Group A streptococcus (GAS) is one of the most common human pathogens and causes a wide array of infections, the most frequent of which is acute pharyngitis (strep throat). Many streptococcal virulence factors have been implicated in the pathogenesis of streptococcal infection including pyrogenic exotoxins (SpeA, SpeB and SpeC) and M protein. M protein protects GAS from phagocytosis by polymorphonuclear leukocytes [Reference Hortsmann1, Reference Fischetti2]. More than 90 of M serotypes have been identified, and a molecular approach to identification of emm (M protein) genes has also been documented [Reference Beall, Facklam and Thompson3]. From the late 1980s, severe invasive infections such as streptococcal toxic shock-like syndrome (TSLS) and necrotizing fasciitis (NF) caused by GAS became a serious problem in both developed and developing countries. The first defined case of TSLS in Japan was reported in 1992 [Reference Shimizu4]. In almost all of the countries which have surveyed the prevalence of M serotypes, strains of emm3 and emm1 genotypes are dominant in causing severe invasive infections. In Japan, where periodic sampling surveys are performed on a 5-year cycle, emm1 remained the most frequent from1992 to 2000, but emm3 strains which predominated from 1992 to 1995 decreased during 1996–2000 [Reference Ikebe5].
In this study, we report the frequency of genotypes of isolates from patients with severe invasive streptococcal infections, TSLS and/or NF, from 2001 to 2005 in Japan and compare these data with the preceding sampling period from 1996 to 2000.
Information on patients with severe invasive streptococcal infections and emm genotypes of the isolates during the period of 2001–2005 was submitted to the National Institute of Infectious Diseases from the branch offices of reference centre and cooperative hospitals [Reference Ikebe5]. The diagnostic criteria of TSLS were based principally on those described by the Working Group on Severe Streptococcal Infections (1993) [6].
A total of 137 GAS isolates were cultured predominantly from sterile sites of patients with severe invasive infections during the period of 2001–2005. The ratio of each genotype compared between the two sampling periods is given in the Figure. Isolates of emm1 remained dominant over the two periods, accounting for 50 out of 93 (53·8%) cases in 1996–2000 and 57 out of 137 (41·6%) cases in 2001–2005; there was a slight reduction in the ratio of emm1 to other genotypes. On the other hand, emm3 isolates increased dramatically from 1·08% (1/93) to 9·49% (13/137) (P<0·01). It is noteworthy that this genotype was already prevalent (33%) among TSLS isolates more than 10 years previously [Reference Ikebe5] and despite the marked decrease in the 1996–2000 sampling period, its prevalence is clearly increasing in more recent years. Fewer than seven emm genotypes were isolated each year in 1996–2000, while 8–15 emm genotypes were recorded in the subsequent years. A total of 15 emm genotypes were isolated during 1996–2000 compared with 23 emm genotypes in the later period; 10 genotypes (emm58, emm60, emm73, emm78, emm91, emm103, emm112, st2147, stg485 and stg652) were isolated from patients in the later period but were absent in the first sampling period (Fig.). These results indicate the change and diversity of genotypes over time in Japan.
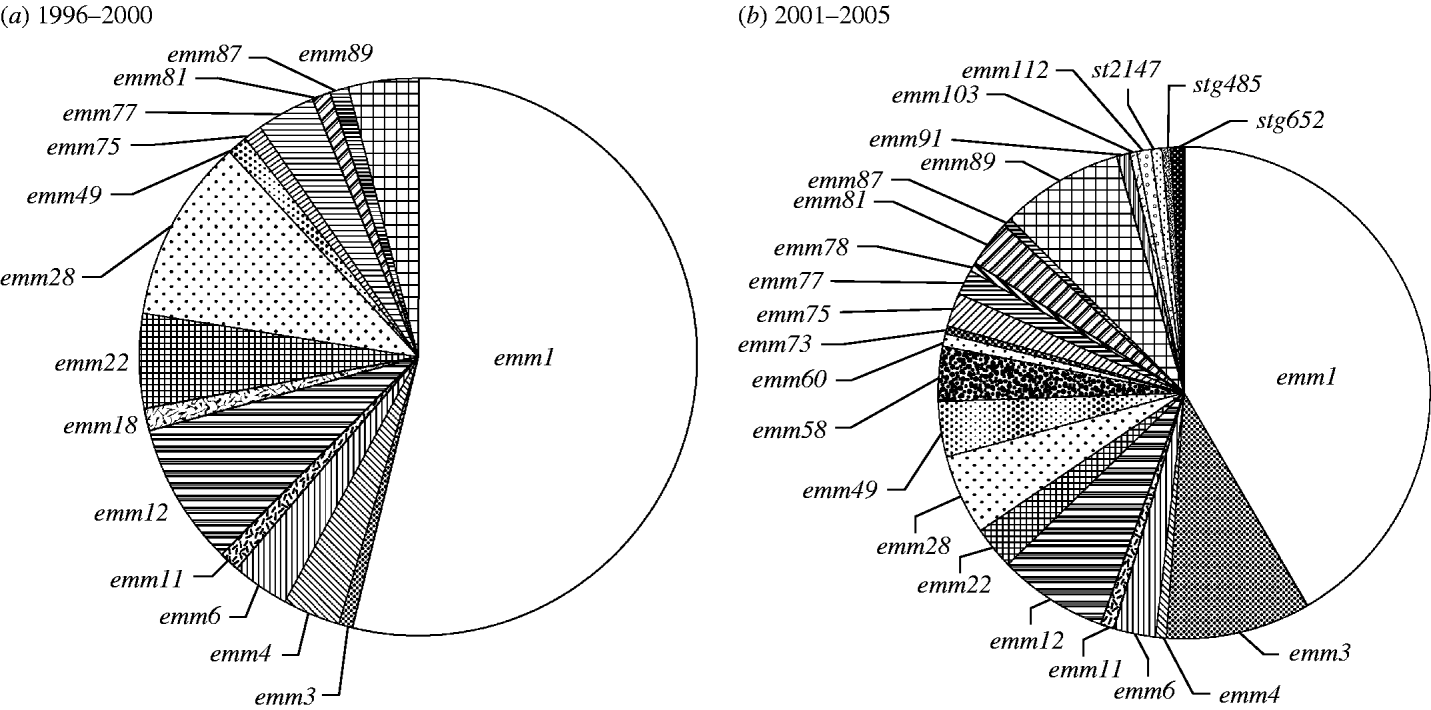
Fig. The rate of emm genotypes of strains isolated from patients of severe invasive streptococcal infections. (a) 1996–2000, (b) 2001–2005. (Number of each emm genotyped isolate during each period/number of severe invasive infections isolates during each period.)
On the basis of epidemiological data demonstrating that the majority of non-invasive and invasive streptococcal infections are caused by a limited number of M-types, a multivalent vaccine containing amino-terminal fragments of M proteins from 26 different serotypes of GAS has recently been developed in the United States [Reference McNeil7]. Based on the seroprevalence data presented here for Japanese GAS from invasive disease the vaccine would provide cover for 87 out of 93 strains (93·5%) isolated in 1996–2000 and for 112 out of 137 (81·8%) in 2001–2005. These data therefore have important implications for the formulation and composition of candidate vaccines against streptococcal infections in Japan.
APPENDIX. Working Group for Beta-haemolytic Streptococci in Japan
The following are Group members: T. Kawai (Sapporo City Institute of Public Health, Sapporo), T. Konno (Akita Prefectural Institute of Public Health, Akita), S. Fujii (Research Institute for Environmental Sciences and Public Health of Iwate Prefecture, Iwate), K. Ootani (Yamagata Prefectural Institute of Public Health, Yamagata), N. Numata (Sendai City Institute of Public Health, Sendai), H. Tamura (Miyagi Prefectural Institute of Public Health and Environment, Miyagi), M. Tanaka (Niigata City Institute of Public Health, Niigata), H. Terasawa (Niigata Prefectural Institute of Public Health and Environmental Sciences, Niigata), K. Kimura (Hokkaido Institute of Public Health, Hokkaido), M. Matsumoto (Aichi Prefectural Institute of Public Health, Nagoya), M. Sakaki (Hiroshima Prefectural Institute of Public Health and Environment, Hiroshima), K. Yoshida (Ehime Prefectural Institute of Public Health and Environmental Science, Ehime), M. Kinuta (Public Health Institute of Kochi Prefecture, Kochi), T. Mori (Tokushima Prefectural Institute of Public Health and Environmental Sciences, Tokushima), K. Kishikawa (The Saga Prefectural Institute of Public Health and Pharmaceutical Research, Saga), J. Kudaka (Okinawa Prefectural Institute of Health and Environment, Okinawa), N. Tosaka (National Institute of Infectious Diseases, Tokyo), M, Ueno (National Institute of Infectious Diseases, Tokyo).
ACKNOWLEDGEMENTS
We are grateful to S. Matsunaga for excellent technical assistance. This work was supported in part by a grant from the Ministry of Health, Labor and Welfare.
DECLARATION OF INTEREST
None.



