Iodine deficiency (ID) is an important micronutrient deficiency, given iodine’s critical role in fetal neurologic development( Reference Zimmermann 1 – Reference Pearce, Lazarus and Moreno-Reyes 4 ). The severe form of ID can cause cretinism, congenital anomalies, intellectual impairment, growth retardation and infant mortality( Reference Bath, Steer and Golding 5 – Reference Zimmermann, Jooste and Pandav 7 ), whereas, even mild ID can impair children’s neuro-intellectual development( Reference Berbel, Mestre and Santamaria 8 – Reference Abel, Caspersen and Meltzer 10 ). Although, there has been substantial progress in the global elimination of ID, lactating mothers and their infants remain susceptible to insufficient iodine intake( Reference Pearce, Andersson and Zimmermann 11 ).
Iodine sufficiency among lactating mothers is particularly important for exclusively breast-fed infants, as breast milk is their only source of iodine during a critical period of growth and development( Reference Azizi and Smyth 12 ). Based on biological mechanisms, it is plausible that urinary iodine concentrations (UIC), which reflect iodine status, are positively associated among mothers and their exclusively breast-fed infants( Reference Azizi and Smyth 12 ). Interestingly, results from Turkey( Reference Simsek, Karabay and Kocabay 13 ), Ireland( Reference Smyth, Hetherton and Smith 14 ), China( Reference Zhang, Xu and Zhan 15 ) and Iran( Reference Nazeri, Mirmiran and Hedayati 16 , Reference Ordookhani, Pearce and Hedayati 17 ) indicate that, the transfer of iodine from mother to infant results is the infant having higher UIC values, it was also accompanied by an increase in the proportion of lactating mothers with inadequate UIC values. Moreover, a few studies that specifically assessed iodine status among formula feeding mothers revealed conflicting findings. For example, one study found no difference in the median UIC values of mothers who were breast-feeding or formula feeding( Reference Smyth, Smith and Sheehan 18 ). However, another study revealed significantly increased UIC values among breast-feeding mothers, compared with formula feeding mothers( Reference Smyth, Hetherton and Smith 14 ).
Neonates and infants receive their entire supply of iodine from breast milk and/or formula. Thus, Europe requires infant formula to contain 10–50 μg of iodine/418·4 kJ (100 kcal), whereas the USA requires approximately 5–75 μg/100kcal. However, despite the importance of iodine during these critical periods, few studies have compared the iodine statuses of breast-fed and formula-fed infants. In addition, studies from areas with iodine sufficiency have revealed inconclusive results. For instance, in the USA, no significant differences in UIC values were detected among infants who were exclusively breast-fed, formula-fed or receiving a combination of breast milk and formula( Reference Gordon, Leung and Hale 19 ). In contrast, the median UIC was significantly lower among Chinese infants who were exclusively formula-fed, compared with those who were exclusively breast-fed( Reference Zhang, Xu and Zhan 15 ). Another study in Switzerland, which is a country with iodine sufficiency, indicated that infants who were exclusively or partially formula-fed had a better iodine status, compared with exclusively breast-fed infants( Reference Andersson, Aeberli and Wust 20 ).
Therefore, based on the limited data and the conflicting results regarding the effect of feeding type on infants’ iodine status in areas with iodine sufficiency, the present study compared the UIC values from breast-fed and formula-fed infants and their mothers in an area that has had iodine sufficiency for more than two decades.
Methods
Subjects
This cross-sectional study was conducted between December 2014 and March 2015 at four randomly selected health care centres in Tehran. Among 140 healthy mothers and their infants who were referred for routine check-ups, the present study included 124 infants who were <3 months old and either exclusively breast-fed or exclusively/partially formula-fed, as well as their mothers. The infants and mothers were enrolled with unequal sample sizes from each health care centre. Mothers and infants with a history of thyroid disorders and those who were currently using iodine-containing supplements and disinfectants were excluded. An interviewer-administered questionnaire was used to collect data regarding maternal age, education, occupation, date of last pregnancy, gravidity, parity, history of abortion and use of iodine-containing supplements during pregnancy. In addition, data were collected regarding the infants’ birth date, sex, birth weight, height and head measurements, type of feeding and brand of infant formula (where applicable). Written informed consent was obtained from the parents after they received an explanation of the study’s protocol and objectives. The study’s protocol was approved by the ethics committee of the Research Institute for Endocrine Sciences (RIES), Shahid Beheshti University of Medical Sciences.
Urine and breast milk sample collection
At the first visit, labelled plastic bottles and adhesive paediatric urine bags (SUPA Medical Services) were provided to collect spot urine samples from each mother and infant, according to the manufacturer’s detailed instructions. Mothers were asked to collect a casual infant urine sample at a random point during the day, after cleaning the infant’s genital region. For boys, the entire penis was placed in the bag and the adhesive was pressed against their skin. For girls, the bag was positioned to cover the labia. If the infant’s urine sample could not be collected after three attempts, the mothers were asked to obtain a sample by holding a specimen bottle in the urine stream. Lactating mothers who were exclusively or partially breast-feeding were asked to manually express their breast milk at the time of urine collection. All samples of urine and breast milk were collected and stored at room temperature, before being sent to the RIES laboratory, where they were transferred into screw-top labelled plastic vials. The aliquots were subsequently frozen at −20°C until the iodine concentrations were measured.
Salt sample collection
Two tablespoon-sized samples of cooking and/or table salt were obtained from each mother at the time of urine collection. Some mothers used two types of salt, and samples of both were collected in those cases. The samples were stored in lightproof, closed plastic containers that were labelled with the mother’s identification code.
Infant formula selection
Information on different types of formula for infants from birth to 6 months of age was obtained from the Food and Drug Administration, Ministry of Health and Medical Education. Based on the list of formula brands currently available in the Tehran markets, the bands most frequently used were selected. From each brand, three samples were purchased from referral pharmacies and sent to the RIES laboratory for iodine testing.
Laboratory measurements
Iodine concentrations in the urine, breast milk and infant formula samples were analysed using the Sandell–Kolthoff (acid-digestion) reaction( Reference Hedayati, Khazan and Yaghmaee 21 ). The results were expressed as μg of iodine per litre of urine and milk, or as μg/100 ml of infant formula. Breast milk and formula samples were carefully homogenised before the alkaline ashing procedure. Intra-assay CV for UIC were 8 % at 8 µg/l, 6 % at 17 µg/l and 8 % at 36 µg/l. The inter-assay CV for UIC were 10 % at 8 µg/l, 10 % at 17 µg/l and 8 % at 36 µg/l. The intra-assay CV for breast milk iodine concentration (BMIC) were 9 % at 3 µg/l, 7 % at 13 µg/l and 9 % at 36 µg/l. The inter-assay CV for BMIC were 10 % at 3 µg/l, 9 % at 13 µg/l and 12 % at 36 µg/l. The intra-assay and inter-assay CV for the infant formula samples were both 9 %. The salt samples’ iodine concentrations in parts per million (ppm) were determined using the iodometric titration method, which has a sensitivity of 1 ppm and a CV of 1 %.
Definitions
Exclusive breast-feeding was defined according to the WHO’s criteria as no non-breast milk food or drink, including water, being used for nourishment. Exclusive formula feeding was defined as the infant only being fed using a standard commercially available cow’s milk-based formula. Partial breast and formula feeding was defined as the infant receiving both breast milk and formula. According to international criteria, median UIC values <100 µg/l and≥100 µg/l were defined as indicating ID and adequacy, respectively, among postpartum mothers and their infants( 22 ). A median BMIC of≥100 µg/l is defined as adequate in areas with iodine sufficiency( Reference Dorea 23 ).
Statistical analysis
Data were reported as numbers and percentages, mean values and standard deviations or medians and interquartile ranges (IQR)), as appropriate. Normality of the variables’ distributions was assessed using the Kolmogorov–Smirnov test and histogram charts. Inter-group comparisons were performed using the χ 2 test, Mann–Whitney test, or t test, as appropriate. Spearman’s correlation analysis was used to examine the correlations between the infants’ UIC values and potentially related variables. ANCOVA was also used to examine the effect of feeding type (exclusive breast-feeding v. partial formula feeding) on the mothers’ and infants’ iodine statuses. All statistical analyses were performed using IBM SPSS software (version 20.0; IBM Corp.), and differences were considered significant at a P value of <0·05. Post hoc power analysis was performed using G*Power software (version 3.1.0) and based on the mean difference in the UIC values according to feeding type. Using an effect size of=0·5 (based on previously reported data)( Reference Ordookhani, Pearce and Hedayati 17 , Reference Simsek, Karabay and Kocabay 13 , Reference Smyth, Smith and Sheehan 18 ), an error probability ratio (β:α) of=4, and the two groups’ sample sizes (breast-feeding: seventy-one cases, formula feeding: fifty-three cases), the present study had an estimated statistical power of 0·787.
Results
The present study included 124 mothers (29·2 (sd 4·9) years old) and 124 infants (2·0 (sd 0·23) months old). Among the infants, 57 % were exclusively breast-fed and 43 % were formula-fed (exclusively or partially). The mothers’ and infants’ characteristics are shown in Table 1. No significant differences were observed between the breast-fed and formula-fed groups in terms of infant sex, birth weight, height and head circumference, weight-for-length z score, or length-for-age z score, as well as maternal age, education, date of last pregnancy, gravidity, parity, history of abortion, or use of iodine- containing supplements during pregnancy. Table 2 shows the UIC and BMIC values for the mothers and infants according to feeding type. The median UIC values were 183 µg/l (IQR 76–285) for breast-fed infants, compared with 140 µg/l (IQR 68–290) for formula-fed infants (P=0·810). The median UIC values were 78 µg/l (IQR 42–145) for breast-feeding mothers, compared with 87 µg/l (IQR 44–159) for formula feeding mothers (P=0·421). Among breast-feeding mothers, UIC values of <100 and <50 µg/l were observed in 68 and 33 %, respectively, compared with 59 and 35 %, respectively, of formula feeding mothers. The median BMIC values were 100 µg/l (IQR 83–147) among breast-feeding mothers, compared with 122 µg/l (IQR 77–172) among formula feeding mothers (P=0·254). The median iodine concentrations in the salt samples were 26 ppm (IQR 20–32) in the breast-feeding group and 25 ppm (IQR 20–29) in the formula feeding group (P=0·155). The iodine content of the selected formula brands (n 9) were 5–18 µg/100 ml, which was often higher than the labelled amount (Table 3), with differences that ranged from −5 to +4 µg/100 ml.
Table 1 Basic characteristic of infants and their mothers by type of feeding (Numbers and percentages; mean values and standard deviations)

* Exclusive breast- feeding.
† Exclusive or partial formula feeding.
Table 2 Urinary and breast milk iodine concentrations in infants and their mothers by type of feeding (Numbers and percentages; medians and interquartile ranges (IQR))
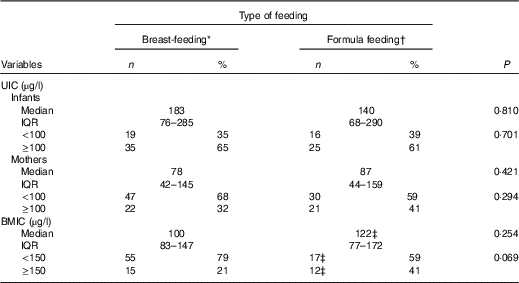
UIC, urinary iodine concentration; BMIC, breast milk iodine concentration.
* Exclusive breast-feeding.
† Exclusive or partial formula feeding.
‡ Data reported from partial formula feeding group.
Table 3 Comparison of labelled and measured iodine content in different brands of infant formula

* Measured using the Sandell–Kolthoff (acid-digestion) reaction.
† Differences of measured iodine concentration compared with labelled iodine concentration based on µg/100 ml formula.
The maternal UIC values were significantly correlated with the BMIC values (r 0·36, P<0·001) and the infants’ UIC values (r 0·21, P=0·04). In addition, the BMIC values were significantly correlated with the infants’ UIC values (r 0·35, P=0·001) (Fig. 1). However, the salt samples’ iodine contents were not significantly correlated with the BMIC or UIC values. Analysis of covariance revealed that BMIC significantly affected the infants’ iodine status (P=0·002). However, after adjustment for BMIC, feeding type (exclusive breast-feeding v. partial formula feeding) did not affect the mothers’ or infants’ UIC values.
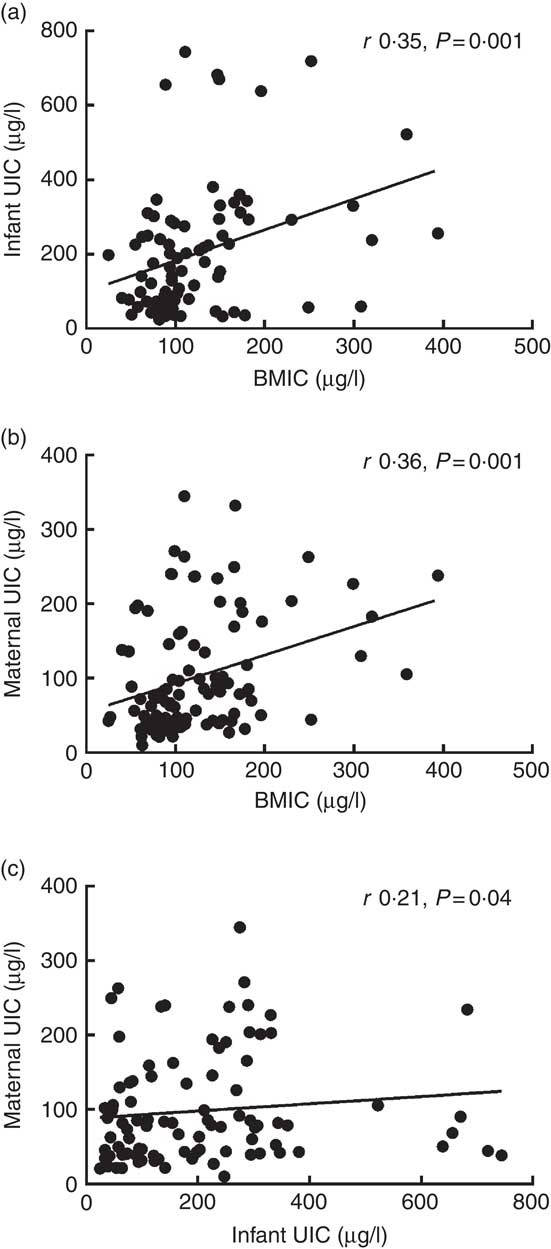
Fig. 1 Correlations between infants' urinary iodine concentration (UIC) and breast milk iodine concentration (BMIC) (a), between maternal UIC and BMIC (b), and between maternal and infants' UIC (c).
Discussion
To the best of our knowledge, the present study is one of the first to compare UIC values between breast-fed and formula-fed infants, as well as their mothers, in Iran, where iodised salt is the main dietary iodine source for all age groups, including lactating mothers and their infants. The present study revealed no significant differences in the mothers’ and infants’ UIC values according to feeding type. Furthermore, although BMIC was significantly correlated with the infants’ iodine status, the feeding type (exclusive breast-feeding v. partial formula feeding) did not affect the infants’ and mothers’ UIC values in the analyses that were adjusted for BMIC.
According to the international criteria, the median UIC in a representative sample of schoolchildren provides a reliable assessment of a population’s iodine status. However, there is significant evidence that pregnant and/or lactating women can have a suboptimal iodine status in areas with iodine sufficiency. For example, iodine sufficiency among school-aged children was not accompanied by adequate UIC values among pregnant and/or lactating women in Switzerland( Reference Andersson, Aeberli and Wust 20 ), Iran( Reference Nazeri, Zarghani and Mirmiran 24 ), Australia( Reference Axford, Charlton and Yeatman 25 ) and New Zealand( Reference Brough, Jin and Shukri 26 ). However, data from different countries have indicated that, in areas with iodine sufficiency or even mild ID, infants’ needs for iodine and thyroid hormones are sufficiently fulfilled using breast milk, because of a compensatory mechanism in the mammary glands that leads to up-regulation of the sodium–iodide symporter and sufficient transfer of iodine from mother to infant( Reference Andersson, Aeberli and Wust 20 , Reference Andersen, Moller and Laurberg 27 – Reference Sukkhojaiwaratkul, Mahachoklertwattana and Poomthavorn 30 ). Present data confirm that iodine adequacy among infants may be accompanied by suboptimal iodine status among lactating mothers, which was observed more in the exclusive breast-feeding group than the formula feeding group. Similarly, a significant proportion of exclusively breast-fed infants and their mothers have ID in India, although the infants’ iodine status is relatively protected( Reference Gupta, Seth and Pandav 31 ). Similar patterns were observed among lactating mothers and their infants in China (122 v. 183 µg/l), Ireland (76 v. 100 µg/l) and Turkey (40 v. 85 µg/l), which indicates that lactating mothers have lower UIC values than their infants( Reference Simsek, Karabay and Kocabay 13 – Reference Zhang, Xu and Zhan 15 ).
Given the broad ranges of BMIC in countries with iodine sufficiency( Reference Azizi and Smyth 12 , Reference Dorea 23 , Reference Semba and Delange 32 , Reference Henjum, Lilleengen and Aakre 33 ), there is no consensus regarding the required iodine levels in breast milk that are needed to adequately fulfil infants’ iodine requirements( Reference Zimmermann, Jooste and Pandav 7 , Reference Swanson and Pearce 34 – Reference Zimmermann 36 ). For example, reported BMIC values are 46–155 µg/l in the USA( Reference Pearce, Leung and Blount 29 , Reference Leung, Braverman and He 37 , Reference Hannan, Faraji and Tanguma 38 ) and 58–170 µg/l in Iran( Reference Ordookhani, Pearce and Hedayati 17 , Reference Bazrafshan, Mohammadian and Ordookhani 39 , Reference Mobasseri, Roshanravan and Mesri Alamdari 40 ). These differences may be related to several factors that can affect BMIC values, including maternal iodine status( Reference Dorea 23 , Reference Semba and Delange 32 , Reference Zimmermann 36 ), recent maternal iodine intake( Reference Leung, Braverman and He 41 ), duration of lactation( Reference Mulrine, Skeaff and Ferguson 42 ) and maternal fluid intake( Reference Andersen, Moller and Laurberg 27 ), of which maternal iodine status plays the most crucial role. In addition, despite the mechanism that enhances iodine excretion in the mother’s mammary glands, different studies have indicated that mothers with suboptimal iodine status have lower BMIC values, compared with mothers with adequate iodine status. For example, lactating mothers with ID have inadequate BMIC values in Australia( Reference Chan, Hams and Wiley 43 ), Turkey( Reference Kurtoglu, Akcakus and Kocaoglu 44 ) and Portugal( Reference Costeira, Oliveira and Ares 45 ), whereas lactating mothers in Iran( Reference Ordookhani, Pearce and Hedayati 17 , Reference Hashemipour, Nasri and Hovsepian 46 ) and the USA( Reference Pearce, Leung and Blount 29 ) (areas with iodine sufficiency) have BMIC values that are within the optimal range. Similarly, a recent study by Dold et al. ( Reference Dold, Zimmermann and Aboussad 47 ) revealed that lactating mothers with an adequate iodine status had greater fractional iodine excretion in their breast milk, compared with their urine, although a relatively constant proportion (approximately 33 %) of total iodine was excreted in breast milk from mothers with ID. There is some evidence that BMIC can be used to monitor the iodine status of breast-fed infants( Reference Dold, Baumgartner and Zeder 48 ), although this relationship remains speculative, given the limited number of relevant studies. The present finding is compatible with our previous reports that revealed a significant positive correlation between BMIC and infants’ UIC values( Reference Ordookhani, Pearce and Hedayati 17 , Reference Bazrafshan, Mohammadian and Ordookhani 39 , Reference Hashemipour, Nasri and Hovsepian 46 ). Furthermore, in the USA and China, iodine sufficiency among infants was also accompanied by adequate iodine levels in breast milk( Reference Zhang, Xu and Zhan 15 , Reference Gordon, Leung and Hale 19 ).
Only a few studies have compared the iodine contents of breast milk and infant formula, and related those findings to infants’ iodine status according to feeding type in areas with iodine sufficiency. A recent report from Switzerland indicates that partially breast-fed and formula-fed infants had higher UIC values than infants who were exclusively breast-fed (109 v. 73 µg/l), which was attributed to the higher iodine content of infant formula (45–148 µg/l), compared with breast milk (40–80 µg/l)( Reference Andersson, Aeberli and Wust 20 ). The present study revealed that the iodine content of infant formula was 50–184 µg/l, whereas the median BMIC values were 100 µg/l (40–320 µg/l) in the exclusive breast-feeding group and 122 µg/l (25–394 µg/l) in the group that received breast milk and formula. In addition, although the difference was not significant, exclusively breast-fed infants had higher UIC values, compared with infants who received formula. Similarly, an American study( Reference Pearce, Leung and Blount 29 ) revealed no significant difference between the median iodine concentrations of breast milk and infant formula (155 µg/l, n 57 v. 145 µg/l, n 17). It also found similar iodine statuses among infants who were exclusively breast-fed (203 µg/l, n 39), formula-fed (182·5 µg/l, n 44) or received breast milk and formula (198 µg/l, n 10)( Reference Gordon, Leung and Hale 19 ).
The main strength of the present study is the simultaneous assessment of iodine status among breast-fed and formula-fed infants, as well as their mothers, in an area with a successful salt iodisation programme. However, the present study only evaluated individuals from randomly selected regions of Tehran, and the data may not be representative of the entire city. Therefore, caution should be taken in generalising these findings to other areas and populations. In addition, the use of a single urine sample is a poor indicator of long-term iodine status( Reference Konig, Andersson and Hotz 49 ), as intra-individual variability could be introduced through maternal dietary intake of iodine (i.e. iodised salt in Iran), maternal hydration and the iodine content and amount of the formula and/or breast milk that the infant receives. Furthermore, programmes promoting the importance of breast-feeding, as well as low family incomes and relatively high formula prices, resulted in a limited number of mothers who exclusively fed their children formula (n 15). Thus, categorisation of the mothers into groups that exclusively breast-fed, partially formula fed, and exclusively formula fed could lead to inadequately powered statistical analyses.
In conclusion, the present study revealed that in Tehran (an area with iodine sufficiency based on the median UIC from the WHO), there were no differences in infants’ or mothers’ urinary iodine levels according to the feeding type and the iodine statuses of mothers and their infants were not affected by the type of feeding. Thus, both breast milk with adequate iodine levels and infant formula that is supplemented with iodine can fulfil infants’ iodine requirements during the first 6 months of life.
Acknowledgements
We would like to thank the mothers and their infants who participated in the study.
This study was supported by financial grant from the Research Institute of Endocrine Sciences, Shahid Beheshti University of Medical Sciences.
P. N. contributed to the design, data analysis and writing of the manuscript; H. D. contributed to the design, writing, reading and final approval of the manuscript; Y. M. contributed to the statistical analysis and writing of the manuscript; M. H. contributed to the design of the study and writing of the manuscript; P. M. and F. A. contributed to the design of the study, writing, reading and final approval of the manuscript.
The authors declare that there are no conflicts of interest.









