Conditions within tropical urban slums, characterized by low-level infrastructure, inadequate sanitation, and poor access to safe water, contribute to the transfer of zoonoses to humans. Within Brazilian slums, these conditions have been shown to contribute to the spread of harmful zoonoses such as leptospirosis [Reference Costa1]. Many other infectious organisms exist within these urban areas, capable of posing substantial risk to the health of the resident human population. The global numbers of slum dwellers is expected to reach 1·4 billion by 2020, with Latin America and the Caribbean having 110·7 million (23·5%) of its population living in these sub-standard conditions [Reference Costa1]. Tropical urban areas are therefore important environments in which to study zoonoses, in an attempt to identify and quantify their risk to humans, and to develop approaches that prevent and control the spread of disease to inhabitants of urban slums.
Calodium hepaticum (Brancroft, 1893) (syn. Capillaria hepatica, Tricocephalus hepaticus, Hepaticola hepatica) is a globally distributed, zoonotic nematode that causes hepatic capillariasis in humans [Reference Fuehrer, Igel and Auer2] and is understudied in tropical urban environments. Most research concerning C. hepaticum has been restricted to temperate areas [Reference Costa1–Reference Childs, Glass and Kroch4]. C. hepaticum has more than 180 documented mammalian hosts, with the Norway rat (Rattus norvegicus) considered to be the most important [Reference Fuehrer5]. Prevalence of C. hepaticum has been reported as high as 87·4% in Baltimore, USA, and >50% within populations of R. norvegicus sampled in Salvador, Brazil [Reference Galvão6]. C. hepaticum infects the liver of its host and requires the death of the host to release the unembryonated ova into the environment [Reference Farhang-Azad3]. With sufficient time and suitable conditions of temperature, oxygen tension and humidity, eggs embryonate after 5–7 weeks [Reference Farhang-Azad3]. Infection then occurs when contaminated soil, water, or vegetation are ingested [Reference Fuehrer, Igel and Auer2].
The result is hepatic infection, where C. hepaticum invades the hepatic parenchyma causing visible inflammatory lesions along with necrosis and fibrosis of the liver. Humans usually present with a characteristic triad of symptoms; high fever, liver enlargement and severe eosinophilia. In serious cases infection can be fatal. Unembryonated eggs are passed through the digestive tract and excreted in the faeces. Both infection types in humans are associated with unsanitary living conditions and high prevalence of C. hepaticum in synantrophic rat populations [Reference Fuehrer, Igel and Auer2]. Over 70 documented cases of human hepatic infection have been reported worldwide, a figure widely believed to be an underestimate, with most cases reported in children [Reference Fuehrer, Igel and Auer2].
Studies of C. hepaticum in Norway rats from urban areas in the tropics have served to determine the prevalence within rat populations, or focused on histopathological effects of the parasite on its host [Reference Galvão6–Reference Simões8]. Thus, the present study systematically evaluates how environmental and demographic factors affect the carriage and level of infection intensity of C. hepaticum in Norway rat populations from an urban slum environment of Salvador, Brazil. Additionally, levels of environmental contamination were assessed in an attempt to better understand the relationship between carriage of C. hepaticum in rats and contamination of the environment. This information will be used to inform risk areas within urban slum environments, in which transmission of C. hepaticum to humans may be possible.
From May 2013 to December 2014, Norway rats were captured from the neighbourhood of Pau da Lima in Salvador, Brazil (13° 32′ 53·47″ S; 38° 43′ 51·10″ W). This area, which is occupied by communities already participating in an active leptospirosis surveillance programme [Reference Costa1], has three distinct valleys (1, 2, 3). This site is typical of a slum community, and ~30% of the populations from Salvador (and Brazil) reside in similar social and environmental conditions. Live trapping of rats using Tomahawk® traps (40·6 × 12·7 × 12·7 cm) (Tomahawk Live Traps, USA) took place in all three of the valleys, with traps being set at sunrise and collected at sunrise the following day. Methods for transporting animals to the outdoor processing site, handling and obtaining tissues were as described previously [Reference Costa1]. Animal-specific factors including weight, body length, sex and the number of open wounds were recorded. Recorded weights of the animals were converted to ages (days) using the von Bertalanffy equation [Reference Burthe9] and previously published data. Moreover, body condition was estimated using a scaled mass index based on mass and body length, while accounting for the effect of age [Reference Peig and Green10]. Environmental variables including location of capture, the presence of an open sewer within 10 m of the capture location, the number of trees at the capture location and number of alternative hosts of C. hepaticum, were recorded at the time of live capture. Data regarding the number of trees and the number of alternative hosts was recorded per household in cases when the capture location was inside the resident's grounds. Trees were recorded within a 10-m radius from the trap location and both cats and dogs were recorded as alternative hosts of C. hepaticum.
The presence and absence of C. hepaticum were evaluated through macroscopic examination of the liver during necropsies, with the presence of C. hepaticum being noted based on visible yellowish-white lesions on the liver surface and scored as described previously [Reference Farhang-Azad3, Reference Childs, Glass and Kroch4, Reference Simões8] Percentage of the liver surface covered with lesions was used as a proxy for intensity level of infection and was classified as slight (1–25%), low (26–50%), heavy (51–75%) or gross (75–100%) liver involvement.
Additionally, soil samples were evaluated for evidence of environmental contamination of C. hepaticum. Using a cross-sectional design, ten locations previously used to trap rodents were randomly selected using a random number generator, and six samples of soil (30 g) were collected at each location during August 2015. Those 10 locations represented ~10% of the total number of capture sites and were homogeneously distributed across the study area. All ten locations were in close proximity to places of residence and were easily accessible by humans, making them potential risk areas for humans. Furthermore, given that monthly rainfall and temperatures are consistently high year round, environmental factors are not limiting egg embryonation at capture locations. Samples were processed as described previously [Reference Sudhakar11].
A logistic regression model was performed using R v. 2.11.1 (R Foundation, Austria), to examine the relationship between carriage of C. hepaticum and explanatory variables. An ordinal logistic regression model was used to examine the relationship between the infection level of C. hepaticum and the explanatory variables. For multivariate analysis, variables associated in univariate models with P < 0·2 were included and the models were then judged based on Akaike's Information Criterion (AIC). This allowed for the selection of a statistical model that best fitted the data available.
The carriage of C. hepaticum was assessed for 402 synanthropic rats. The carriage level was 83% (337/402) in the Norway rat. In univariate analyses, carriage tended to increase with age, location (valley) of capture, proximity (within 10 m) to an open sewer and the number of dogs recorded at households next to capture locations. Multivariate analysis supported strongly the inclusion of age and location (valley) of capture. This model formula produced an AIC score of 228·5, of which was the lowest AIC score relative to the other models that were assessed (Table 1, Supplementary Table S1). This model was therefore the best fit to the available data. The age of an individual was a significant predictor of whether or not an individual was carrying C. hepaticum, after controlling for the location of capture, i.e. valley [unadjusted odds ratio (OR) 1·03, 95% confidence interval (CI) 1·02–1·05)]. Of individuals that were sexually mature, i.e. aged ⩾65 days, 91% (235/261) of the individuals were infected compared to 69% (97/141) of juvenile rats. The increased probability of infection in older individuals may be due to higher levels of activity and therefore an increased likelihood of coming into contact with infective ova, but may simply be the result of cumulative exposure. Once the parasite has infected the hepatic parenchyma infection remains for the duration of the individuals’ lifetime [Reference Farhang-Azad3]. In valley 1, 81% (65/80) of R. norvegicus individuals were infected compared to 92% (144/157) captured in valley 2 and 75% (123/165) captured in valley 3. Univariate analysis showed the presence of an open sewer within 10 m of the capture location, to be a significant predictor of carriage of C. hepaticum within individuals (unadjusted OR 1·97, 95% CI 1·01–3·83) (Table 1). Open sewers run along the bottom of valleys and it is possible that heavy rainfall, typical in the region between the months of April and July, could wash infective ova into low-lying areas, increasing the environmental concentration of ova and therefore the probability of infection. However, the inclusion of this variable in the multivariate analysis was not strongly supported (Table 1, Supplementary Table S1).
Table 1. Unadjusted and adjusted logistic regression predicting carriage and intensity of infection of Calodium hepaticum within individuals of Rattus norvegicus
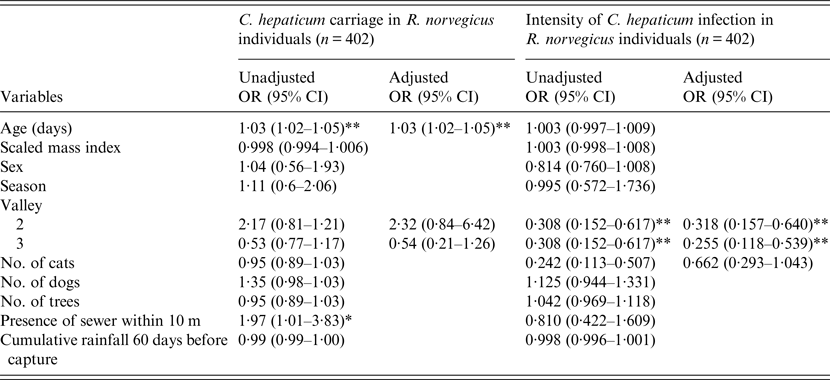
OR, Odds ratio; CI, confidence interval.
* P < 0·05, ** P < 0·001.
Regarding intensity, of the infected individuals 71% (237/337) were classified as having a slight level of infection (<25% liver involvement) and only 8% (26/337) had gross liver involvement. Multivariate analyses supported the inclusion of valley, i.e. location of capture and cats, an alternative host of C. hepaticum. This model formula was shown to be the best fit for the data available, producing an AIC score of 371·48, the lowest score relative to the other evaluated models (Table 1, Supplementary Table S1). Multivariate analyses showed location of capture, i.e. valley, to be a significant predictor of infection intensity after adjusting for the number of cats recorded at the residence where capture points were located (Table 1). The odds of individuals possessing higher levels of infection intensity were significantly greater in valley 1 compared to those in valley 2 (adjusted OR 0·318, 95% CI 0·157–0·640) and valley 3 (adjusted OR 0·255, 95% CI 0·118–0·539) (Table 1). This difference between valleys has no obvious explanation. Previous work has shown higher levels of C. hepaticum carriage within populations of R. norvegicus to be associated with higher the levels of infection intensity [Reference Farhang-Azad3]; however, carriage level did not show a significant difference between valleys in either the univariate or multivariate models (Table 1). It is possible that unexamined factors, including the effects of co-infection with other parasites and the health of the individual hosts could be contributing to the observed result.
Data regarding environmental contamination of C. hepaticum revealed the proportion of samples contaminated with C. hepaticum was low (10%, 6/60) as were the number of eggs found in positive samples (mean 3·25, range 1–7). These results suggest C. hepaticum is broadly distributed within the area but at low environmental concentrations. A more systematic evaluation of the environmental levels of C. hepaticum would be necessary to determine how the prevalence of C. hepaticum in rats translates to the levels in the environment, thus providing a better indication of the risk to humans of acquiring this infection. It is possible that infected rats are eaten by other animals (dogs, cats, etc.) prior to decomposition, affecting the distribution of environmental C. hepaticum. Therefore additional evaluation of alternative and intermediate hosts may also be of benefit.
This study demonstrates the role of specific demographic and environmental factors in predicting both the carriage and intensity levels of C. hepaticum in R. norvegicus in an urban slum. Additionally, the confirmation of environmental contamination suggests that humans could be at some risk of becoming infected with C. hepaticum. The extent of which, however, is unknown without further investigation. This is the first published report of C. hepaticum prevalence in R. norvegicus from Salvador since 1976 when carriage in captured rats was 57% [Reference Galvão6]. The carriage level of 83% determined by this study is higher than the most recent documentations of C. hepaticum in R. norvegicus from cities in Brazil: Rio de Janerio (47%) [Reference Simões8] and Porto Velho (2%) [Reference Rocha7]. This is potentially due to the high host density and year round conditions suitable for egg embryonation. There is a lack of epidemiological studies of C. hepaticum in humans and such studies are needed given the high levels of C. hepaticum carriage within the rat population. Further study is required to assess whether humans are becoming infected within this community and whether C. hepaticum is posing a significant risk to human health.
The Institutional Animal Care and Use Committees at the Oswaldo Cruz Foundation, Salvador, Brazil (003/2012) and Yale University in the United States (2012–11 498) approved all animal procedures and methods.
SUPPLEMENTARY MATERIAL
For supplementary material accompanying this paper visit http://dx.doi.org/10.1017/S0950268816002259.
ACKNOWLEDGEMENTS
The authors thank the staff of Zoonosis Control Centre from Salvador for their assistance in conducting the study; Luciano Lima, Roberval Arcanjo, Alex Maia, Alex Márcio for their critical advice during the preparation of the manuscript and fieldwork. This work could not have been accomplished without the joint collaborative effort of the resident associations, community leaders and residents, which constitute the Urban Health Council of Pau da Lima.
This work was supported by the Oswaldo Cruz Foundation and Secretariat of Health Surveillance, Brazilian Ministry of Health, the National Institutes of Health (grants F31 AI114245, R01 AI052473, U01 AI088752, R01 TW009504 and R25 TW009338) and by the Wellcome Trust (102330/Z/13/Z).
DECLARATION OF INTEREST
None.



