Book contents
- The Brain and Behavior
- The Brain and Behavior
- Copyright page
- Dedication
- Contents
- Preface to the Fourth Edition
- Abbreviations
- Chapter 1 Introduction
- Chapter 2 Anatomy of the Gross Brain
- Chapter 3 Histology
- Chapter 4 Occipital and Parietal Lobes
- Chapter 5 Temporal Lobe: Neocortical Structures
- Chapter 6 Frontal Lobe
- Chapter 7 Basal Ganglia
- Chapter 8 Diencephalon: Hypothalamus and Epithalamus
- Chapter 9 Diencephalon: Thalamus
- Chapter 10 Brainstem
- Chapter 11 Limbic System: Temporal Lobe
- Chapter 12 Limbic System: Cingulate Cortex
- Chapter 13 Asymmetry and Interhemispheric Connections
- Index
- References
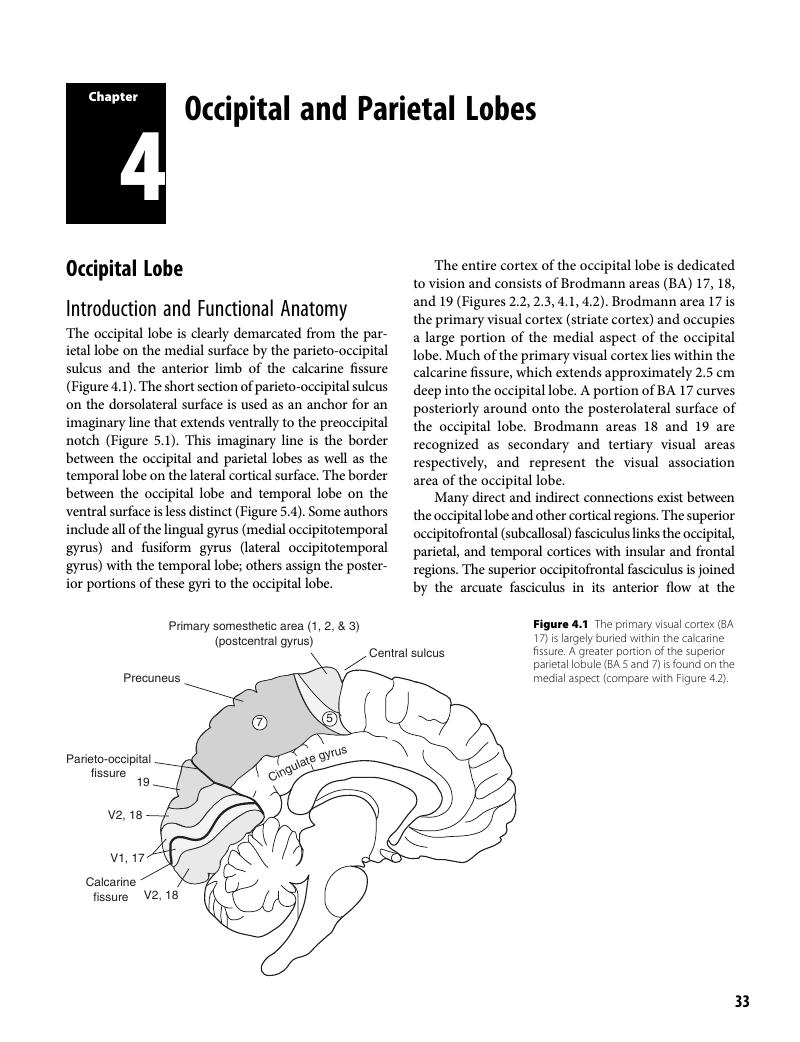
Chapter 4 - Occipital and Parietal Lobes
Published online by Cambridge University Press: 22 February 2018
- The Brain and Behavior
- The Brain and Behavior
- Copyright page
- Dedication
- Contents
- Preface to the Fourth Edition
- Abbreviations
- Chapter 1 Introduction
- Chapter 2 Anatomy of the Gross Brain
- Chapter 3 Histology
- Chapter 4 Occipital and Parietal Lobes
- Chapter 5 Temporal Lobe: Neocortical Structures
- Chapter 6 Frontal Lobe
- Chapter 7 Basal Ganglia
- Chapter 8 Diencephalon: Hypothalamus and Epithalamus
- Chapter 9 Diencephalon: Thalamus
- Chapter 10 Brainstem
- Chapter 11 Limbic System: Temporal Lobe
- Chapter 12 Limbic System: Cingulate Cortex
- Chapter 13 Asymmetry and Interhemispheric Connections
- Index
- References
Summary
- Type
- Chapter
- Information
- The Brain and BehaviorAn Introduction to Behavioral Neuroanatomy, pp. 33 - 55Publisher: Cambridge University PressPrint publication year: 2018
References
Select Bibliography
References
- 1
- Cited by

