Background
Bronchiolitis is the leading cause of infant hospitalizations in North AmericaReference Njoo, Pelletier and Spika 1 - Reference Shay, Holman and Newman 4 and results in a significant health burden on infants, their families, and the health care system.Reference Njoo, Pelletier and Spika 1 - Reference Mansbach, Emond and Camargo 6 Despite the common nature of bronchiolitis, its treatment remains controversial and substantial practice variation exists.Reference Mansbach, Emond and Camargo 6 - Reference Plint, Johnson and Wiebe 8 The utilization of unproven therapies and investigations for bronchiolitis management increases health care costs.Reference Schuh, Lalani and Allen 9 Several pediatric centres have demonstrated a reduction in the use of beta-agonist therapy, chest x-rays, and viral testing with implementation of a bronchiolitis clinical practice guideline (CPG).Reference Todd, Bertoch and Dolan 10 - Reference Kotagal, Robbins and Kini 13 However, most children with bronchiolitis who present to a hospital are treated and discharged from the emergency department (ED).Reference Mansbach, Emond and Camargo 6 - Reference Plint, Johnson and Wiebe 8 Many are also managed outside children’s hospitals where physicians may be less comfortable with bronchiolitis and less familiar with recent literature regarding bronchiolitis management. Little is known regarding the detailed management of children with bronchiolitis in community hospital settings both in the ED and on inpatient wards.
We examined the ED management and disposition of children presenting with bronchiolitis to community EDs across Ontario, Canada, as well as the management of children admitted to the inpatient wards at those same hospitals. Our main objectives were to compare current management to clinical guideline recommendations and examine areas of inter-hospital practice variability.
Methods
Study design and patient population
We conducted a retrospective chart review of a cohort of children who presented consecutively to 28 participating community EDs with bronchiolitis. Data were collected for two bronchiolitis seasons (December 2005 to April 2006, and December 2006 to April 2007). Community-based hospitals throughout Ontario that had 24-hour EDs and accepted pediatric patients to the ED and inpatient wards were eligible for the study. A community hospital was defined as a hospital that is not primarily a referral centre. The ethics board of the principal investigator’s institution as well those of participating hospitals approved this study.
Sampling method and data extraction
At the time of this study, 115 hospitals in Ontario had 24-hour EDs and admitted children to the ED and inpatient wards. We contacted hospitals located within seven provincial health districts, representing 76 hospitals. These health districts represented a wide geographic distribution and were considered feasible to visit from the coordinating site (in Ottawa, Ontario). Medical directors of the 76 hospitals were sent a letter explaining the study. We then attempted contact by telephone to confirm hospital eligibility and obtain participation. Since this project examined practice variation in smaller communities where physicians may practice at more than one hospital within the community, only one hospital per community was eligible, in order to prevent the over-representation of one or more physician’s practice patterns. While all hospitals within a given community were approached, we planned to randomly select hospitals for inclusion if more than one hospital per community was eligible and interested in participating.
At participating hospitals the medical records for all children less than 12 months of age treated in an ED or inpatient setting and discharged with a diagnosis of bronchiolitis (ICD-10 codes J21, J21.0, J21.8, J21.9), RSV (respiratory syncytial virus), wheezing, asthma, or reactive airways (ICD-10 codes B97.4, J12.1, J20.5 J45.00, J45.01, R06.2) were retrieved. Given the number of sites, a single trained data record analyst visited all sites to complete data extraction. Prior to data extraction, the data record analyst and principal investigator independently reviewed and then discussed a sample of training charts to ensure accurate data extraction. The analyst reviewed the retrieved charts to identify eligible patients. Children were included if they met the study definition of bronchiolitis. Bronchiolitis was defined as the first episode of wheezing in a child less than 12 months of age, associated with signs of an upper respiratory tract infection (such as fever, coryza, cough) during the period deemed to be peak season for RSV bronchiolitis (December to April). This definition was operationalized by including only children who had wheezing on physical examination documented on their medical record at the time of their index ED visit. As well, children were excluded if a previous diagnosis of asthma, any previous episodes of wheezing or cough treated with bronchodilators or inhaled steroids, or any previous episodes of wheezing diagnosed by a physician were documented on their ED medical record or elsewhere in their hospital chart. Data were extracted to an electronic data form with built-in prompts to ensure data quality. If a child had more than one ED visit during their illness, data regarding presentation, medications, investigations, and disposition were collected for each visit.
We also interviewed each study site’s medical director or operations director to determine whether a bronchiolitis clinical guideline was in use at their site.
Key outcome measures
Data regarding age of child, gestational age at birth, sex, medications on ED presentation, and clinical presentation (presenting oxygen saturation, respiratory rate, and heart rate) were collected. The main outcome measure of interest was patient disposition (admission, discharge, and transfers to other hospitals). Other outcome measures were ED and inpatient ward management, including medication usage and investigations, and discharge medications. Medications examined included bronchodilators, corticosteroids, and antibiotics. Investigations examined included viral testing, chest x-rays, blood tests (including electrolytes, urea, creatinine, complete blood count, and blood cultures), and urine tests (including routine analysis, microscopy, and culture). Repeat ED visits for bronchiolitis symptoms in the 21 days following the index ED visit were also reviewed. We chose 21 days following the index ED visit, as bronchiolitis symptoms often last up to three weeks.Reference Plint, Johnson and Wiebe 8 , Reference Plint, Johnson and Patel 14 , Reference Swingler, Hussey and Zwarenstein 15
Data analysis
Patient demographic and clinical characteristics on ED presentation were described using means and standard deviations for continuous variables (or median and interquartile range in the case of skewness), and frequencies and proportions for categorical variables. The main outcome measures were described using frequencies and proportions with 95% confidence intervals (CIs). Standard errors were adjusted for the intra-cluster correlation coefficient (ICC).Reference Donner and Klar 16 Outcomes relating to management were described using frequencies and proportions. To describe inter-hospital variation in outcomes, we calculated the proportion of patients with each outcome at each hospital and then the interquartile range (IQR) of these proportions across the study hospitals. The IQR was considered more meaningful than the range between hospitals, as it avoided extreme variation due to small numbers of patients at some hospitals. In addition, we calculated the ICC for each outcome using the Analysis of Variance approach.Reference Donner 17 The ICC may be defined as the between-hospital variance divided by the total variance (between plus within hospitals). Thus, it ranges between 0 and 1, where a value of 0 indicates no variation and a value of 1 indicates maximum variation among hospitals. Negative values for the ICC are usually considered as sampling error and set to 0. All analyses were conducted using SAS (version 9.3). We also intended to examine practice variation based on presence of a clinical practice guideline; however, small sample sizes precluded this analysis.
Results
Hospital characteristics
Of the 76 hospitals across the seven health districts, 28 (37%) agreed to participate. No hospital that expressed interest in participating needed to be excluded for being in the same community as another study hospital. Participating hospitals had from 6,700 to 103,000 ED visits each year. Seven participating hospitals had less than 15,000 ED visits per year, 10 hospitals had between 15,000 and 30,000 ED visits per year, and 11 had greater than 30,000 ED visits per year. The number of pediatric ED visits at participating sites ranged from 1,400 to 18,900 per year, with a median of 5,300 visits per year and a mean of 7,113 visits per year. Thirteen hospitals had less than 5,000 pediatric visits per year, nine had less than 10,000 pediatric visits per year, and six had less than 19,000 pediatric visits per year. The number of children with bronchiolitis per site ranged from two to 51 children over the study years, with a median 12 visits per site and a mean of 19.4 visits per site. Five sites had less than 10 patient visits, 11 had less than 30 visits, and the remainder had greater than 30 patient visits. Driving distances from our study centres to a pediatric referral site ranged from 40 to greater than 1,500 kilometres (km) with a mean distance of 102 km and a median of 193 km. Thirteen study sites were less than 100 km from a pediatric referral centre, 11 were less than 200 km, and four were greater than 200 km away. Seven hospitals reported having a clinical practice guideline in place for managing patients with bronchiolitis.
Patient characteristics
During the study period, our screening criteria identified 953 infants, with 543 meeting eligibility criteria. Of the 410 children excluded, 89 had previous wheezing episodes documented on their medical record, and 326 had no wheezing recorded on physical examination in the ED. The number of eligible children per site ranged from four to 51. Ten hospitals had less than 10 eligible children, 12 hospitals had between 10-30 eligible children, and six had more than 30 eligible children. Table 1 describes the clinical characteristics of the eligible children at their initial ED visit.
Table 1 Patient demographic and clinical characteristics on initial presentation to the emergency department (N=543)

* Data reported for all patients in the study except for the following variables: gestational age (n=285), medication usage on presentation (n=414), weight (n=506), temperature (n=535), oxygen saturation (n=500), heart rate (n=514), and respiratory rate (n=500).
† Temperature includes all measurement methods (per rectum, axilla, or tympanic).
Admissions, transfers, and repeat ED visits
Results regarding admissions, transfers, and repeat ED visits are presented in Table 2. Of the 543 children in the study, 161 children (29.7%, 95% CI 22.3-37.0%) were admitted to hospital. No child was admitted twice. The admission rates varied widely among sites (from 0% to 64.3%, ICC=0.086). Due to the relatively small number of children with return visits within 21 days, the remainder of results are reported for each child’s initial ED visit only.
Table 2 Admissions, transfers, and return visits to the emergency department

* Interquartile range (IQR) of the percentage of patients with the specified outcome across the study hospitals.
† Intracluster correlation co-efficient (ICC).
‡ All transfers occurred at the patient’s initial visit to the ED for bronchiolitis.
§ 31 children had 2 ED visits, 7 children had 3 visits, and 1 child had 5 visits within 21 days of index visit.
Medication usage
Medication usage in the ED, on inpatient wards, and on discharge is described in Table 3. Bronchodilator use was widespread in the ED (79.7% of patients) and on the inpatient wards (94.4% of patients). Salbutamol by metered-dose inhaler (MDI) or nebulizer was the most commonly used bronchodilator. Other bronchodilator medications used included ipratropium bromide and epinephrine. Approximately one-third of ED patients and half of inpatients were treated with corticosteroids in some form. Antibiotic use was not common in the ED, but about one-third of admitted patients were treated with antibiotics. At discharge from the ED and hospital, 46% of patients were prescribed a bronchodilator medication for use at home. Salbutamol administered by MDI was the most commonly prescribed discharge medication (35.4% of patients), but 9.3% of ED patients and 15.5% of inpatients were discharged on oral bronchodilators.
Table 3 Medications received during the initial emergency department visit, during admission, and prescribed upon discharge
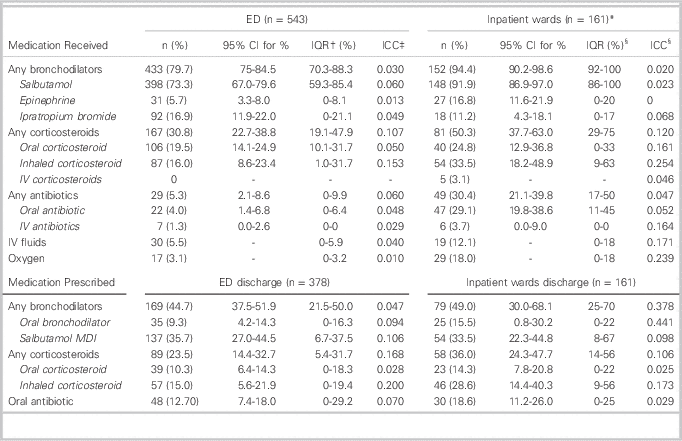
* Children admitted to the inpatient wards have their ED management described under the “ED” column and their inpatient management described separately under “inpatient” management.
† Interquartile range (IQR) of the percentage of patients with the specified outcome across the study hospitals.
‡ Intracluster correlation co-efficient (ICC)
§ Data from only 18 hospitals included in these calculations, as the remaining hospitals had no inpatient admissions.
There was substantial variation between hospitals in use of corticosteroids as well as several other interventions. Inter-hospital practice variation existed for the use of any corticosteroids (IQR 19.1-47.9%), oral corticosteroids (IQR 10.3-31.7%) and inhaled corticosteroids (IQR 1.0-31.7%) in the ED and at discharge from the ED (IQR 5.4-31.7%, 0-18.3%, 0-19.4% respectively). Similarly, there was inter-hospital practice variation in the use of any corticosteroids (IQR 29-75%), oral corticosteroids (IQR 0-33%), and inhaled corticosteroids (IQR 9-63%) on the inpatient wards and at discharge from inpatient wards (IQR 14-56%, 0-22%, 9-56% respectively). The use of oral bronchodilators (such as liquid salbutamol and orciprenaline) at inpatient ward discharge also varied between hospitals (IQR 0-22%). There was also substantial inter-hospital practice variation in use of IV fluids (IQR 0-18%), oxygen (IQR 0-18%), and oral antibiotics (IQR 11-45%).
Investigations in ED and on inpatient wards
Investigations completed in the ED and during inpatient admission are presented in Table 4. Overall, 216 children (60.2%) had at least one chest x-ray taken and 174 (32.0%) had at least one panel of viral studies completed either during their ED visit or during admission. Twenty-four children (4.4%) had more than one chest x-ray during their illness; 20 of these children were admitted and had chest x-rays during their ED visit and during their hospital admission. During their initial ED visit, 54.5% of all children had chest x-rays, 22.7% had nasal viral studies, 6.8% had at least one blood test, and 3.0% had urine tests. Among admitted children, 30% had chest x-rays, 33% had viral studies, 20% had at least one blood test, and 11% had urine tests. Large inter-hospital variability was observed for the use of viral studies, both in the ED (IQR 1.0-28.0%) and on the inpatient wards (IQR 0-36%). There was also large inter-hospital variation for obtaining blood tests on inpatient wards (IQR 10-50%).
Table 4 Investigations completed during the index emergency department visit and during admission on inpatient wards
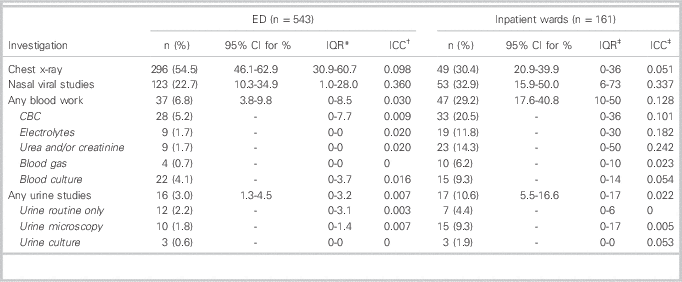
* Interquartile range (IQR) of the percentage of patients with the specified outcome across the study hospitals.
† Intracluster correlation co-efficient (ICC)
‡ Data from only 18 hospitals included in these calculations as the remaining hospitals had no inpatient admissions.
Intracluster correlation coefficients
We calculated ICCs for 66 measures (all process measures). The ICC for hospital admission was 0.086. ICCs for investigations and medications in the ED ranged from 0 to 0.36 (median 0.044, IQR 0.017-0.082) and in the inpatient wards from 0 to 0.44 (median 0.083, IQR 0.028-0.172).
Discussion
We found that children in our study region received treatment with multiple medications for bronchiolitis, many of which have never been demonstrated to show significant benefit. As has been seen in other North American ED and inpatient studies, the overwhelming majority of children were treated with salbutamol.Reference Mansbach, Pelletier and Camargo 5 , Reference Mansbach, Emond and Camargo 6 , Reference Plint, Johnson and Wiebe 8 , Reference Law and De Carvalho 18 This is despite systematic reviews published during our study time frame which found no benefit to salbutamol use in outpatient or inpatient settings,Reference Kellner, Ohlsson and Gadomski 19 and which suggested that in the ED only epinephrine, but not salbutamol, might be beneficial in reducing hospital admission.Reference Hartling, Wiebe and Russell 20 The American Academy of Pediatrics (AAP) Bronchiolitis Practice Guideline, published during the time period our cohort was treated, advised against routine use of bronchodilators, but did support a limited trial in an observed setting. 21 Research published since the initial AAP guideline reaffirms that selective beta2agonists reduce neither hospital admission rates or length of stay; there is, however, some evidence that epinephrine may reduce hospital admissions but not length of hospital stay.Reference Hartling, Fernandes and Bialy 22 , Reference Skjerven, Hunderi and Brügmann-Pieper 23 The AAP guideline has been recently revised and continues to advise against the routine use of bronchodilator (both salbutamol and epinephrine) and no longer supports a limited trial of bronchodilator use.Reference Ralston, Lieberthal and Meissner 24 The Canadian Pediatric Society (CPS) bronchiolitis guideline recommends only supportive care, reports equivocal evidence of benefit to epinephrine for outpatients, and suggests, at most, a limited observed trial of its use.Reference Friedman, Rieder and Walton 25
The use of systemic corticosteroids for ED patients was similar to studies that have included community EDs within their study population.Reference Mansbach, Emond and Camargo 6 , Reference Johnson, Adair and Brant 26 Among inpatients, corticosteroid use was double that reported at pediatric centers.Reference Ralston, Garber and Narang 27 The majority of trials and systematic reviews available at the time of our cohort and the AAP practice guideline did not suggest a benefit of corticosteroids.Reference Garrison, Christakis and Harvey 28
We found a surprising number of children were treated with medications we had not anticipated. Many children received inhaled ipratropium bromide and inhaled corticosteroids in the ED and during their admission, and were discharged from the ED and inpatient wards with a prescription for oral bronchodilators. The use of oral bronchodilators and inhaled corticosteroids in managing bronchiolitis is not supported by the available evidence.Reference Hartling, Fernandes and Bialy 22 , Reference Patel, Gouin and Platt 29 - Reference Everard, Bara and Kurian 31 While use of ipratropium bromide in bronchiolitis management has not been reported in North American studies, several European studies report its use.Reference Hartling, Fernandes and Bialy 22 , Reference Walker, Danby and Turner 32 In our study, 16.0% of children in the ED and 33.5% of children on inpatient wards were treated with inhaled corticosteroids, markedly more than the reported 6% of children treated on inpatient wards in children’s hospitals.Reference Christakis, Cowan and Garrison 33 It is possible that community emergency physicians, who may have limited experience with managing bronchiolitis, have been influenced by the medical literature showing benefit to inhaled corticosteroids in the management of croupReference Russell, Wiebe and Saenz 34 and of ipratropium bromide (in combination with salbutamol) for the management of severe asthma.Reference Plotnick and Ducharme 35 However, since we used a conservative and explicit diagnosis of bronchiolitis, it is unlikely that many, if any, of the children in this cohort actually had either asthma or croup as an explanation for the use of these medications.
Chest x-rays were commonly obtained. High utilization of chest x-rays has been seen in other studies,Reference Mansbach, Emond and Camargo 6 , Reference Plint, Johnson and Wiebe 8 including one that examined management at pediatric academic centresReference Plint, Johnson and Wiebe 8 . Neither the original, 21 the revised AAP bronchiolitis CPG,Reference Ralston, Lieberthal and Meissner 24 nor the current CPS bronchiolitis guidelineReference Friedman, Rieder and Walton 25 recommend chest x-rays. Chest x-rays have been determined to add little to management and increase the use of antibiotics.Reference Schuh, Lalani and Allen 9 , 21 Viral studies also have no clinical benefit. 21 , Reference Ralston, Lieberthal and Meissner 24 The inter-hospital variation we found may reflect differing hospital isolation policies. Blood work and urine tests were uncommonly performed in the ED but were more common among admitted children, likely reflecting either more severe illness or greater concern by the treating physician for dehydration and other illnesses. The inter-hospital variation we found in the oxygen use, IV fluids, and oral antibiotics among admitted children may reflect practice variation or differences in illness severity among children admitted at different hospitals.
Given the widespread use of medications and investigations shown to provide little benefit to bronchiolitis among both community and pediatric centres found in our study, it is interesting to consider the effects CPGs have had in improving care for children with bronchiolitis. Several studies evaluating the implementation of bronchiolitis CPGs for inpatient management at pediatric centres have found a reduction in the use of beta-agonist therapy, chest x-rays, and viral testing.Reference Todd, Bertoch and Dolan 10 - Reference Kotagal, Robbins and Kini 13 A recent study comparing children’s hospitals with bronchiolitis CPGs to hospitals without CPGs found that the presence of a CPG was not associated with less use of unnecessary medication and investigations.Reference Mittal, Hall and Morse 36 Interestingly, when change in bronchiolitis inpatient management was examined across many of these same pediatric hospitals before and after publication of the AAP bronchiolitis CPG, a significant improvement was seen in the use of chest x-rays, systemic corticosteroids, and bronchodilators over time.Reference Parikh, Hall and Teach 37 Clearly, as with many medical conditions, changing bronchiolitis care is a complex endeavour. There is evidence that exposure to increased volumes of patients with bronchiolitis is associated with decreased use of investigations and medications.Reference Van Cleve and Christakis 38 The overall small numbers of children with bronchiolitis seen at many of our study sites could heighten diagnostic and management uncertainty among the treating physicians. Finally, while we know that seven study centres had CPGs for the management of bronchiolitis, our sample size precluded a subgroup analysis for this variable. Future studies are needed to examine whether introduction of bronchiolitis CPGs within community EDs influences clinical outcomes or resource utilization.
The admission rate we found in our cohort is broadly similar to that reported previously. Admission rates have between reported at 13% among a cohort of patients treated at either a pediatric or a general hospital,Reference Johnson, Robles and Hudgins 39 at 31% among a cohort treated at pediatric emergency departments,Reference Schuh, Lalani and Allen 9 and at 37% among a cohort treated at a general hospital.Reference Johnson, Adair and Brant 26 The variation may reflect differences in illness severity as well as differences in physician comfort in managing bronchiolitis.Reference Johnson, Adair and Brant 26
Two other cohort studies have also reported how commonly children return to the ED for ongoing bronchiolitis symptoms.Reference Plint, Johnson and Wiebe 8 , Reference Norwood, Mansbach and Clark 40 One study, which followed a multicentre cohort seen in Canadian pediatric EDs, found that 17% of children returned to the ED for ongoing symptoms,Reference Plint, Johnson and Wiebe 8 while the second, which followed a multicentre cohort of children seen at 30 EDs across the United States, reported 13% returned to the ED,Reference Norwood, Mansbach and Clark 40 in comparison to about 7% in our study. Bronchiolitis clearly places a significant burden on children and families in terms of admissions to hospitals and the need to seek further ED care, especially given evidence that hospitalization causes significant stress for parents.Reference Lapillonne, Regnault and Gournay 41
Given the practice variation seen in our study regarding the use of treatments and investigations with little evidence of benefit, future work could include evaluation of knowledge translation strategies for the appropriate management of bronchiolitis. The ICCs presented within this paper are important parameters to understand, as they can aid in the design of future cluster randomized trials of knowledge translation strategies. Failure to adjust for the ICC during sample size calculations risks underestimating the required sample size.
Our study has several limitations. Children screened for study inclusion were identified by ICD-10 coding and thus cohort identification was dependent on the diagnosis of site physicians. In an attempt to capture a broad cohort of potentially eligible children, we selected a wide range of diagnostic codes for initial screening, and confirmed through detailed chart review that these children met our definition of bronchiolitis. The retrospective nature of our data collection meant that we were dependent upon nursing and physician documentation. Furthermore, despite this wide range of diagnostic codes for initial screening and large number of participating hospitals, our overall sample size was relatively small at 543 patients. The small sample size likely reflects that many of these centres see few pediatric patients, with almost half seeing less than 5,000 pediatric patients per year. Small sample sizes at many sites also limited our ability to explore site-to-site variation, as the precision of the estimated ICCs may be low in those cases. ICC estimates were not adjusted for patient case mix at the participating hospitals, and may therefore over-estimate the proportion of variation due to differences in management practices. It is our intention that a future study will investigate case-mix adjusted differences in practices in this sample. Further, the hospitals participating in our study were not randomly selected, and participating sites may have different practices than those that did not participate. For example, hospitals that chose to participate may, by their very interest in the study, be more homogenous than those that chose not to participate and higher-performing centres, and our results may in fact underestimate the true extent of variability and lack of adherence to guidelines. Finally, the data from this study reflect the management of patients between 2005 and 2007. It is difficult to know whether practice may have changed significantly since that time, although recent evidence suggests that significant practice variation, unnecessary investigations, and use of ineffective medications persist among pediatric centres.Reference Mittal, Hall and Morse 36 Given the fact there has been no province-wide implementation of a standardized bronchiolitis clinical guideline, we do not suspect a significant change in practice has occurred since the time our data were collected.
Conclusion
Our results provide a better understanding of how children with bronchiolitis are managed in community hospitals. Our results suggest infants with bronchiolitis receive medications and investigations for which there is little evidence of benefit. This suggests a need for knowledge translation strategies directed to community hospitals. The evidence of inter-hospital variation in medication use and laboratory testing suggests an opportunity to explore barriers and facilitators across hospitals in order to design and tailor interventions based on baseline performance.
Competing Interests: The study was funded through the Canadian Institutes of Health Research Team Grant in Pediatric Emergency Medicine (grant 200510CTP-155421-TEB-CBAA-52742). The funding agency played no role in the design, in the collection, analysis, and interpretation of data; in the writing of the manuscript; and in the decision to submit the manuscript for publication. Amy C. Plint holds a University of Ottawa Tier II Clinical Research Chair in Pediatric Emergency Medicine. Jeremy Grimshaw holds a Canada Research Chair in Health Knowledge Transfer and Update. Shannon D. Scott holds a Canada Research Chair (Tier 2) for Knowledge Translation in Child Health and a Population Health Investigator Award from Alberta Innovates Health Solutions.






