patterns, risk factors and treatments associated with PICCs and deep vein thrombosis (DVT) in hospitalised adults and found that hospitalised patients receiving a combination of aspirin and a statin were less likely to develop PICC-induced DVT [Reference Chopra11]. Our study found that the chronic use of statins was associated with a decreased HCU rate in patients discharged on OPAT. Furthermore, patients taking both an antiplatelet agent and a statin also showed a decreasing trend in HCU, though not statistically significant. The protective mechanism associated with a statin may be due to its known biological effects including anti-inflammatory, antithrombotic and pleiotropic activities [Reference Chopra11, Reference Nkemdirim Okere and Renier15, Reference Patel16]. Interestingly, of the nine patients readmitted due to mechanical problems or thrombosis related to PICC, eight were not regularly taking a statin. Further research is necessary to elucidate whether concurrent medications impact on OPAT-related outcomes.
There were numerous reasons for HCU, the major ones being non-infection-related problems and worsening of the primary infection. These reasons for HCU have been seen in previous studies but without details of the non-infection-related reasons [Reference Yan6, Reference Allison8]. The most commonly seen non-infection-related problem in our study was an exacerbation of chronic comorbidities, which was expected as more severely ill patients tend to utilise the healthcare system more frequently. The second non-infection-related reason was associated with pain control. Opiate therapy is common among medical inpatients and has been shown to be associated with hospital readmission [Reference Mosher17]. Given the prevalence of opioid therapy, it is important to understand and improve the utilisation and long-term health outcomes associated with hospitalisation.
The main limitation of this study is its retrospective nature, which includes the biases and confounders implicit in this type of study. The study relied on CPT codes to identify OPAT patients and the validity of these codes has not been formally assessed. Further, useful information such as socioeconomic status and treatment adherence was often not available in the medical records and was therefore omitted. The statistical power to detect differences between cases and controls is limited given our sample size. A larger follow-up study including multiple medical centres is necessary to further understand risk factors related HCU among OPAT patients. In addition, we did not have integrated medical record access to other health systems. Thus, this restricted data to only our institution and limited our ability to collect HCU at other healthcare facilities. It is possible that we did not capture all HCU, which would underestimate the true HCU incidence rate.
Conclusion
HCU including hospital readmissions and ED visits was high in patients receiving OPAT via a PICC. A formal OPAT clinic to proactively monitor and follow-up the care of these patients may reduce the unnecessary use of inappropriate healthcare resources. Although multiple HCU risk factors were found, further work is needed to risk stratify these patients and target the highest risk groups. OPAT represents an opportunity to improve patient care but requires a coordinated and evidence-based approach to reduce unwanted outcomes.
Acknowledgements
This study was supported by a grant from Research for Health in Erie County, Inc.
Declaration of interest
Authors have nothing to declare.








